REGONOL- pyridostigmine bromide injection, solution
Regonol by
Drug Labeling and Warnings
Regonol by is a Prescription medication manufactured, distributed, or labeled by Sandoz Inc, Delpharm Boucherville Canada Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
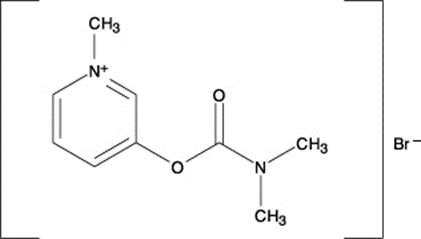
REGONOL® (pyridostigmine bromide injection, USP) is an active cholinesterase inhibitor chemically designated as 3-hydroxy-1-methylpyridinium bromide dimethyl-carbamate.
Its structural formula is:
REGONOL® is supplied as a sterile, isotonic, nonpyrogenic solution for injection. Each mL contains 5 mg of pyridostigmine bromide with 1% BENZYL ALCOHOL, WHICH IS NOT INTENDED FOR USE IN NEWBORNS, as the preservative. The pH is buffered with sodium citrate and citric acid and adjusted with sodium hydroxide if necessary.
-
CLINICAL PHARMACOLOGY
REGONOL® (pyridostigmine bromide injection, USP) an analogue of neostigmine, facilitates the transmission of impulses across the myoneural junction by inhibiting the destruction of acetylcholine by cholinesterase. Currently available data indicate that pyridostigmine may have a significantly lower degree and incidence of bradycardia, salivation and gastrointestinal stimulation than does neostigmine. Animal studies using the injectable form of pyridostigmine and human studies using the oral preparation have indicated that pyridostigmine has a longer duration of action than does neostigmine measured under similar circumstances.1,2 REGONOL® is effective in reversing the neuromuscular blocking effects of nondepolarizing muscle relaxants.
Anticholinesterase agents such as REGONOL® and neostigmine may produce depolarization block when administered at doses above their recommended therapeutic ranges. The therapeutic index of REGONOL® (ratio of reversal dose to blocking dose) is approximately 1:6 (see OVERDOSAGE).3
The antagonism of neuromuscular blockade by anticholinesterase agents may be influenced by the degree of spontaneous recovery achieved when the reversal agent is administered, by the particular relaxant administered, acid-base balance, body temperature, electrolyte imbalance, concomitant medications such as potent inhalational anesthetics, antibiotics or other drugs which enhance or antagonize the action of nondepolarizing muscle relaxants.4 The use of peripheral nerve stimulation to determine the degree of neuromuscular blockade is recommended in evaluating the effects of the reversal agents.
Failure of anticholinesterase agents to produce prompt (within 30 minutes) reversal of neuromuscular blockade may occur in the presence of extreme debilitation, carcinomatosis, and with concomitant use of certain broad-spectrum antibiotics, or anesthetic agents and other drugs which enhance neuromuscular blockade or cause respiratory depression through their own pharmacologic actions.
As with other anticholinesterase agents, the administration of REGONOL® may be associated with muscarinic and nicotinic side effects, notably bradycardia and excessive bronchial secretions; the use of glycopyrrolate or atropine sulfate simultaneously with or prior to administration of REGONOL® is recommended to counteract these side effects (see DOSAGE AND ADMINISTRATION).5
Pharmacokinetics
It has been postulated that the clearance of pyridostigmine is almost equally dependent on metabolism and on urinary elimination of the unchanged drug.6 Other studies in man indicated that approximately 75 percent of the plasma clearance of pyridostigmine is dependent on renal excretion and the remainder on nonrenal mechanisms.7
- INDICATIONS AND USAGE
- CONTRAINDICATIONS
-
WARNINGS
NOT FOR USE IN NEONATES
REGONOL® (pyridostigmine bromide injection, USP) should be used with particular caution in patients with bronchial asthma or cardiac dysrhythmias. Transient bradycardia may occur and be relieved by atropine sulfate. Atropine sulfate should also be used with caution in patients with cardiac dysrhythmias. When large doses of pyridostigmine bromide are administered, as during reversal of muscle relaxants, prior or simultaneous injection of atropine sulfate or an equipotent dose of glycopyrrolate is advisable. Because of the possibility of hypersensitivity in an occasional patient, atropine and antishock medication should always be readily available.
When used as an antagonist to nondepolarizing muscle relaxants, adequate recovery of voluntary respiration and neuromuscular transmission must be obtained prior to discontinuation of respiratory assistance, and there should be continuous patient observation. Satisfactory recovery may be judged by adequacy of skeletal muscle tone, respiratory measurements, and by observation of the response to peripheral nerve stimulation. A patent airway should be maintained and manual or mechanical ventilation should be continued until complete recovery of normal respiration is assured.
Exposure to excessive amounts of benzyl alcohol has been associated with toxicity (hypotension, metabolic acidosis), particularly in neonates, and an increased incidence of kernicterus, particularly in small preterm infants. There have been rare reports of deaths, primarily in preterm infants, associated with exposure to excessive amounts of benzyl alcohol. The amount of benzyl alcohol from medications is usually considered negligible compared to that received in flush solutions containing benzyl alcohol. Administration of high dosages of medications containing this preservative must take into account the total amount of benzyl alcohol administered. The amount of benzyl alcohol at which toxicity may occur is not known. If the patient requires more than the recommended dosages or other medications containing this preservative, the practitioner must consider the daily metabolic load of benzyl alcohol from these combined sources (see PRECAUTIONS: Pediatric Use).
-
PRECAUTIONS
THE USE OF A PERIPHERAL NERVE STIMULATOR TO MONITOR RECOVERY OF NEUROMUSCULAR FUNCTION WILL MINIMIZE THE POSSIBILITY OF EXCESS DOSING OR INADEQUATE REVERSAL.
Inadequate reversal of the neuromuscular blockade induced by nondepolarizing (curariform) muscle relaxants is possible. This can be managed by manual or mechanical ventilation until recovery is judged adequate. The administration of additional doses of anticholinesterase reversal agents is not recommended since excessive dosages of such drugs may produce depolarizing block through their own pharmacologic actions.
Pyridostigmine is mainly excreted unchanged by the kidney.2,7,8 Therefore, lower doses may be required in patients with renal disease, and treatment should be based on titration of drug dosage to effect.2,7
Drug Interactions
Concomitant administration of REGONOL® (pyridostigmine bromide injection, USP) and 4-aminopyridine has been reported to delay the onset of action of REGONOL®.9
Antibiotics
Parenteral administration of high doses of certain antibiotics may intensify or produce neuromuscular block through their own pharmacologic actions. The following antibiotics have been associated with various degrees of paralysis: aminoglycosides (such as neomycin, streptomycin, kanamycin, gentamicin, and dihydrostreptomycin); tetracyclines; bacitracin; polymyxin B; colistin; and sodium colistimethate. If these or other newly introduced antibiotics are used in conjunction with nondepolarizing neuromuscular blocking drugs during surgery, unexpected prolongation of neuromuscular block or resistance to its reversal should be considered a possibility.
Other
Experience concerning injection of quinidine during recovery from use of nondepolarizing muscle relaxants suggest that recurrent paralysis may occur. This possibility must be considered when administering anticholinesterase agents to antagonize neuromuscular blockade induced by nondepolarizing muscle relaxants.
Electrolyte imbalance and diseases which lead to electrolyte imbalance, such as adrenal cortical insufficiency, have been shown to alter neuromuscular blockade. Depending on the nature of the imbalance, either enhancement or inhibition may be expected. Magnesium salts, administered for the management of toxemia of pregnancy, may enhance the neuromuscular blockade. The possibility that such circumstances may interfere with the restoration of neuromuscular function should be considered when administering REGONOL®.
Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term studies in animals have not been performed to evaluate carcinogenic or mutagenic potential or impairment of fertility.
Pregnancy
Pregnancy Category C
It is not known whether REGONOL® (pyridostigmine bromide injection, USP) can cause fetal harm when administered to a pregnant woman or can affect reproductive capacity. REGONOL® should be given to a pregnant woman only if the administering clinician decides that the benefits outweigh the risks.
Pediatric Use
Safety and efficacy in pediatric patients have not been established.
Benzyl alcohol, a component of this drug product, has been associated with serious adverse events and death, particularly in pediatric patients. The “gasping syndrome”, (characterized by central nervous system depression, metabolic acidosis, gasping respirations, and high levels of benzyl alcohol and its metabolites found in the blood and urine) has been associated with benzyl alcohol dosages >99 mg/kg/day in neonates and low-birth-weight neonates. Additional symptoms may include gradual neurological deterioration, seizures, intracranial hemorrhage, hematologic abnormalities, skin breakdown, hepatic and renal failure, hypotension, bradycardia, and cardiovascular collapse. Although normal therapeutic doses of this product deliver amounts of benzyl alcohol that are substantially lower than those reported in association with the “gasping syndrome”, the minimum amount of benzyl alcohol at which toxicity may occur is not known. Premature and low-birth-weight infants, as well as patients receiving high dosages, may be more likely to develop toxicity. Practitioners administering this and other medications containing benzyl alcohol should consider the combined daily metabolic load of benzyl alcohol from all sources.
-
ADVERSE REACTIONS
The side effects of pyridostigmine bromide are most commonly related to overdosage and generally are of two varieties, muscarinic and nicotinic. Among those in the former group are nausea, vomiting, diarrhea, abdominal cramps, increased peristalsis, increased salivation, increased bronchial secretions, miosis, and diaphoresis. Nicotinic side effects are comprised chiefly of muscle cramps, fasciculation, and weakness. Muscarinic side effects can usually be counteracted by atropine. As with any compound containing the bromide radical, a skin rash may be seen in an occasional patient. Such reactions usually subside promptly upon discontinuance of the medication. Thrombophlebitis has been reported subsequent to intravenous administration.
-
OVERDOSAGE
THE POSSIBILITY OF IATROGENIC OVERDOSAGE CAN BE MINIMIZED BY CAREFULLY MONITORING THE MUSCLE TWITCH RESPONSE TO PERIPHERAL NERVE STIMULATION. Should overdosage occur, ventilation should be supported by artificial means until the adequacy of spontaneous respiration is assured, and cardiac function should be monitored.
Respiratory depression following administration of nondepolarizing neuromuscular blocking agents may be due either wholly or in part to other drugs used during the conduct of general anesthesia, such as narcotics, thiobarbiturates and other central nervous system depressants. A peripheral nerve stimulator may be used to assess the degree of residual neuromuscular blockade and to help differentiate residual neuromuscular blockade from other causes of decreased respiratory reserve.
-
DOSAGE AND ADMINISTRATION
REGONOL® (pyridostigmine bromide injection, USP) is for intravenous use only. This drug should be administered by or under the supervision of experienced clinicians familiar with the use of agents which reverse or antagonize the effects of neuromuscular blocking agents. Dosage must be individualized in each case. The dosage information which follows is derived from studies based upon units of drug per unit of body weight and is intended to serve as a guide only. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
NOTE: CONTAINS BENZYL ALCOHOL (see PRECAUTIONS)
Reversal doses of REGONOL® range from 0.1 to 0.25 mg/kg.5,10,11,12,13 The onset time to peak effect is dose-dependent; return of twitch height to 90% of control occurs within approximately 6 minutes following administration of a 0.25 mg/kg dose of REGONOL®.5,12 At lower doses, full recovery usually occurs within 15 minutes in most patients, although others may require a half-hour or more.
When REGONOL® is given intravenously to reverse the action of muscle relaxant drugs, it is recommended that atropine sulfate (0.6 to 1.2 mg) or an equipotent dose of glycopyrrolate be given immediately prior to or simultaneously with the administration of REGONOL®. Side effects, notably excessive secretions and bradycardia are thereby minimized. Please refer to the appropriate prescribing information prior to the use of glycopyrrolate or atropine sulfate.
To obtain maximum clinical benefits of REGONOL® and to minimize the possibility of overdosage, the monitoring of muscle twitch response to peripheral nerve stimulation is advised. REGONOL® should be administered after spontaneous recovery of neuromuscular function has begun.
Satisfactory reversal can be evident by adequate voluntary respiration, respiratory measurements and use of a peripheral nerve stimulator device. It is recommended that the patient be well-ventilated and a patent airway maintained until complete recovery of normal respiration is assured. Once satisfactory reversal has been attained following administration of REGONOL®, recurrence of paralysis is unlikely to occur.
Inadequate reversal of neuromuscular blockade by anticholinesterase drugs is possible with all curariform drugs, and is managed by manual or mechanical ventilation until recovery is judged adequate. The administration of additional doses of anticholinesterase reversal agents is not recommended since excessive dosages of such drugs may produce depolarizing block through their own pharmacological actions.
Use in Pediatrics
The safety and efficacy of REGONOL® (pyridostigmine bromide injection, USP) in pediatric patients have not been established, therefore no dosing recommendations can be made (see PRECAUTIONS).
-
HOW SUPPLIED
REGONOL® (pyridostigmine bromide injection USP) for injection is available as:
REGONOL® 2 mL ampules containing 10 mg pyridostigmine bromide injection (5 mg/mL) and supplied as:
NDC: 0781-3040-95 boxes of 10
CONTAINS BENZYL ALCOHOL.
Store at 25°C (77°F); excursions permitted to 15°-30°C (59°-86°F) (see USP Controlled Room Temperature). Protect from light.
-
REFERENCES
- 1. Baker PR, Calvey TN, Chan K, Macnee CM, Taylor K. Plasma clearance of neostigmine and pyridostigmine in the dog. Br J Pharmacol 1978;63:509-512.
- 2. Cronnelly R, Stanski DR, Miller RD, Sheiner LB. Pyridostigmine kinetics with and without renal function. Clin Pharmacol & Ther 1980;28:78-81.
- 3. Katz RL. Pyridostigmine (Mestinon) as an antagonist of d-tubocurarine. Anesthesiology 1967;28:528-534.
- 4. Miller RD. Antagonism of neuromuscular blockade. Anesthesiology 1967; 44: 318-329.
- 5. Gyermek L. Clinical studies on the reversal of the neuromuscular blockade produced by pancuronium bromide. 1. The effects of glycopyrrolate and pyridostigmine. Curr Ther Res 1975; 18:20-23.
- 6. Williams ME, Calvey TN, Chan K. Plasma concentration of pyridostigmine during the antagonism of neuromuscular block. Br J Anesth 1983; 55:27-30.
- 7. Miller RD. Pharmacodynamics and pharmacokinetics of anticholinesterase. In: Ruegheimer E, Zindler M, eds., Anaesthesiology, Amsterdam, Netherlands: Excerpta Medica 1981: 222-223.
- 8. Breyer-Pfaff U, Maier U, Brinkmann AM, Schumm F. Pyridostigmine kinetics in healthy subjects and patients with myasthenia gravis. Clin Pharmacol Ther 1985;5:494-501.
- 9. Miller RD, Booij LH, Agoston S, Crul JF. 4-aminopyridine potentiates neostigmine and pyridostigmine in man. Anesthesiology 1979;50:416-420.
- 10. Gyermek L. The Glycopyrrolate-Pyridostigmine Combination. Anesthesiology Review 1978;5:19-22.
- 11. Zsigmond EK. New Safe and Effective Antagonist of Pancuronium Bromide: Pyridostigmine Bromide. A Scientific Exhibit Presented at the American Medical Association Annual Convention in New York City, NY; June 23-27, 1973.
- 12. Rusin WD. Comparison of Neostigmine and Pyridostigmine as Antagonists of Pancuronium Neuromuscular Blockade. ASA Clinical Papers, 299-300, 1976.
- 13. Katz R. Pyridostigmine as an Antagonist of d-Tubocurarine. Anesthesiology 1967;3:528-534.
- 14. Miller RD, Van Nyhuis LS, Eger EI, Vitez TS, Way WL. Comparative Times to Peak Effect and Durations of Action of Neostigmine and Pyridostigmine. Anesthesiology 1974;41:27-33.
- 15. Fogdall RP, Miller RD. Antagonism of d-Tubocurarine and Pancuronium Induced Neuromuscular Blockades by Pyridostigmine in Man. Anesthesiology 1973;39:504-509.
Rev. January 2019
46238566
Distributed by
Sandoz Inc., Princeton, NJ 08540
-
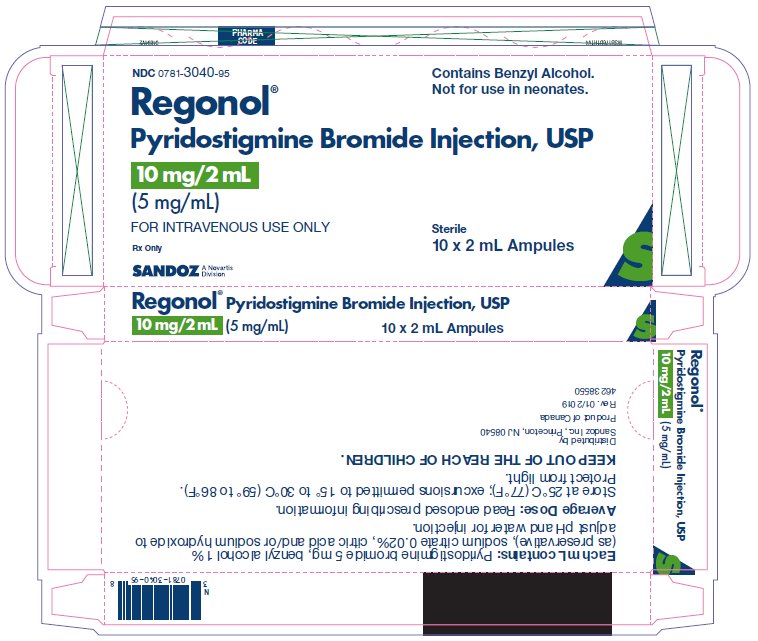
10 mg 2 mL Carton
NDC: 0781-3040-95
Regonol® Contains Benzyl Alcohol. Not for use in neonates.
Pyridostigmine Bromide Injection, USP
10 mg/2 mL Sterile
(5 mg/mL)
FOR INTRAVENOUS USE ONLY
Rx only
Sterile
10 x 2 mL Ampules
SANDOZ – A Novartis Division
-
INGREDIENTS AND APPEARANCE
REGONOL
pyridostigmine bromide injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 0781-3040 Route of Administration INTRAVENOUS, PARENTERAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PYRIDOSTIGMINE BROMIDE (UNII: KVI301NA53) (PYRIDOSTIGMINE - UNII:19QM69HH21) PYRIDOSTIGMINE BROMIDE 5 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZYL ALCOHOL (UNII: LKG8494WBH) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0781-3040-95 10 in 1 CARTON 05/10/2005 1 NDC: 0781-3040-72 2 mL in 1 AMPULE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA017398 05/10/2005 Labeler - Sandoz Inc (005387188)
Trademark Results [Regonol]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 REGONOL 78912946 3357938 Dead/Cancelled |
Novartis AG 2006-06-21 |
 REGONOL 73003241 0991089 Dead/Expired |
ORGANON INC. 1973-10-10 |
 REGONOL 71648125 0591580 Dead/Expired |
ORGANON INC. 1953-06-02 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.