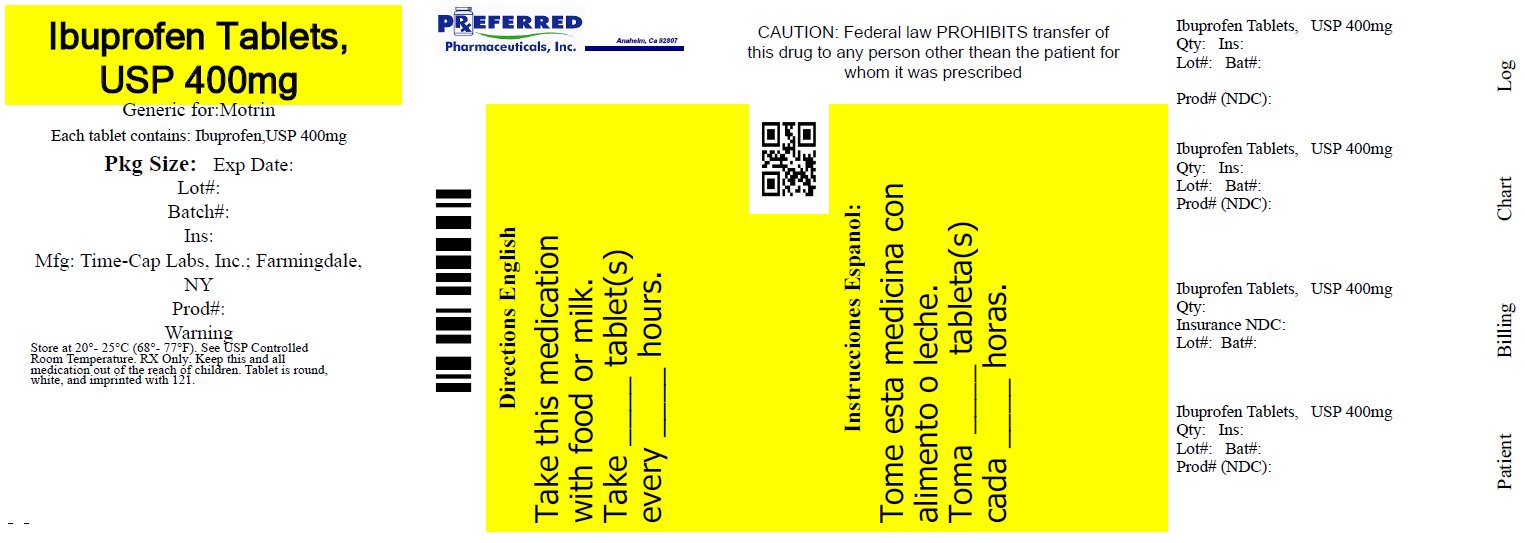
IBUPROFEN 400 MG - 600 MG AND 800 MG TABLETS
IBUPROFEN by
Drug Labeling and Warnings
IBUPROFEN by is a Prescription medication manufactured, distributed, or labeled by Preferred Pharmaceuticals, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
IBUPROFEN- ibuprofen tablet, film coated
Preferred Pharmaceuticals, Inc.
----------
IBUPROFEN 400 MG - 600 MG AND 800 MG TABLETS
HOW SUPPLIED
400mg (white to of white, round, biconvex, film coated tablets debossed with '121' on one side and plain on the other side)
-
Bottles of 20 NDC: 68788-7745-02
Bottles of 28 NDC: 68788-7745-08
Bottles of 30 NDC: 68788-7745-03
Bottles of 50 NDC: 68788-7745-05
Bottles of 60 NDC: 68788-7745-06
Bottles of 90 NDC: 68788-7745-09
Bottles of 100 NDC: 68788-7745-01
Repackaged by Preferred Pharmaceuticals, Inc.
| IBUPROFEN
ibuprofen tablet, film coated |
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||
| Labeler - Preferred Pharmaceuticals, Inc. (791119022) |
| Registrant - Preferred Pharmaceuticals, Inc. (791119022) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Preferred Pharmaceuticals, Inc. | 791119022 | REPACK(68788-7745) | |
Revised: 7/2025
Document Id: 2032b7fb-2c25-42e1-b85d-94babfd7273e
Set id: 20fda75a-c560-428b-b9fd-c12e97df4a74
Version: 6
Effective Time: 20250702