Omnilux Mineral Facial Sunscreen
Omnilux Mineral Facial Sunscreen by
Drug Labeling and Warnings
Omnilux Mineral Facial Sunscreen by is a Otc medication manufactured, distributed, or labeled by The Lotus Global Group, Inc, dba GlobalMed Technologies, Nanophase Technologies Corporation. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
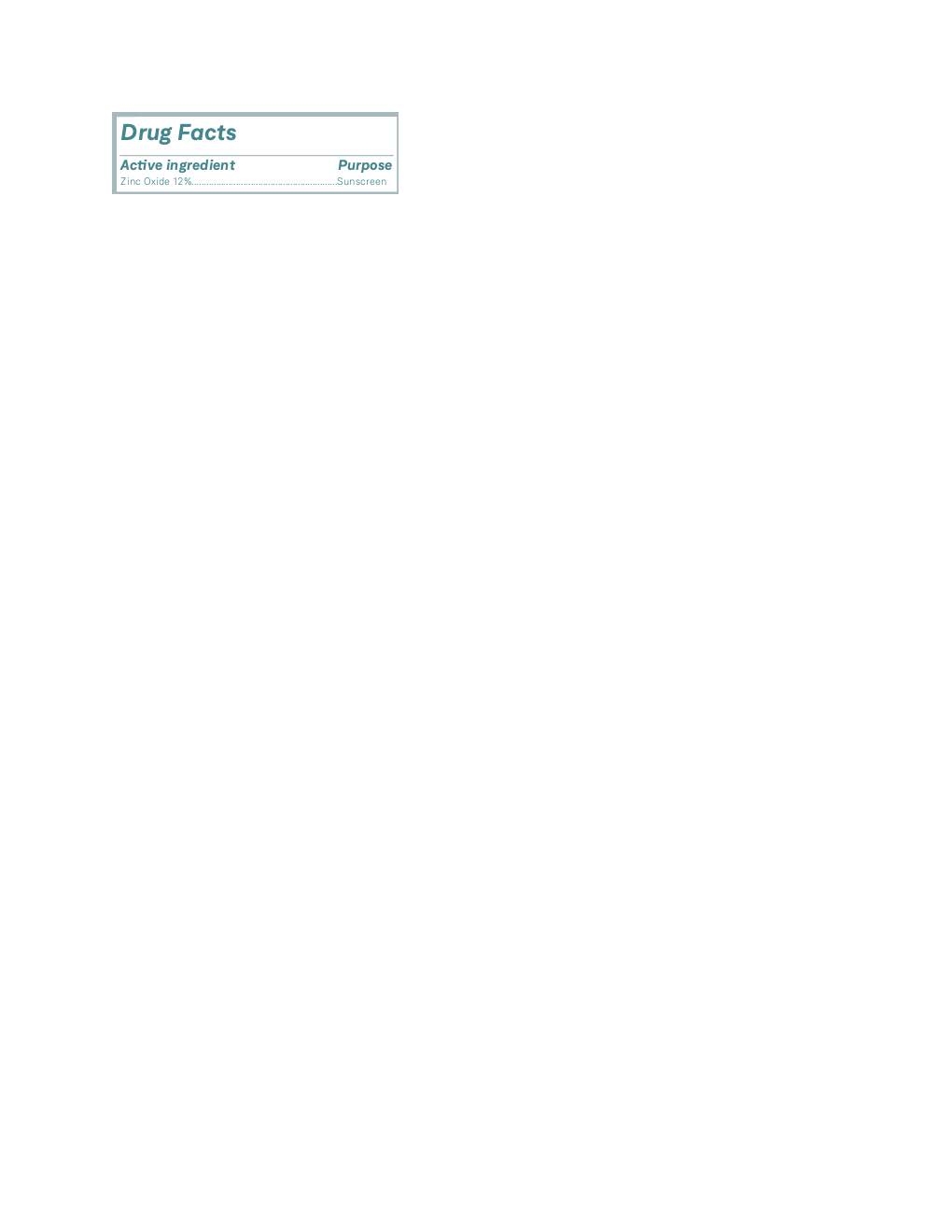
OMNILUX MINERAL FACIAL SUNSCREEN- zinc oxide lotion
The Lotus Global Group, Inc, dba GlobalMed Technologies
----------
Omnilux Mineral Facial Sunscreen
| OMNILUX MINERAL FACIAL SUNSCREEN
zinc oxide lotion |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - The Lotus Global Group, Inc, dba GlobalMed Technologies (969797406) |
Revised: 1/2026
Document Id: 47e1b275-7274-a3c0-e063-6294a90adb1c
Set id: 1fab0031-59f8-36cd-e063-6394a90a8aef
Version: 2
Effective Time: 20260108
The Lotus Globa
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.