MEROPENEM injection, powder, for solution
Meropenem by
Drug Labeling and Warnings
Meropenem by is a Prescription medication manufactured, distributed, or labeled by WG Critical Care, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use MEROPENEM FOR INJECTION safely and effectively. See full prescribing information for MEROPENEM FOR INJECTION.
MEROPENEM for injection, for intravenous use
Initial U.S. Approval: 1996RECENT MAJOR CHANGES
Warnings and Precautions, Rhabdomyolysis (5.3) 12/2024
INDICATIONS AND USAGE
Meropenem for Injection is a penem antibacterial indicated for the treatment of bacterial meningitis in pediatric patients 3 months of age and older only. (1.1)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Meropenem for Injection and other antibacterial drugs, Meropenem for Injection should only be used to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. (1.2)
DOSAGE AND ADMINISTRATION
- Pediatric patients 3 months of age and older with bacterial meningitis (2.1)
Recommended Meropenem for Injection Dosage Schedule for Pediatric Patients 3 Months of Age and Older with Bacterial Meningitis and Normal Renal Function (2.1)
Type of Infection
Dose (mg/kg)
Up to a Maximum Dose
Dosing Interval
Bacterial Meningitis
40
2 grams
Every 8 hours
- Intravenous infusion is to be given over approximately 15 minutes to 30 minutes.
- There is no experience with the use of Meropenem for Injection in pediatric patients with renal impairment.
DOSAGE FORMS AND STRENGTHS
Meropenem for Injection: 2 grams of meropenem as a powder in a single-dose vial for reconstitution (3)
CONTRAINDICATIONS
- Known hypersensitivity to product components or other drugs in the same class or in patients who have demonstrated anaphylactic reactions to beta (β)-lactams. (4)
WARNINGS AND PRECAUTIONS
- Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients receiving β-lactams. (5.1)
- Severe cutaneous adverse reactions have been reported in patients receiving meropenem for injection. (5.2)
- Rhabdomyolysis: If signs or symptoms of rhabdomyolysis are observed, discontinue meropenem for injection and initiate appropriate therapy. (5.3)
- Seizures and other adverse CNS experiences have been reported during treatment. (5.4)
- Co-administration of meropenem for injection with valproic acid or divalproex sodium reduces the serum concentration of valproic acid potentially increasing the risk of breakthrough seizures. (5.5, 7.2)
- Clostridioides difficile-associated diarrhea (ranging from mild diarrhea to fatal colitis) has been reported. Evaluate if diarrhea occurs. (5.6)
- In patients with renal dysfunction, thrombocytopenia has been observed (5.9)
ADVERSE REACTIONS
Most common adverse reactions (incidence ≥ 1%) are: diarrhea, rash (mostly diaper area moniliasis), oral moniliasis, and glossitis (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact WG Critical Care, LLC at 1-866-562-4708 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
- Co-administration of meropenem for injection with probenecid inhibits renal excretion of meropenem and therefore Meropenem for Injection is not recommended (7.1)
- The concomitant use of Meropenem for Injection and valproic acid or divalproex sodium is generally not recommended. Antibacterial drugs other than carbapenems should be considered to treat infections in patients whose seizures are well controlled on valproic acid or divalproex sodium. (5.4, 7.2)
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 7/2023
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Bacterial Meningitis (Pediatric Patients 3 Months of Age and Older Only)
1.2 Usage
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage in Pediatric Patients 3 Months of Age and Older with Bacterial Meningitis
2.2 Preparation and Administration of Meropenem for Injection
2.3 Compatibility
2.4 Stability and Storage
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions
5.2 Severe Cutaneous Adverse Reactions
5.3 Rhabdomyolysis
5.4 Seizure Potential
5.5 Risk of Breakthrough Seizures Due to Drug Interaction with Valproic Acid
5.6 Clostridioides difficile–associated Diarrhea
5.7 Development of Drug-Resistant Bacteria
5.8 Overgrowth of Nonsusceptible Organisms
5.9 Thrombocytopenia
5.10 Potential for Neuromotor Impairment
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Post-marketing Experience
7 DRUG INTERACTIONS
7.1 Probenecid
7.2 Valproic Acid
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
12.4 Microbiology
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Bacterial Meningitis
15 REFERENCES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
1.1 Bacterial Meningitis (Pediatric Patients 3 Months of Age and Older Only)
Meropenem for Injection is indicated for the treatment of bacterial meningitis caused by Haemophilus influenzae, Neisseria meningitidis andpenicillin-susceptible isolates of Streptococcus pneumoniae, in pediatric patients 3 months of age and older. Meropenem for injection has been found to be effective in eliminating concurrent bacteremia in association with bacterial meningitis.
1.2 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Meropenem for Injection and other antibacterial drugs, Meropenem for Injection should only be used to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage in Pediatric Patients 3 Months of Age and Older with Bacterial Meningitis
- For pediatric patients 3 months of age and older with bacterial meningitis, the Meropenem for Injection recommended dosage is 40 mg/kg every 8 hours (maximum dosage is 2 grams every 8 hours).
- For pediatric patients weighing over 50 kg administer Meropenem for Injection at a dosage of 2 grams every 8 hours for bacterial meningitis.
- Administer diluted Meropenem for Injection as an intravenous infusion over approximately 15 minutes to 30 minutes [see Dosage and Administration (2.2)].
- There is limited safety data available to support the administration of a 40 mg/kg (up to a maximum of 2 grams) intravenous bolus injection. There is no experience with the use of Meropenem for Injection in pediatric patients with renal impairment [see Clinical Pharmacology (12.3)].
2.2 Preparation and Administration of Meropenem for Injection
Preparation instructions for Intravenous Infusion (Reconstitution and Dilution)
- Reconstitute Meropenem for Injection vial (2 grams) with Sterile Water for Injection according to the instructions in Table 1 below.
- Shake well to dissolve and let stand until clear.
- Table 1: Volume of Sterile Water for Injection for Reconstitution of Injection Vials
Vial Size
Amount of Diluent Added (mL)
Approximate Withdrawable Volume (mL)
Approximate Average Concentration (mg/mL)
2 grams
40 mL
40 mL
50 mg/mL
- Add the resulting reconstituted solution to an intravenous container and further dilute with an appropriate infusion fluid [see Dosage and Administration(2.3)and (2.4)].
- Discard unused portion.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. When reconstituted, the solution varies from colorless to yellow depending on the concentration.
- Do not use flexible container in series connections.
Administration Instructions for Meropenem Intravenous Infusion Solution
After reconstitution and dilution of Meropenem for Injection, administer the appropriate dose of diluted Meropenem solution as an intravenous infusion over approximately 15 minutes to 30 minutes [see Dosage and Administration (2.1)].
2.3 Compatibility
Compatibility of Meropenem for Injection with other drugs has not been established. Meropenem for Injection should not be mixed with or physically added to solutions containing other drugs.
2.4 Stability and Storage
Reconstituted Solution with Sterile Water for Injection
Use the reconstituted solution with Sterile Water for Injection immediately. However, re-constituted solutions of Meropenem for Injection maintain satisfactory potency under the conditions described below. Do not freeze reconstituted solutions of Meropenem for Injection.
Reconstituted Solution Diluted with Sodium Chloride Injection, 0.9 %
Solutions prepared for infusion (meropenem concentrations ranging from 1 mg/mL to 20 mg/mL) reconstituted with Water for Injection and further diluted with Sodium Chloride Injection 0.9% may be stored for 1 hour at up to 25°C (77°F) or 2 hours at up to 5°C (41°F).
Reconstituted Solution Diluted with Dextrose Injection, 5 %
Immediately use solutions (meropenem concentrations ranging from 1 mg/mL to 20 mg/mL) reconstituted with Water for Injection and subsequently diluted with Dextrose Injection 5%.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Hypersensitivity Reactions
Serious and occasionally fatal hypersensitivity (anaphylactic) reactions have been reported in patients receiving therapy with β-lactams. These reactions are more likely to occur in individuals with a history of sensitivity to multiple allergens.
There have been reports of individuals with a history of penicillin hypersensitivity who have experienced severe hypersensitivity reactions when treated with another β-lactam. Before initiating therapy with meropenem for injection, it is important to inquire about previous hypersensitivity reactions to penicillins, cephalosporins, other β-lactams, and other allergens. If an allergic reaction to meropenem for injection occurs, discontinue the drug immediately.
5.2 Severe Cutaneous Adverse Reactions
Severe cutaneous adverse reactions (SCAR) such as Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), erythema multiforme (EM) and acute generalized exanthematous pustulosis (AGEP) have been reported in patients receiving meropenem for injection [see Adverse Reactions (6.2)]. If signs and symptoms suggestive of these reactions appear, meropenem should be withdrawn immediately and an alternative treatment should be considered.
5.3 Rhabdomyolysis
Rhabdomyolysis has been reported with the use of meropenem [see Adverse Reactions (6.2)]. If signs or symptoms of rhabdomyolysis such as muscle pain, tenderness or weakness, dark urine, or elevated creatine phosphokinase are observed, discontinue meropenem for injection and initiate appropriate therapy.
5.4 Seizure Potential
Seizures and other adverse CNS experiences have been reported during treatment with meropenem for injection. These experiences have occurred most commonly in patients with CNS disorders (e.g., brain lesions or history of seizures) or with bacterial meningitis and/or compromised renal function [see Adverse Reactions(6.1) and Drug Interactions(7.2)].
During clinical investigations, 2904 immunocompetent adult patients were treated for non-CNS infections with the overall seizure rate being 0.7% (based on 20 patients with this adverse event). All meropenem-treated patients with seizures had pre-existing contributing factors. Among these included a prior history of seizures or CNS abnormality and concomitant medications with seizure potential.
Close adherence to the recommended dosage regimens is urged, especially in patients with known factors that predispose to convulsive activity. Continue anti-convulsant therapy in patients with known seizure disorders. If focal tremors, myoclonus, or seizures occur, evaluate neurologically, placed on anti-convulsant therapy if not already instituted, and re-examine the dosage of meropenem for injection to determine whether it should be decreased or discontinued.
5.5 Risk of Breakthrough Seizures Due to Drug Interaction with Valproic Acid
The concomitant use of meropenem and valproic acid or divalproex sodium is generally not recommended. Case reports in the literature have shown that co-administration of carbapenems, including meropenem, to patients receiving valproic acid or divalproex sodium results in a reduction in valproic acid concentrations. The valproic acid concentrations may drop below the therapeutic range as a result of this interaction, therefore increasing the risk of breakthrough seizures. Increasing the dose of valproic acid or divalproex sodium may not be sufficient to overcome this interaction. Consider administration of antibacterial drugs other than carbapenems to treat infections in patients whose seizures are well controlled on valproic acid or divalproex sodium. If administration of meropenem for injection is necessary, consider supplemental anti-convulsant therapy [see Drug Interactions (7.2)].
5.6 Clostridioides difficile–associated Diarrhea
Clostridioides difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including meropenem for injection, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing isolates of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial drug use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial drug use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial drug treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
5.7 Development of Drug-Resistant Bacteria
Prescribing meropenem for injection in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
5.8 Overgrowth of Nonsusceptible Organisms
As with other broad-spectrum antibacterial drugs, prolonged use of meropenem may result in overgrowth of nonsusceptible organisms. Repeated evaluation of the patient is essential. If superinfection does occur during therapy, appropriate measures should be taken.
5.9 Thrombocytopenia
In patients with renal impairment, thrombocytopenia has been observed but no clinical bleeding reported [see Dosage and Administration (2.2), Adverse Reactions (6.1), and Clinical Pharmacology (12.3)].
5.10 Potential for Neuromotor Impairment
Alert patients receiving meropenem for injection on an outpatient basis regarding adverse events such as seizures, delirium, headaches and/or paresthesias that could interfere with mental alertness and/or cause motor impairment. Until it is reasonably well established that meropenem for injection is well tolerated, advise patients not to operate machinery or motorized vehicles [see Adverse Reactions (6.1)].
-
6 ADVERSE REACTIONS
The following are discussed in greater detail in other sections of labeling:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
- Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.2)]
- Rhabdomyolysis [see Warnings and Precautions (5.3)]
- Seizure Potential [see Warnings and Precautions (5.4)]
- Risk of Breakthrough Seizures Due to Drug Interaction with Valproic Acid [see Warnings and Precautions (5.5)]
- Clostridioides difficile – Associated Diarrhea [see Warnings and Precautions (5.6)]
- Development of Drug-Resistant Bacteria [see Warnings and Precautions (5.7)]
- Overgrowth of Nonsusceptible Organisms [see Warnings and Precautions (5.8)]
- Thrombocytopenia [see Warnings and Precautions (5.9)]
- Potential for Neuromotor Impairment [see Warnings and Precautions (5.10)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reactions rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Adverse Reactions in Pediatric Patients with Bacterial Meningitis
Meropenem for injection was studied in 321 pediatric patients (3 months to less than 17 years of age) with bacterial meningitis at a dosage of 40 mg/kg every 8 hours. The most common adverse reactions and their rates of occurrence were as follows:
- Diarrhea 4.7%
- Rash (mostly diaper area moniliasis) 3.1%
- Oral Moniliasis 1.9%
- Glossitis 1%
In these studies, the rates of seizure activity during therapy were comparable between patients with no CNS abnormalities who received meropenem for injection and those who received comparator agents (either cefotaxime or ceftriaxone). In the meropenem for injection-treated group, 12/15 patients with seizures had late onset seizures (seizures that occurred on day 3 or later) versus 7/20 patients in the comparator arm.
The meropenem for injection group had a statistically higher number of patients than the comparator with transient elevation of liver enzymes.
Adverse Reactions from Studies of Meropenem for Injectionin Other Serious Bacterial Infections (not bacterial meningitis)
The following adverse reactions occurred in studies of 2,904 immunocompetent adult patients who received meropenem for injection (500 mg or 1 gram every 8 hours) for the treatment of other serious bacterial infections (not bacterial meningitis). This meropenem for injection product is indicated only for the treatment of bacterial meningitis caused by certain organisms in pediatric patients 3 months of age and older [see Indications and Usage (1.1)]. Many patients in these studies were severely ill and had multiple background diseases, physiological impairments and were receiving multiple other drug therapies. In the seriously ill patient population, it was not possible to determine the relationship between observed adverse events and therapy with meropenem for injection.
Deaths in 5 patients were assessed as possibly related to meropenem for injection; 36 (1.2%) patients had meropenem for injection discontinued because of adverse events.
Local Adverse Reactions
Local adverse reactions that were reported with meropenem for injection were as follows: Inflammation at the injection site (2.4%), injection site reaction (0.9%), phlebitis/thrombophlebitis (0.8%), pain at the injection site (0.4%), and edema at the injection site (0.2%)
Systemic Adverse Reactions
Systemic adverse reactions that occurred in greater than 1% of the meropenem for injection-treated patients were diarrhea (4.8%), nausea/vomiting (3.6%), headache (2.3%), rash (1.9%), sepsis (1.6%), constipation (1.4%), apnea (1.3%), shock (1.2%), and pruritus (1.2%). Additional systemic adverse reactions that occurred in less than or equal to 1% but greater than 0.1% of the meropenem for injection-treated patients are listed below within each body system in order of decreasing frequency:
Bleeding Events: gastrointestinal hemorrhage (0.5%), melena (0.3%), epistaxis (0.2%), hemoperitoneum (0.2%).
Body as a Whole: pain, abdominal pain, chest pain, fever, back pain, abdominal enlargement, chills, pelvic pain
Cardiovascular: heart failure, heart arrest, tachycardia, hypertension, myocardial infarction, pulmonary embolus, bradycardia, hypotension, syncope
Digestive System: oral moniliasis, anorexia, cholestatic jaundice/jaundice, flatulence, ileus, hepatic failure, dyspepsia, intestinal obstruction
Hemic/Lymphatic: anemia, hypochromic anemia, hypervolemia
Metabolic/Nutritional: peripheral edema, hypoxia
Nervous System: insomnia, agitation, delirium, confusion, dizziness, seizure, nervousness, paresthesia, hallucinations, somnolence, anxiety, depression, asthenia
Respiratory: respiratory disorder, dyspnea, pleural effusion, asthma, cough increased, lung edema
Skin and Appendages: urticaria, sweating, skin ulcer
Urogenital System: dysuria, kidney failure, vaginal moniliasis, urinary incontinence
Laboratory Abnormalities
Laboratory abnormalities that occurred in greater than 0.2% of the meropenem for injection-treated patients were as follows:
Hepatic: increased alanine transaminase (ALT), aspartate transaminase (AST), alkaline phosphatase, lactate dehydrogenase (LDH), and bilirubin
Hematologic: increased platelets, increased eosinophils, decreased platelets, decreased hemoglobin, decreased hematocrit, decreased white blood cell (WBC), shortened prothrombin time and shortened partial thromboplastin time, leukocytosis, hypokalemia
Renal: increased creatinine and increased blood urea nitrogen (BUN)
Urinalysis: presence of red blood cells
6.2 Post-marketing Experience
The following adverse reactions have been identified during post-approval use of meropenem, including meropenem for injection. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Worldwide post-marketing adverse reactions not otherwise listed under the Clinical Trials Experience subsection [see Adverse Reactions (6.1)] and reported as possibly, probably, or definitely drug related are listed within each body system in order of decreasing severity.
Blood and Lymphatic System Disorders: agranulocytosis, neutropenia, and leukopenia; a positive direct or indirect Coombs test, and hemolytic anemia.
Immune System Disorders: angioedema
Skin and Subcutaneous Disorders: Stevens-Johnson syndrome, toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms (DRESS), erythema multiforme and acute generalized exanthematous pustulosis.
Musculoskeletal Disorders: rhabdomyolysis
-
7 DRUG INTERACTIONS
7.1 Probenecid
Probenecid competes with meropenem for active tubular secretion, resulting in increased plasma concentrations of meropenem. Co-administration of probenecid with meropenem is not recommended.
7.2 Valproic Acid
Case reports in the literature have shown that co-administration of carbapenems, including meropenem, to patients receiving valproic acid or divalproex sodium results in a reduction in valproic acid concentrations. The valproic acid concentrations may drop below the therapeutic range as a result of this interaction, therefore increasing the risk of breakthrough seizures. Although the mechanism of this interaction is unknown, data from in vitro and animal studies suggest that carbapenems may inhibit the hydrolysis of valproic acid’s glucuronide metabolite (VPA-g) back to valproic acid, thus decreasing the serum concentrations of valproic acid. If administration of meropenem for injection is necessary, then supplemental anti-convulsant therapy should be considered [see Warnings and Precautions(5.4)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are insufficient human data to establish whether there is a drug-associated risk of major birth defects or miscarriages with meropenem in pregnant women.
No fetal toxicity or malformations were observed in pregnant rats and Cynomolgus monkeys administered intravenous meropenem during organogenesis at doses up to 2.4 and 2.3 times the maximum recommended human dose (MRHD) based on body surface area comparison, respectively. In rats administered intravenous meropenem in late pregnancy and during the lactation period, there were no adverse effects on offspring at doses equivalent to approximately 3.2 times the MRHD based on body surface area comparison (see Data).
The background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Meropenem administered to pregnant rats during organogenesis (Gestation Day 6 to Gestation Day 17) in intravenous doses of 240, 500, and 750 mg/kg/day was associated with mild maternal weight loss at all doses, but did not produce malformations or fetal toxicity. The no-observed-adverse-effect-level (NOAEL) for fetal toxicity in this study was considered to be the high dose of 750 mg/kg/day (equivalent to approximately 2.4 times the MRHD of 1 gram every 8 hours based on body surface area comparison). Meropenem administered intravenously to pregnant Cynomolgus monkeys during organogenesis from Day 20 to 50 after mating at doses of 120, 240, and 360 mg/kg/day did not produce maternal or fetal toxicity at the NOAEL dose of 360 mg/kg/day (approximately 2.3 times the MRHD based on body surface area comparison).
In a peri-postnatal study in rats described in the published literature1, intravenous meropenem was administered to dams from Gestation Day 17 until Lactation Day 21 at doses of 240, 500, and 1000 mg/kg/day. There were no adverse effects in the dams and no adverse effects in the first generation offspring (including developmental, behavioral, and functional assessments and reproductive parameters) except that female offspring exhibited lowered body weights which continued during gestation and nursing of the second generation offspring. Second generation offspring showed no meropenem-related effects. The NOAEL value was considered to be 1000 mg/kg/day (approximately 3.2 times the MRHD based on body surface area comparisons).
8.2 Lactation
Risk Summary
Meropenem has been reported to be excreted in human milk. No information is available on the effects of meropenem on the breast-fed child or on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for meropenem for injection and any potential adverse effects on the breast-fed child from meropenem for injection or from the underlying maternal conditions.
8.4 Pediatric Use
The safety and effectiveness of Meropenem for Injection have been established for pediatric patients 3 months of age and older for the treatment of bacterial meningitis caused by Haemophilus influenzae, Neisseria meningitidis and penicillin-susceptible isolates of Streptococcus pneumoniae, and the information on this use is discussed throughout the labeling.
The safety and effectiveness of Meropenem for Injection have not been established in pediatric patients younger than 3 months of age.
-
10 OVERDOSAGE
In mice and rats, large intravenous doses of meropenem (2200 mg/kg to 4000 mg/kg) have been associated with ataxia, dyspnea, convulsions, and mortalities.
Intentional overdosing of meropenem for injection is unlikely, although accidental overdosing might occur if large doses are given to patients with reduced renal function. The largest dose of meropenem administered in clinical trials has been 2 grams given intravenously every 8 hours. At this dosage, no adverse pharmacological effects or increased safety risks have been observed.
Limited postmarketing experience indicates that if adverse events occur following overdosage, they are consistent with the adverse event profile described in the Adverse Reactions section and are generally mild in severity and resolve on withdrawal or dose reduction. Consider symptomatic treatments. In individuals with normal renal function, rapid renal elimination takes place. Meropenem and its metabolite are readily dialyzable and effectively removed by hemodialysis; however, no information is available on the use of hemodialysis to treat overdosage.
-
11 DESCRIPTION
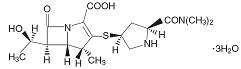
Meropenem for Injection contains meropenem a synthetic carbapenem antibacterial. Meropenem is (4R,5S,6S)-3-[[(3S,5S)-5-(Dimethylcarbamoyl)-3-pyrrolidinyl]thio]-6-[(1R)-1-hydroxyethyl]-4-methyl-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid trihydrate. Its empirical formula is C17H25N3O5S3H2O with a molecular weight of 437.52. Its structural formula is:
Meropenem is soluble in 5% monobasic potassium phosphate solution, sparingly soluble in water, very slightly soluble in hydrated ethanol, and practically insoluble in acetone or ether.
Meropenem for Injection is a white to pale yellow sterile powder for intravenous administration. Each vial contains meropenem equivalent to 2 grams and 416 mg of sodium bicarbonate, anhydrous. When re-constituted as instructed Meropenem for Injection will deliver 2 grams of meropenem (on anhydrous basis) and approximately 180 mg of sodium as sodium carbonate (7.8 mEq) [see Dosage and Administration (2.2)]. The solution varies from colorless to yellow depending on the concentration. The pH of freshly constituted solutions is between 7.3 and 8.3.
-
12 CLINICAL PHARMACOLOGY
12.2 Pharmacodynamics
The percentage of time of a dosing interval that unbound plasma concentration of meropenem exceeds the meropenem minimum inhibitory concentration (MIC) against the infecting organism has been shown to best correlate with efficacy in animal and in vitro models of infection.
12.3 Pharmacokinetics
Plasma Concentrations
At the end of a 30-minute intravenous infusion of a single dose of meropenem for injection in healthy volunteers, mean peak plasma concentrations of meropenem are approximately 49 mcg/mL (range 39 to 58) for the 1 gram dose.
Following intravenous doses of 500 mg, mean plasma concentrations of meropenem usually decline to approximately 1 mcg/mL at 6 hours after administration.
No accumulation of meropenem in plasma was observed with regimens using 1 gram administered every 6 hours in healthy volunteers with normal renal function.
Distribution
The plasma protein binding of meropenem is approximately 2%.
After a single intravenous dose of meropenem for injection, the highest mean concentrations of meropenem were found in tissues and fluids at 1 hour (0.5 hours to 1.5 hours) after the start of infusion, except where indicated in the tissues and fluids listed in Table 2 below.
Table 1: Meropenem Concentrations in Selected Tissues (Highest Concentrations Reported) - * at 1 hour unless otherwise noted
- † obtained from blister fluid
- ‡ in pediatric patients of age 5 months to 8 years
- § in pediatric patients of age 1 month to 15 years (Meropenem for Injection is not approved for use in pediatric patients younger than 3 months of age)
Tissue
Intravenous Dose
(gram)
Number of Samples
Mean [mcg/mL
or mcg/(gram)]*
Range [mcg/mL or
mcg/(gram)]
Endometrium
0.5
7
4.2
1.7 to 10.2
Myometrium
0.5
15
3.8
0.4 to 8.1
Ovary
0.5
8
2.8
0.8 to 4.8
Cervix
0.5
2
7
5.4 to 8.5
Fallopian tube
0.5
9
1.7
0.3 to 3.4
Skin
0.5
22
3.3
0.5 to 12.6
Interstitial fluid†
0.5
9
5.5
3.2 to 8.6
Skin
1
10
5.3
1.3 to 16.7
Interstitial fluid†
1
5
26.3
20.9 to 37.4
Colon
1
2
2.6
2.5 to 2.7
Bile
1
7
14.6 (3 hours)
4 to 25.7
Gall bladder
1
1
-
3.9
Peritoneal fluid
1
9
30.2
7.4 to 54.6
Lung
1
2
4.8 (2 hours)
1.4 to 8.2
Bronchial mucosa
1
7
4.5
1.3 to 11.1
Muscle
1
2
6. 1 (2 hours)
5.3 to 6.9
Fascia
1
9
8.8
1.5 to 20
Heart valves
1
7
9.7
6.4 to 12.1
Myocardium
1
10
15.5
5.2 to 25.5
CSF (inflamed)
20 mg/kg‡
40 mg/kg§
8
5
1.1 (2 hours)
3.3 (3 hours)
0.2 to 2.8
0.9 to 6.5
CSF (uninflamed)
1
4
0.2 (2 hours)
0.1 to 0.3
Elimination
In subjects with normal renal function, the elimination half-life of meropenem is approximately 1 hour. The elimination half-life for meropenem was approximately 1.5 hours in pediatric patients 3 months to 2 years of age.
Metabolism
There is one metabolite of meropenem that is microbiologically inactive.
Excretion
Meropenem is primarily excreted unchanged by the kidneys. Approximately 70% (50% to 75%) of the dose is excreted unchanged within 12 hours. A further 28% is recovered as the microbiologically inactive metabolite. Fecal elimination represents only approximately 2% of the dose. The measured renal clearance and the effect of probenecid show that meropenem undergoes both filtration and tubular secretion. Urinary concentrations of meropenem in excess of 10 mcg/mL are maintained for up to 5 hours after a 500 mg dose.
Specific Populations
Patients with Hepatic Impairment
A pharmacokinetic study with meropenem for injection in patients with hepatic impairment has shown no effects of liver disease on the pharmacokinetics of meropenem.
Patients with Renal Impairment
Pharmacokinetic (PK) studies with meopenem for injection in adult patients with renal impairment have shown that the reduction in plasma clearance of meropenem correlates with creatinine clearance. The effect of renal impairment on the PK of meropenem has not been studied in the pediatric patients. Therefore, there is no experience in pediatric patients with renal impairment [see Dosage and Administration (2.3)].
Drug Interaction Studies
Probenecid competes with meropenem for active tubular secretion and thus inhibits the renal excretion of meropenem. Following administration of probenecid with meropenem, the mean systemic exposure increased 56% and the mean elimination half-life increased 38% [see Drug Interactions (7.1)].
12.4 Microbiology
Mechanism of Action
The bactericidal activity of meropenem results from the inhibition of cell wall synthesis. Meropenem penetrates the cell wall of most gram-positive and gram-negative bacteria to bind penicillin-binding-protein (PBP) targets. Meropenem binds to PBPs 2, 3 and 4 of Escherichia coli and Pseudomonas aeruginosa; and PBPs 1, 2 and 4 of Staphylococcus aureus. Bactericidal concentrations (defined as a 3 log10 reduction in cell counts within 12 hours to 24 hours) are typically 1-2 times the bacteriostatic concentrations of meropenem, with the exception of Listeria monocytogenes, against which lethal activity is not observed.
Meropenem does not have in vitro activity against methicillin-resistant Staphylococcus aureus (MRSA) or methicillin-resistant Staphylococcus epidermidis (MRSE).
Resistance
There are several mechanisms of resistance to carbapenems: 1) decreased permeability of the outer membrane of gram-negative bacteria (due to diminished production of porins) causing reduced bacterial uptake, 2) reduced affinity of the target PBPs, 3) increased expression of efflux pump components, and 4) production of antibacterial drug-destroying enzymes (carbapenemases, metallo-β-lactamases).
Cross-resistance is sometimes observed with isolates resistant to other carbapenems.
Interaction with Other Antimicrobials
In vitro tests show meropenem to act synergistically with aminoglycoside antibacterial drugs against some isolates of Pseudomonas aeruginosa.
Antimicrobial Activity
Meropenem has been shown to be active against most isolates of the following microorganisms, both in vitro and in clinical infections [see Indications and Usage (1)].
Gram-positive bacteria
Streptococcus pneumoniae (penicillin-susceptible isolates only)
Gram-negative bacteria
Haemophilus influenzae
Neisseria meningitidis
The following in vitro data are available, but their clinical significance is unknown. At least 90% of the following bacteria exhibit an in vitro minimum inhibitory concentration (MIC) less than or equal to the susceptible breakpoint for meropenem against isolates of similar genus or organism group. However, the efficacy of meropenem in treating clinical infections caused by these bacteria have not been established in adequate and well-controlled clinical trials.
Gram-positive bacteria
Staphylococcus epidermidis (methicillin-susceptible isolates only)
Gram-negative bacteria
Aeromonas hydrophila
Campylobacter jejuni
Citrobacter freundii
Citrobacter koseri
Enterobacter cloacae
Hafnia alvei
Klebsiella oxytoca
Moraxella catarrhalis
Morganella morganii
Pasteurella multocida
Proteus vulgaris
Serratia marcescens
Anaerobic bacteria
Bacteroides ovatus
Bacteroides uniformis
Bacteroides vulgatus
Clostridium difficile
Clostridium perfringens
Eggerthella lenta
Fusobacterium species
Prevotella intermedia
Prevotella melaninogenica
Propionibacterium acnes
Susceptibility TestingFor specific information regarding susceptibility test interpretive criteria and associated test methods and quality control standards recognized by FDA for this drug, please see: https://www.fda.gov/STIC.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Carcinogenesis studies have not been performed.
Mutagenesis
Genetic toxicity studies were performed with meropenem using the bacterial reverse mutation test, the Chinese hamster ovary HGPRT assay, cultured human lymphocytes cytogenic assay, and the mouse micronucleus test. There was no evidence of mutagenic potential found in any of these tests.
Impairment of Fertility
In fertility studies, intravenous meropenem was administered to male rats beginning 11 weeks before mating and throughout mating and to female rats from 2 weeks before mating through Gestation Day 7 at doses of 240, 500, and 1000 mg/kg/day. There was no evidence of impaired fertility at doses up to 1000 mg/kg/day (on the basis of body surface area comparison, approximately 3.2 times to the MRHD of 1 gram every 8 hours).
-
14 CLINICAL STUDIES
14.1 Bacterial Meningitis
Four hundred forty-six patients (397 pediatric patients 3 months to less than 17 years of age) were enrolled in 4 separate clinical trials and randomized to treatment with meropenem for injection (n = 225) at a dosage of 40 mg/kg every 8 hours or a comparator drug, i.e., cefotaxime (n = 187) or ceftriaxone (n = 34), at the approved dosing regimens. A comparable number of patients were found to be clinically evaluable (ranging from 61 to 68%) and with a similar distribution of pathogens isolated on initial CSF culture.
A patient was defined as clinically not cured if any one of the following three criteria were met:
- 1 At the 5-7 week post-completion of therapy visit, the patient had any one of the following: moderate to severe motor, behavior or development deficits, hearing loss of greater than 60 decibels in one or both ears, or blindness.
- 2 During therapy the patient’s clinical status necessitated the addition of other antibacterial drugs.
- 3 Either during or post-therapy, the patient developed a large subdural effusion needing surgical drainage, or a cerebral abscess, or a bacteriologic relapse.
Using these criteria, the following efficacy rates were obtained, per organism (noted in Table 3). The values represent the number of patients clinically cured/number of clinically evaluable patients, with the percent cure in parentheses. Sequelae were the most common reason patients were assessed as clinically not cured.
Table 3: Efficacy rates by Pathogen in the Clinically Evaluable Population with Bacterial Meningitis
- * (+) β-lactamase-producing
- † (-/NT) non-β-lactamase-producing or not tested
Microorganisms
Meropenem for Injection
Comparator
S. pneumoniae
17/24 (71)
19/30 (63)
H. influenzae (+)*
8/10 (80)
6/6 (100)
H. influenzae (-/NT) †
44/59 (75)
44/60 (73)
N. meningitidis
30/35 (86)
35/39 (90)
Total (including others)
102/131 (78)
108/140 (77)
Five patients were found to be bacteriologically not cured, 3 in the comparator group (1 relapse and 2 patients with cerebral abscesses) and 2 in the meropenem for injection group (1 relapse and 1 with continued growth of Pseudomonas aeruginosa).
With respect to hearing loss, 263 of the 271 evaluable patients had at least one hearing test performed post-therapy. Table 4 shows the degree of hearing loss between the meropenem for injection-treated patients and comparator-treated patients.
Table 4: Hearing Loss at Post-Therapy in the Evaluable Population Treated with Meropenem
Degree of Hearing Loss
(in one or both ears)
Meropenem for Injection
n = 128
Comparator
n = 135
No loss
61%
56%
20 to 40 decibels
20%
24%
Greater than 40 to 60 decibels
8%
7%
Greater than 60 decibels
9%
10%
- 15 REFERENCES
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Meropenem for Injection is supplied in a single-dose vial containing meropenem as a white to light yellow powder to deliver 2 grams of meropenem for intravenous administration after reconstitution. The dry powder should be stored at 20º C to 25ºC (68º F to 77ºF). Brief exposure to 15ºC to 30ºC (59ºF to 86ºF) is permitted [see USP Controlled Room Temperature].
NDC: 44567-402-01 2 gram single-dose injection vial
NDC: 44567-402-06 2 gram single-dose injection vial packaged in a carton of 6
The container closure is not made with natural rubber latex.
-
17 PATIENT COUNSELING INFORMATION
Antibacterial Resistance
Patients should be counseled that antibacterial drugs including Meropenem for Injection should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When Meropenem for Injection is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by Meropenem for Injection or other antibacterial drugs in the future [see Warnings and Precautions (5.7)].
Diarrhea
Counsel patients that diarrhea is a common problem caused by antibacterial drugs which usually ends when the antibacterial drug is discontinued. Sometimes after starting treatment with antibacterial drugs, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibacterial drug. If this occurs, patients should contact their physician as soon as possible [see Warnings and Precautions (5.6)].
Risk of Breakthrough Seizures
Counsel patients to inform their physician if they are taking valproic acid or divalproex sodium. Valproic acid concentrations in the blood may drop below the therapeutic range upon co-administration with Meropenem for Injection. If treatment with Meropenem for Injection is necessary and continued, alternative or supplemental anti-convulsant medication to prevent and/or treat seizures may be needed [see Warnings and Precautions (5.4)].
Potential of Neuromotor Impairment
Patients receiving Meropenem for Injection on an outpatient basis must be alerted of adverse events such as seizures, delirium, headaches and/or paresthesias that could interfere with mental alertness and/or cause motor impairment. Until it is reasonably well established that Meropenem for Injection is well tolerated, patients should not operate machinery or motorized vehicles [see Warnings and Precautions (5.10)].
Manufactured for:
WG Critical Care, LLC
Paramus, NJ 07652
Made in Italy
-
PRINCIPAL DISPLAY PANEL
NDC: 44567-402-01
Meropenem for Injection, USP
2 grams per vial
For Intravenous Infusion Only
Single-Dose Vial – Discard Unused Portion
Rx only -
INGREDIENTS AND APPEARANCE
MEROPENEM
meropenem injection, powder, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 44567-402 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MEROPENEM (UNII: FV9J3JU8B1) (MEROPENEM ANHYDROUS - UNII:YOP6PX0BAO) MEROPENEM 2 g Inactive Ingredients Ingredient Name Strength SODIUM CARBONATE (UNII: 45P3261C7T) 180 mg Other Ingredients Ingredient Kind Ingredient Name Quantity Does not contain NATURAL LATEX RUBBER (UNII: 2LQ0UUW8IN) 0 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 44567-402-06 6 in 1 CARTON 09/08/2023 1 NDC: 44567-402-01 1 in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA215212 09/08/2023 Labeler - WG Critical Care, LLC (829274633)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.