PHENERGAN Injection (promethazine hydrochloride Injection, USP) Rx only
PHENERGAN by
Drug Labeling and Warnings
PHENERGAN by is a Prescription medication manufactured, distributed, or labeled by REMEDYREPACK INC.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
PHENERGAN- promethazine hydrochloride injection
REMEDYREPACK INC.
----------
PHENERGAN Injection (promethazine hydrochloride Injection, USP)
Rx only
WARNINGS
Respiratory Depression – Pediatrics
PHENERGAN Injection should not be used in pediatric patients less than 2 years of age because of the potential for fatal respiratory depression. Postmarketing cases of respiratory depression, including fatalities, have been reported with use of promethazine in pediatric patients less than 2 years of age. Caution should be exercised when administering PHENERGAN Injection to pediatric patients 2 years of age and older (seeWARNINGS - Respiratory Depression).
Severe Tissue Injury, Including Gangrene
PHENERGAN Injection can cause severe chemical irritation and damage to tissues regardless of the route of administration. Irritation and damage can result from perivascular extravasation, unintentional intra-arterial injection, and intraneuronal or perineuronal infiltration. Adverse reactions include burning, pain, thrombophlebitis, tissue necrosis, and gangrene. In some cases, surgical intervention, including fasciotomy, skin graft, and/or amputation have been required (see WARNINGS - Severe Tissue Injury, Including Gangrene).
Due to the risks of intravenous injection, the preferred route of administration of PHENERGAN Injection is deep intramuscular injection. Subcutaneous injection is contraindicated. SeeDOSAGE AND ADMINISTRATIONfor important notes on administration.
DESCRIPTION
PHENERGAN Injection (promethazine hydrochloride injection, USP), is a sterile, pyrogen-free solution for deep intramuscular or intravenous administration. Promethazine hydrochloride (10 H-Phenothiazine-10-ethanamine, N,N,α-trimethyl-, monohydrochloride, (±)-) is a racemic compound and has the following structural formula:
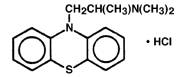
C 17H 21ClN 2S MW 320.88
Each mL contains promethazine hydrochloride, either 25 mg or 50 mg, edetate disodium 0.1 mg, calcium chloride 0.04 mg, sodium metabisulfite 0.25 mg and phenol 5 mg in Water for Injection. pH 4.0 to 5.5; buffered with acetic acid-sodium acetate.
PHENERGAN Injection (promethazine hydrochloride injection, USP) is a clear, colorless solution. The product is light sensitive. It should be inspected before use and discarded if either color or particulate is observed.
CLINICAL PHARMACOLOGY
Promethazine hydrochloride is a phenothiazine derivative which possesses antihistaminic, sedative, antimotion-sickness, antiemetic, and anticholinergic effects. Promethazine is a competitive H 1receptor antagonist, but does not block the release of histamine. Structural differences from the neuroleptic phenothiazines result in its relative lack (1 /10 that of chlorpromazine) of dopamine antagonist properties. Clinical effects are generally apparent within 5 minutes of an intravenous injection and within 20 minutes of an intramuscular injection. Duration of action is four to six hours, although effects may persist up to 12 hours. Promethazine hydrochloride is metabolized in the liver, with the sulfoxides of promethazine and N-desmethylpromethazine being the predominant metabolites appearing in the urine. Following intravenous administration in healthy volunteers, the plasma half-life for promethazine has been reported to range from 9 to 16 hours. The mean plasma half-life for promethazine after intramuscular administration in healthy volunteers has been reported to be 9.8 ± 3.4 hours.
INDICATIONS AND USAGE
PHENERGAN Injection is indicated for the following conditions:
- Amelioration of allergic reactions to blood or plasma.
- In anaphylaxis as an adjunct to epinephrine and other standard measures after the acute symptoms have been controlled.
- For other uncomplicated allergic conditions of the immediate type when oral therapy is impossible or contraindicated.
- For sedation and relief of apprehension and to produce light sleep from which the patient can be easily aroused.
- Active treatment of motion sickness.
- Prevention and control of nausea and vomiting associated with certain types of anesthesia and surgery.
- As an adjunct to analgesics for the control of postoperative pain.
- Preoperative, postoperative, and obstetric (during labor) sedation.
- Intravenously in special surgical situations, such as repeated bronchoscopy, ophthalmic surgery, and poor-risk patients, with reduced amounts of meperidine or other narcotic analgesic as an adjunct to anesthesia and analgesia.
CONTRAINDICATIONS
Children Less Than 2 Years of Age
PHENERGAN Injection is contraindicated for use in pediatric patients less than two years of age due to the risk of respiratory depression (see WARNINGS - Respiratory Depression).
Intra-Arterial Injection
Under no circumstances should PHENERGAN Injection be given by intra-arterial injection due to the likelihood of severe arteriospasm and the possibility of resultant gangrene (see WARNINGS - Severe Tissue Injury, Including Gangrene).
WARNINGS
Respiratory Depression
Pediatrics
PHENERGAN Injection should not be used in pediatric patients less than 2 years of age because of the potential for fatal respiratory depression. Postmarketing cases of respiratory depression, including fatalities, have been reported with use of promethazine in pediatric patients less than 2 years of age. A wide range of weight-based doses of PHENERGAN Injection have resulted in respiratory depression in these patients.
Caution should be exercised when administering PHENERGAN Injection to pediatric patients 2 years of age and older. It is recommended that the lowest effective dose of PHENERGAN Injection be used in pediatric patients 2 years of age and older. Avoid concomitant administration of other drugs with respiratory depressant effects because of an association with respiratory depression, and sometimes death, in pediatric patients.
Severe Tissue Injury, Including Gangrene
PHENERGAN Injection can cause severe chemical irritation and damage to tissues regardless of the route of administration. Irritation and damage can result from perivascular extravasation, unintentional intra-arterial injection, and intraneuronal or perineuronal infiltration. Adverse event reports include burning, pain, erythema, swelling, sensory loss, palsies, paralysis, severe spasm of distal vessels, thrombophlebitis, venous thrombosis, phlebitis, abscesses, tissue necrosis, and gangrene. In some cases, surgical intervention, including fasciotomy, skin graft, and/or amputation have been required.
Because of the risks of intravenous injection, the preferred route of administration of PHENERGAN Injection is deep intramuscular injection (seeDOSAGE AND ADMINISTRATION). Subcutaneous injection is contraindicated. Due to the close proximity of arteries and veins in the areas most commonly used for intravenous injection, extreme care should be exercised to avoid perivascular extravasation or unintentional intra-arterial injection as pain, severe chemical irritation, severe spasm of distal vessels, and resultant gangrene requiring amputation are likely under such circumstances. Aspiration of dark blood does not preclude intra-arterial needle placement because blood is discolored upon contact with PHENERGAN Injection. Use of syringes with rigid plungers or of small-bore needles might obscure typical arterial backflow if this is relied upon alone.
In the event that a patient complains of pain during intravenous injection of Phenergan Injection, the injection should be stopped immediately to evaluate for possible arterial injection or perivascular extravasation.
There is no proven successful management of unintentional intra-arterial injection or perivascular extravasation after it occurs. Sympathetic block and heparinization have been employed during the acute management of unintentional intra-arterial injection, because of the results of animal experiments with other known arteriolar irritants.
CNS Depression
PHENERGAN Injection may impair the mental and/or physical abilities required for the performance of potentially hazardous tasks, such as driving a vehicle or operating machinery. The impairment may be amplified by concomitant use of other central-nervous-system depressants such as alcohol, sedative/hypnotics (including barbiturates), general anesthetics, narcotics, narcotic analgesics, tricyclic antidepressants, and tranquilizers; therefore such agents should either be eliminated or given in reduced dosage in the presence of promethazine hydrochloride (see PRECAUTIONS - Information for Patientsand Drug Interactions).
Lower Seizure Threshold
PHENERGAN Injection may lower seizure threshold and should be used with caution in persons with seizure disorders or in persons who are using concomitant medications, such as narcotics or local anesthetics, which may also affect seizure threshold.
Bone-Marrow Depression
PHENERGAN Injection should be used with caution in patients with bone-marrow depression. Leukopenia and agranulocytosis have been reported, usually when promethazine hydrochloride has been used in association with other known marrow-toxic agents.
Neuroleptic Malignant Syndrome
A potentially fatal symptom complex sometimes referred to as Neuroleptic Malignant Syndrome (NMS) has been reported in association with promethazine hydrochloride alone or in combination with antipsychotic drugs. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis and cardiac dysrhythmias).
The diagnostic evaluation of patients with this syndrome is complicated. In arriving at a diagnosis, it is important to identify cases where the clinical presentation includes both serious medical illness (e.g., pneumonia, systemic infection, etc.) and untreated or inadequately treated extrapyramidal signs and symptoms (EPS). Other important considerations in the differential diagnosis include central anticholinergic toxicity, heat stroke, drug fever and primary central nervous system (CNS) pathology.
The management of NMS should include 1) immediate discontinuation of promethazine hydrochloride, antipsychotic drugs, if any, and other drugs not essential to concurrent therapy, 2) intensive symptomatic treatment and medical monitoring, and 3) treatment of any concomitant serious medical problems for which specific treatments are available. There is no general agreement about specific pharmacological treatment regimens for uncomplicated NMS.
Since recurrences of NMS have been reported with phenothiazines, the reintroduction of promethazine hydrochloride should be carefully considered.
Sulfite Sensitivity
PHENERGAN Injection contains sodium metabisulfite, a sulfite that may cause allergic-type reactions, including anaphylactic symptoms and life-threatening or less severe asthma episodes, in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in nonasthmatic people.
PRECAUTIONS
General
Drugs having anticholinergic properties should be used with caution in patients with narrow-angle glaucoma, prostatic hypertrophy, stenosing peptic ulcer, pyloroduodenal obstruction, and bladder-neck obstruction.
PHENERGAN Injection should be used cautiously in persons with cardiovascular disease or impairment of liver function.
Information for Patients
Patients should be advised of the risk of respiratory depression, including potentially fatal respiratory depression in children less than 2 years of age (see WARNINGS - Respiratory Depression).
Patients should be advised of the risk of severe tissue injury, including gangrene (see WARNINGS - Severe Tissue Injury, Including Gangrene). Patients should be advised to immediately report persistent or worsening pain or burning at the injection site.
PHENERGAN Injection may cause marked drowsiness or impair the mental or physical abilities required for the performance of potentially hazardous tasks, such as driving a vehicle or operating machinery. Pediatric patients should be supervised to avoid potential harm in bike riding or in other hazardous activities. The concomitant use of alcohol, sedative/hypnotics (including barbiturates), general anesthetics, narcotics, narcotic analgesics, tricyclic antidepressants, and tranquilizers may enhance impairment (see WARNINGS - CNS Depressionand PRECAUTIONS - Drug Interactions).
Patients should be advised to report any involuntary muscle movements (see ADVERSE REACTIONS - Paradoxical Reactions).
Patients should be advised to avoid prolonged exposure to the sun (see ADVERSE REACTIONS - Dermatologic).
Repackaged By / Distributed By: RemedyRepack Inc.
625 Kolter Drive, Indiana, PA 15701
(724) 465-8762
Drug Interactions
CNS Depressants
PHENERGAN Injection may increase, prolong, or intensify the sedative action of central-nervous-system depressants, such as alcohol, sedative/hypnotics (including barbiturates), general anesthetics, narcotics, narcotic analgesics, tricyclic antidepressants, and tranquilizers; therefore, such agents should be avoided or administered in reduced dosage to patients receiving promethazine hydrochloride. When given concomitantly with PHENERGAN Injection, the dose of barbiturates should be reduced by at least one-half, and the dose of narcotics should be reduced by one-quarter to one-half. Dosage must be individualized. Excessive amounts of PHENERGAN Injection relative to a narcotic may lead to restlessness and motor hyperactivity in the patient with pain; these symptoms usually disappear with adequate control of the pain.
Epinephrine
Because of the potential for promethazine hydrochloride to reverse epinephrine’s vasopressor effect, epinephrine should NOT be used to treat hypotension associated with PHENERGAN Injection overdose.
Drug/Laboratory Test Interactions
The following laboratory tests may be affected in patients who are receiving therapy with PHENERGAN Injection:
Carcinogenesis, Mutagenesis and Impairment of Fertility
Long-term animal studies have not been performed to assess the carcinogenic potential of PHENERGAN Injection, nor are there other animal or human data concerning carcinogenicity, mutagenicity, or impairment of fertility. PHENERGAN Injection was nonmutagenic in the Salmonellatest system of Ames.
Pregnancy
Teratogenic Effects—Pregnancy Category C
Teratogenic effects have not been demonstrated in rat-feeding studies at doses of 6.25 and 12.5 mg/kg (approximately 2.1 and 4.2 times the maximum recommended human daily dose) of PHENERGAN Injection. Daily doses of 25 mg/kg intraperitoneally have been found to produce fetal mortality in rats.
There are no adequate and well-controlled studies of PHENERGAN Injection in pregnant women. Because animal reproduction studies are not always predictive of human response, PHENERGAN Injection should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Adequate studies to determine the action of the drug on parturition, lactation and development of the animal neonate have not been conducted.
Labor and Delivery
PHENERGAN Injection may be used alone or as an adjunct to narcotic analgesics during labor (see DOSAGE AND ADMINISTRATION). Limited data suggest that use of PHENERGAN Injection during labor and delivery does not have an appreciable effect on the duration of labor or delivery and does not increase the risk of need for intervention in the newborn. The effect on later growth and development of the newborn is unknown. (See also Pregnancy - Nonteratogenic Effects.)
Nursing Mothers
It is not known whether PHENERGAN Injection is excreted in human milk. Because many drugs are excreted in human milk, and because of the potential for serious adverse reactions in nursing infants from PHENERGAN Injection, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
PHENERGAN Injection is contraindicated for use in pediatric patients less than 2 years of age, because of the potential for fatal respiratory depression. PHENERGAN Injection should be used with caution in pediatric patients 2 years of age and older (see WARNINGS - Respiratory Depression).
Antiemetics are not recommended for treatment of uncomplicated vomiting in pediatric patients, and their use should be limited to prolonged vomiting of known etiology. The extrapyramidal symptoms which can occur secondary to PHENERGAN Injection administration may be confused with the CNS signs of undiagnosed primary disease, e.g. encephalopathy or Reye's syndrome. The use of PHENERGAN Injection should be avoided in pediatric patients whose signs and symptoms may suggest Reye's syndrome or other hepatic diseases.
Excessively large dosages of antihistamines, including PHENERGAN Injection, in pediatric patients may cause sudden death (see OVERDOSAGE). Hallucinations and convulsions have occurred with therapeutic doses and overdoses of PHENERGAN Injection in pediatric patients. In pediatric patients who are acutely ill associated with dehydration, there is an increased susceptibility to dystonias with the use of PHENERGAN Injection.
ADVERSE REACTIONS
Respiratory Depression
PHENERGAN Injection is contraindicated in pediatric patients less than 2 years of age, because of the potential for fatal respiratory depression. PHENERGAN Injection should be used with caution in pediatric patients 2 years of age and older (see WARNINGS - Respiratory Depression).
Severe Tissue Injury, Including Gangrene
PHENERGAN Injection can cause severe chemical irritation and damage to tissues regardless of the route of administration. Irritation and damage can result from perivascular extravasation, unintentional intra-arterial injection, and intraneuronal or perineuronal infiltration. Adverse reactions include burning, pain, erythema, swelling, sensory loss, palsies, paralysis, severe spasm of distal vessels, thrombophlebitis, venous thrombosis, phlebitis, abscesses, tissue necrosis, and gangrene. In some cases, surgical intervention, including fasciotomy, skin graft, and/or amputation have been required (see WARNINGS - Severe Tissue Injury, Including Gangrene; and DOSAGE AND ADMINISTRATION).
Central Nervous System
Drowsiness is the most prominent CNS effect of this drug. Sedation, somnolence, blurred vision, dizziness, confusion, disorientation, and extrapyramidal symptoms such as oculogyric crisis, torticollis, and tongue protrusion; lassitude, tinnitus, incoordination, fatigue, euphoria, nervousness, diplopia, insomnia, tremors, convulsive seizures, excitation, catatonic-like states, hysteria. Hallucinations have also been reported.
Respiratory
Asthma, nasal stuffiness, respiratory depression (potentially fatal) and apnea (potentially fatal). (See WARNINGS - Respiratory Depression.)
Other
Angioneurotic edema. Neuroleptic Malignant Syndrome (potentially fatal) has also been reported. (See WARNINGS - Neuroleptic Malignant Syndrome.)
Paradoxical Reactions
Hyperexcitability and abnormal movements have been reported in patients following a single administration of PHENERGAN Injection. Consideration should be given to the discontinuation of PHENERGAN Injection and to the use of other drugs if these reactions occur. Respiratory depression, nightmares, delirium, and agitated behavior have also been reported in some of these patients.
OVERDOSAGE
Signs and symptoms of overdosage range from mild depression of the central nervous system and cardiovascular system to profound hypotension, respiratory depression, unconsciousness and sudden death. Other reported reactions include hyperreflexia, hypertonia, ataxia, athetosis, and extensor-plantar reflexes (Babinski reflex).
Stimulation may be evident, especially in pediatric patients and geriatric patients. Convulsions may rarely occur. A paradoxical-type reaction has been reported in pediatric patients receiving single doses of 75 mg to 125 mg orally, characterized by hyperexcitability and nightmares.
Atropine-like signs and symptoms-dry mouth; fixed, dilated pupils; flushing; etc., as well as gastrointestinal symptoms, may occur.
Treatment
Treatment of overdosage is essentially symptomatic and supportive. Only in cases of extreme overdosage or individual sensitivity do vital signs, including respiration, pulse, blood pressure, temperature, and EKG, need to be monitored. Attention should be given to the reestablishment of adequate respiratory exchange through provision of a patent airway and institution of assisted or controlled ventilation. Diazepam may be used to control convulsions. Acidosis and electrolyte losses should be corrected. Note that any depressant effects of PHENERGAN Injection are not reversed by naloxone.
Avoid analeptics, which may cause convulsions. The treatment of choice for resulting hypotension is administration of intravenous fluids, accompanied by repositioning if indicated. In the event that vasopressors are considered for the management of severe hypotension which does not respond to intravenous fluids and repositioning, the administration of norepinephrine or phenylephrine should be considered. EPINEPHRINE SHOULD NOT BE USED, since its use in a patient with partial adrenergic blockade may further lower the blood pressure. Extrapyramidal reactions may be treated with anticholinergic antiparkinson agents, diphenhydramine, or barbiturates. Oxygen may also be administered. Limited experience with dialysis indicates that it is not helpful.
DOSAGE AND ADMINISTRATION
Important Notes on Administration
PHENERGAN Injection can cause severe chemical irritation and damage to tissues regardless of the route of administration. Irritation and damage can result from perivascular extravasation, unintentional intra-arterial injection, and intraneuronal or perineuronal infiltration (see WARNINGS - Severe Tissue Injury, Including Gangrene).
- The preferred parenteral route of administration for PHENERGAN Injection is by deep intramuscular injection.
- Under no circumstances should PHENERGAN Injection be given by intra-arterial injection due to the likelihood of severe arteriospasm and the possibility of resultant gangrene (see WARNINGS - Severe Tissue Injury, Including Gangrene).
- Subcutaneous injection is contraindicated as it may result in tissue necrosis.
- When administered intravenously, PHENERGAN Injection should be given in a concentration no greater than 25 mg per mL and at a rate not to exceed 25 mg per minute.It is preferable to inject through the tubing of an intravenous infusion set that is known to be functioning satisfactorily.
- In the event that a patient complains of pain during intravenous injection of PHENERGAN Injection, the injection should be stopped immediately to evaluate for possible arterial injection or perivascular extravasation.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
Do not use PHENERGAN Injection if solution has developed color or contains precipitate.
To avoid the possibility of physical and/or chemical incompatibility, consult specialized literature before diluting with any injectable solution or combining with any other medication. Do not use if there is a precipitate or any sign of incompatibility.
Allergic Conditions
The average adult dose is 25 mg. This dose may be repeated within two hours if necessary, but continued therapy, if indicated, should be via the oral route as soon as existing circumstances permit. After initiation of treatment, dosage should be adjusted to the smallest amount adequate to relieve symptoms. The average adult dose for amelioration of allergic reactions to blood or plasma is 25 mg.
Sedation
In hospitalized adult patients, nighttime sedation may be achieved by a dose of 25 to 50 mg of PHENERGAN Injection.
Nausea and Vomiting
For control of nausea and vomiting, the usual adult dose is 12.5 to 25 mg, not to be repeated more frequently than every four hours. When used for control of postoperative nausea and vomiting, the dosage of analgesics and barbiturates should be reduced accordingly (see PRECAUTIONS – Drug Interactions).
Antiemetics should not be used in vomiting of unknown etiology in children and adolescents (see PRECAUTIONS – Pediatric Use).
Preoperative and Postoperative Use
As an adjunct to preoperative or postoperative medication, 25 to 50 mg of PHENERGAN Injection in adults may be combined with appropriately reduced doses of analgesics and atropine-like drugs as desired. Dosage of concomitant analgesic or hypnotic medication should be reduced accordingly (see PRECAUTIONS – Drug Interactions).
PHENERGAN is contraindicated for use in pediatric patients less than two years of age.
Obstetrics
PHENERGAN Injection in doses of 50 mg will provide sedation and relieve apprehension in the early stages of labor. When labor is definitely established, 25 to 75 mg (average dose, 50 mg) PHENERGAN Injection may be given with an appropriately reduced dose of any desired narcotic (see PRECAUTIONS – Drug Interactions). If necessary, PHENERGAN Injection with a reduced dose of analgesic may be repeated once or twice at four-hour intervals in the course of a normal labor. A maximum total dose of 100 mg of PHENERGAN Injection may be administered during a 24-hour period to patients in labor.
Pediatric Patients
PHENERGAN Injection is contraindicated for use in pediatric patients less than 2 years of age (seeWARNINGS – Respiratory Depression).Caution should be exercised when administering promethazine hydrochloride to pediatric patients 2 years of age or older. It is recommended that the lowest effective dose of promethazine hydrochloride be used in pediatric patients 2 years of age and older and concomitant administration of other drugs with respiratory depressant effects be avoided (seeWARNINGS – Respiratory Depression).
In pediatric patients 2 years of age and older, the dosage should not exceed half that of the suggested adult dose. As an adjunct to premedication, the suggested dose is 1.1 mg per kg of body weight in combination with an appropriately reduced dose of narcotic or barbiturate and the appropriate dose of an atropine-like drug (see PRECAUTIONS – Drug Interactions). Antiemetics should not be used in vomiting of unknown etiology in pediatric patients.
HOW SUPPLIED
PHENERGAN Injection (promethazine hydrochloride injection, USP) is available as follows:
25 mg/mL
NDC: 70518-2656-00
NDC: 70518-2656-01
PACKAGING: 25 in 1 CARTON
PACKAGING: 1 mL in 1 VIAL, TYPE 0
Storage
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Protect from light. Keep covered in carton until time of use.
Do not use if solution has developed color or contains a precipitate.
Repackaged and Distributed By:
Remedy Repack, Inc.
625 Kolter Dr. Suite #4 Indiana, PA 1-724-465-8762
DRUG: PHENERGAN
GENERIC: Promethazine Hydrochloride
DOSAGE: INJECTION
ADMINSTRATION: INTRAMUSCULAR
NDC: 70518-2656-0
NDC: 70518-2656-1
PACKAGING: 1 mL in 1 VIAL
OUTER PACKAGING: 25 in 1 CARTON
ACTIVE INGREDIENT(S):
- PROMETHAZINE HYDROCHLORIDE 25mg in 1mL
INACTIVE INGREDIENT(S):
- EDETATE DISODIUM
- CALCIUM CHLORIDE
- PHENOL
- WATER
- ACETIC ACID
- SODIUM ACETATE


| PHENERGAN
promethazine hydrochloride injection |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - REMEDYREPACK INC. (829572556) |
Trademark Results [PHENERGAN]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 PHENERGAN 98588669 not registered Live/Pending |
Rising Pharma Holdings, Inc. 2024-06-06 |
 PHENERGAN 97852996 not registered Live/Pending |
WOODWARD SPECIALTY LLC 2023-03-23 |
 PHENERGAN 77909935 3818335 Live/Registered |
DELCOR ASSET CORPORATION 2010-01-12 |
 PHENERGAN 71550837 0507057 Live/Registered |
SOCIETE DES USINES CHIMIQUES RHONE-POULENC 1948-02-28 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.