UNDA 700- berberis vulgaris, calcarea fluorica liquid UNDA 312- angelica archangelica, drosera, grindelia, lamium album, pinus sylvestris, rubus fruticosus, senega officinalis liquid
UNDA 312 by
Drug Labeling and Warnings
UNDA 312 by is a Homeopathic medication manufactured, distributed, or labeled by Seroyal USA, SAN’UP. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
ACTIVE INGREDIENT
Active ingredients
Each drop contains:
Angelica archangelica (European angelica) Root 6X
Drosera (Sundew) Whole Plant 6X
Grindelia (Gum-plant) Aerial Parts 6X
Lamium album (Dead nettle) Aerial Parts 6X
Pinus sylvestris (Scotch pine) Shoots 6X
Rubus fruticosus (Shrubby blackberry) Leaf 6X
Senega officinalis (Snakeroot) Root 6X - PURPOSE
-
WARNINGS
Warnings
Sore throat warning: Severe or persistent sore throat or sore throat accompanied by high fever, headache,
nausea and vomiting may be serious.
Consult physician promptly.Do not use more than 2 days or administer to children under 3 years of age unless directed by physician.
Ask a doctor before use if you have Persistent or chronic cough such as occurs with smoking, asthma, chronic
bronchitis, or emphysema.
Cough that is accompanied by excessive phlegm (mucus) or fever.Stop use and ask a doctor if Cough persists for more than 1 week, tends to recur, or is accompanied by a
fever, rash, or persistent headache. A persistent cough may be a sign of a serious condition.Stop use and ask a doctor if symptoms persist or worsen.
If pregnant or breastfeeding, ask a health professional before use.
Keep out of reach of children.In case of overdose, get medical help or contact a Poison Control Center right away.
- ASK DOCTOR
- STOP USE
- DO NOT USE
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- OVERDOSAGE
- INACTIVE INGREDIENT
- OTHER SAFETY INFORMATION
- DOSAGE & ADMINISTRATION
- INDICATIONS & USAGE
- ACTIVE INGREDIENT
- PURPOSE
- WARNINGS
- STOP USE
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- OVERDOSAGE
- INACTIVE INGREDIENT
- OTHER SAFETY INFORMATION
- DOSAGE & ADMINISTRATION
- INDICATIONS & USAGE
-
PRINCIPAL DISPLAY PANEL

UNDA
NDC: 62106-1169-8
numbered compounds
UNDA 312
Homeopathic PreparationFor the relief of symptoms associated with sore throat.
Contains 31% Alcohol
0.7 fl oz (20 mL) -
PRINCIPAL DISPLAY PANEL
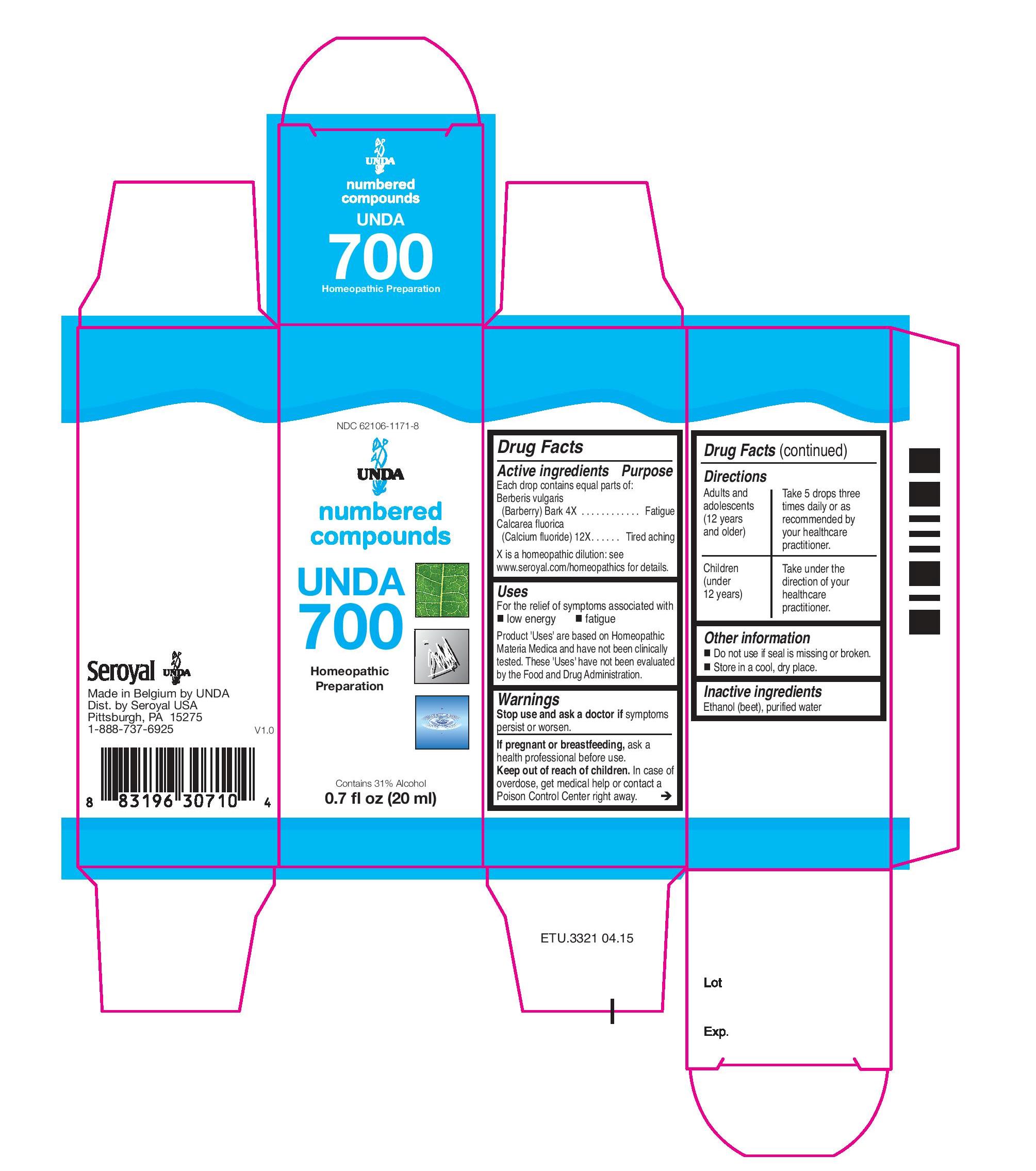
UNDA
NDC: 62106-1171-8
numbered compounds
UNDA 700
Homeopathic Preparation
Contains 31% Alcohol
0.7 fl oz (20 mL) -
INGREDIENTS AND APPEARANCE
UNDA 700
berberis vulgaris, calcarea fluorica liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 62106-1171 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BERBERIS VULGARIS ROOT BARK (UNII: 1TH8Q20J0U) (BERBERIS VULGARIS ROOT BARK - UNII:1TH8Q20J0U) BERBERIS VULGARIS ROOT BARK 4 [hp_X] in 20 mL CALCIUM FLUORIDE (UNII: O3B55K4YKI) (FLUORIDE ION - UNII:Q80VPU408O) CALCIUM FLUORIDE 12 [hp_X] in 20 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 62106-1171-8 1 in 1 CARTON 09/23/2015 1 20 mL in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 09/23/2015 UNDA 312
angelica archangelica, drosera, grindelia, lamium album, pinus sylvestris, rubus fruticosus, senega officinalis liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 62106-1169 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DROSERA ROTUNDIFOLIA FLOWERING TOP (UNII: 75O014T1HG) (DROSERA ROTUNDIFOLIA FLOWERING TOP - UNII:75O014T1HG) DROSERA ROTUNDIFOLIA FLOWERING TOP 6 [hp_X] in 20 mL GRINDELIA HIRSUTULA FLOWERING TOP (UNII: IDB0NAZ6AI) (GRINDELIA HIRSUTULA FLOWERING TOP - UNII:IDB0NAZ6AI) GRINDELIA HIRSUTULA FLOWERING TOP 6 [hp_X] in 20 mL PINUS SYLVESTRIS LEAFY TWIG (UNII: Q1RGP4UB73) (PINUS SYLVESTRIS LEAFY TWIG - UNII:Q1RGP4UB73) PINUS SYLVESTRIS LEAFY TWIG 6 [hp_X] in 20 mL ANGELICA ARCHANGELICA ROOT (UNII: DTN01M69SN) (ANGELICA ARCHANGELICA ROOT - UNII:DTN01M69SN) ANGELICA ARCHANGELICA ROOT 6 [hp_X] in 20 mL LAMIUM ALBUM (UNII: 046Y1357I6) (LAMIUM ALBUM - UNII:046Y1357I6) LAMIUM ALBUM 6 [hp_X] in 20 mL RUBUS FRUTICOSUS LEAF (UNII: YQ2S06L8S9) (RUBUS FRUTICOSUS LEAF - UNII:YQ2S06L8S9) RUBUS FRUTICOSUS LEAF 6 [hp_X] in 20 mL POLYGALA SENEGA ROOT (UNII: M7T6H7D4IF) (POLYGALA SENEGA ROOT - UNII:M7T6H7D4IF) POLYGALA SENEGA ROOT 6 [hp_X] in 20 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 62106-1169-8 1 in 1 CARTON 09/23/2015 1 20 mL in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 09/23/2015 Labeler - Seroyal USA (018361118) Establishment Name Address ID/FEI Business Operations SAN’UP 401010287 manufacture(62106-1171, 62106-1169)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.