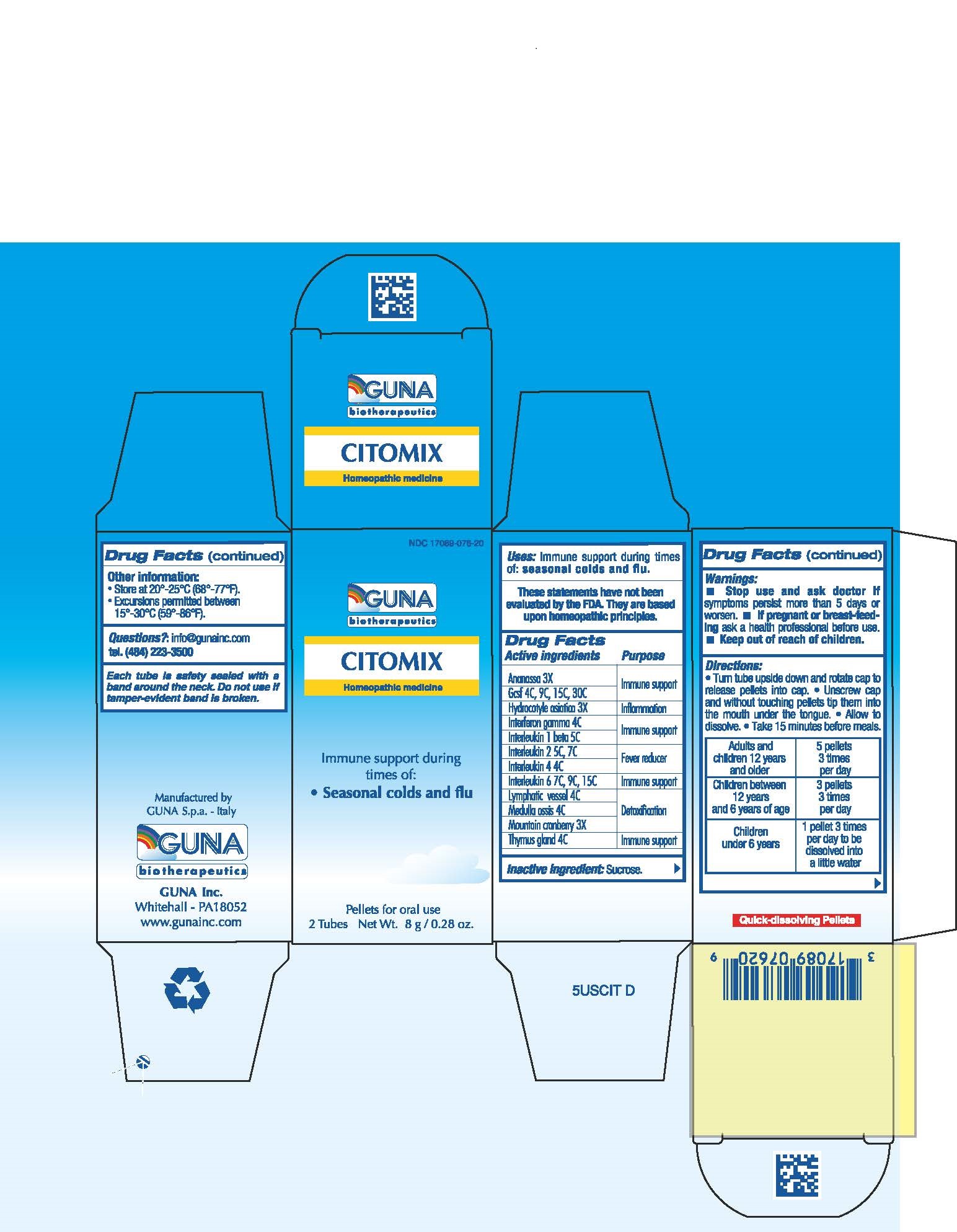
CITOMIX (aldesleukin - binetrakin - canakinumab - centella asiatica - cranberry - human interleukin-6- nonglycosylated - interferon gamma-1b - lenograstim - pineapple - sus scrofa bone marrow - sus scrofa small intestine mucosa lymph follicle - sus scrofa thymus - pellet
CITOMIX by
Drug Labeling and Warnings
CITOMIX by is a Homeopathic medication manufactured, distributed, or labeled by Guna spa. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
ACTIVE INGREDIENTS/PURPOSE
ANANASSA 3X IMMUNE SUPPORT
GCSF 4C, 9C, 15C, 30C IMMUNE SUPPORT
HYDROCOTYLE ASIATICA 3X INFLAMMATION
INTERFERON GAMMA 4C IMMUNE SUPPORT
INTERLEUKIN 1 BETA 5C IMMUNE SUPPORT
INTERLEUKIN 2 5C, 7C FEVER REDUCER
INTERLEUKIN 4 4C FEVER REDUCER
INTERLEUKIN 6 7C, 9C, 15C IMMUNE SUPPORT
LYMPHATIC VESSEL 4C DETOXIFICATION
MEDULLA OSSIS 4C DETOXIFICATION
MOUNTAIN CRANBERRY 3X DETOXIFICATION
THYMUS GLAND 4C IMMUNE SUPPORT - USES
- WARNINGS
- PREGNANCY
- WARNINGS
- DIRECTIONS
- QUESTIONS
- DOSAGE & ADMINISTRATION
- WARNINGS
- INACTIVE INGREDIENT
- INDICATIONS & USAGE
- KEEP OUT OF REACH OF CHILDREN
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CITOMIX
aldesleukin - binetrakin - canakinumab - centella asiatica - cranberry - human interleukin-6 (nonglycosylated) - interferon gamma-1b - lenograstim - pineapple - sus scrofa bone marrow - sus scrofa small intestine mucosa lymph follicle - sus scrofa thymus - pelletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 17089-076 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PINEAPPLE (UNII: 2A88ZO081O) (PINEAPPLE - UNII:2A88ZO081O) PINEAPPLE 3 [hp_X] in 4 g LENOGRASTIM (UNII: 6WS4C399GB) (LENOGRASTIM - UNII:6WS4C399GB) LENOGRASTIM 4 [hp_C] in 4 g CENTELLA ASIATICA (UNII: 7M867G6T1U) (CENTELLA ASIATICA - UNII:7M867G6T1U) CENTELLA ASIATICA 3 [hp_X] in 4 g INTERFERON GAMMA-1B (UNII: 21K6M2I7AG) (INTERFERON GAMMA-1B - UNII:21K6M2I7AG) INTERFERON GAMMA-1B 4 [hp_C] in 4 g CANAKINUMAB (UNII: 37CQ2C7X93) (CANAKINUMAB - UNII:37CQ2C7X93) CANAKINUMAB 5 [hp_C] in 4 g ALDESLEUKIN (UNII: M89N0Q7EQR) (ALDESLEUKIN - UNII:M89N0Q7EQR) ALDESLEUKIN 5 [hp_C] in 4 g BINETRAKIN (UNII: 751635Z921) (BINETRAKIN - UNII:751635Z921) BINETRAKIN 4 [hp_C] in 4 g HUMAN INTERLEUKIN-6 (NONGLYCOSYLATED) (UNII: 92QVL9080Y) (HUMAN INTERLEUKIN-6 (NONGLYCOSYLATED) - UNII:92QVL9080Y) HUMAN INTERLEUKIN-6 (NONGLYCOSYLATED) 7 [hp_C] in 4 g SUS SCROFA SMALL INTESTINE MUCOSA LYMPH FOLLICLE (UNII: 308LM01C72) (SUS SCROFA SMALL INTESTINE MUCOSA LYMPH FOLLICLE - UNII:308LM01C72) SUS SCROFA SMALL INTESTINE MUCOSA LYMPH FOLLICLE 4 [hp_C] in 4 g SUS SCROFA BONE MARROW (UNII: VP2CN2G7Y8) (SUS SCROFA BONE MARROW - UNII:VP2CN2G7Y8) SUS SCROFA BONE MARROW 4 [hp_C] in 4 g CRANBERRY (UNII: 0MVO31Q3QS) (CRANBERRY - UNII:0MVO31Q3QS) CRANBERRY 3 [hp_X] in 4 g SUS SCROFA THYMUS (UNII: 7B69B0BD62) (SUS SCROFA THYMUS - UNII:7B69B0BD62) SUS SCROFA THYMUS 4 [hp_C] in 4 g Inactive Ingredients Ingredient Name Strength SUCROSE (UNII: C151H8M554) 3.5 g in 4 g Product Characteristics Color white (white) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 17089-076-20 2 in 1 BOX 05/23/2006 1 4 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 05/23/2006 Labeler - Guna spa (430538264) Establishment Name Address ID/FEI Business Operations Guna spa 338587646 manufacture(17089-076)
Trademark Results [CITOMIX]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 CITOMIX 79267669 not registered Live/Pending |
GUNA S.p.a. 2019-07-31 |
 CITOMIX 78825262 3882592 Live/Registered |
GUNA, INC. 2006-02-28 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.