Salsalate by Westminster Pharmaceuticals, LLC SALSALATE tablet
Salsalate by
Drug Labeling and Warnings
Salsalate by is a Prescription medication manufactured, distributed, or labeled by Westminster Pharmaceuticals, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
-
BOXED WARNING
(What is this?)
Cardiovascular Risk
- NSAIDs may cause an increased risk of serious cardiovascular thrombotic events, myocardial infarction, and stroke, which can be fatal. This risk may increase with duration of use. Patients with cardiovascular disease or risk factors for cardiovascular disease may be at greater risk (see WARNINGS and CLINICAL TRIALS).
- Salsalate tablets, USP are contraindicated for the treatment of perioperative pain in the setting of coronary artery bypass graft (CABG) surgery (see WARNINGS).
Gastrointestinal Risk
- NSAIDs cause an increased risk of serious gastrointestinal adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients are at greater risk for serious gastrointestinal events (see WARNINGS).
-
DESCRIPTION
Salsalate USP, is a nonsteroidal anti-inflammatory agent for oral administration.
Chemically, salsalate, USP (salicylsalicylic acid or 2-hydroxy-benzoic acid, 2-carboxyphenyl ester) is a dimer of salicylic acid; its structural formula is shown below.
Chemical Structure:
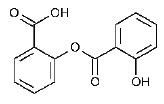
Tablets:
Inactive Ingredients: Colloidal Silicon Dioxide, D&C Yellow #10, Hypromellose, Microcrystalline Cellulose, Sodium Starch Glycolate, Stearic Acid, Titanium Dioxide, Triacetin.
-
CLINICAL PHARMACOLOGY
Salsalate is insoluble in acid gastric fluids (<0.1 mg/ml at pH 1.0), but readily soluble in the small intestine where it is partially hydrolyzed to two molecules of salicylic acid. A significant portion of the parent compound is absorbed unchanged and undergoes rapid esterase hydrolysis in the body: its half-life is about one hour. About 13% is excreted through the kidneys as a glucuronide conjugate of the parent compound, the remainder as salicylic acid and its metabolites. Thus, the amount of salicylic acid available from salsalate is about 15% less than from aspirin, when the two drugs are administered on a salicylic acid molar equivalent basis (3.6 g salsalate/5 g aspirin). Salicylic acid biotransformation is saturated at anti-inflammatory doses of salsalate. Such capacity-limited biotransformation results in an increase in the half-life of salicylic acid from 3.5 to 16 or more hours. Thus, dosing with salsalate twice a day will satisfactorily maintain blood levels within the desired therapeutic range (10 to 30 mg/100 mL) throughout the 12-hour intervals. Therapeutic blood levels continue for up to 16 hours after the last dose. The parent compound does not show capacity-limited biotransformation, nor does it accumulate in the plasma on multiple dosing. Food slows the absorption of all salicylates including salsalate.
The mode of anti-inflammatory action of salsalate and other nonsteroidal anti-inflammatory drugs is not fully defined. Although salicylic acid (the primary metabolite of salsalate) is a weak inhibitor of prostaglandin synthesis in vitro, salsalate appears to selectively inhibit prostaglandin synthesis in vivo1, providing anti-inflammatory activity equivalent to aspirin2 and indomethacin3. Unlike aspirin, salsalate does not inhibit platelet aggregation4.
The usefulness of salicylic acid, the active in vivo product of salsalate, in the treatment of arthritic disorders has been established5,6. In contrast to aspirin, salsalate causes no greater fecal gastrointestinal blood loss than placebo7.
-
INDICATIONS AND USAGE
Carefully consider the potential benefits and risks of Salsalate tablets, USP and other treatment options before deciding to use Salsalate tablets, USP. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS).
Salsalate, USP is indicated for relief of the signs and symptoms of rheumatoid arthritis, osteoarthritis and related rheumatic disorder.
-
CONTRAINDICATIONS
Salsalate tablets, USP are contraindicated in patients with known hypersensitivity to salsalate, USP.
Salsalate tablets, USP should not be given to patients who have experienced asthma, urticaria, or allergic-type reactions after taking aspirin or other NSAIDs. Severe, rarely fatal, anaphylactic-like reactions to NSAIDs have been reported in such patients (see WARNINGS – Anaphylactoid Reactions, and PRECAUTIONS - Preexisting Asthma).
Salsalate tablets, USP are contraindicated for the treatment of peri-operative pain in the setting of coronary artery bypass graft (CABG) surgery (see WARNINGS).
-
WARNINGS
Reye's Syndrome may develop in individuals who have chicken pox, influenza, or flu symptoms. Some studies suggest a possible association between the development of Reye's Syndrome and the use of medicines containing salicylate or aspirin. Salsalate contains a salicylate and therefore is not recommended for use in patients with chicken pox, influenza, or flu symptoms.
CARDIOVASCULAR EFFECTS
Cardiovascular Thrombotic Events
Clinical trials of several COX-2 selective and nonselective NSAIDs of up to three years duration have shown an increased risk of serious cardiovascular (CV) thrombotic events, myocardial infarction, and stroke, which can be fatal. All NSAIDs, both COX-2 selective and nonselective, may have a similar risk. Patients with known CV disease or risk factors for CV disease may be at greater risk. To minimize the potential risk for an adverse CV event in patients treated with an NSAID, the lowest effective dose should be used for the shortest duration possible. Physicians and patients should remain alert for the development of such events, even in the absence of previous CV symptoms. Patients should be informed about the signs and/or symptoms of serious CV events and the steps to take if they occur.
There is no consistent evidence that concurrent use of aspirin mitigates the increased risk of serious CV thrombotic events associated with NSAID use. The concurrent use of aspirin and an NSAID does increase the risk of serious GI events (see GI WARNINGS).
Two large, controlled, clinical trials of a COX-2 selective NSAID for the treatment of pain in the first 10 to 14 days following CABG surgery found an increased incidence of myocardial infarction and stroke (see CONTRAINDICATIONS).
Hypertension
NSAIDs, including Salsalate tablets, can lead to onset of new hypertension or worsening of pre-existing hypertension, either of which may contribute to the increased incidence of CV events.
Patients taking thiazides or loop diuretics may have impaired response to these therapies when taking NSAIDs. NSAIDs, including Salsalate tablets, should be used with caution in patients with hypertension. Blood pressure (BP) should be monitored closely during the initiation of NSAID treatment and throughout the course of therapy.
Gastrointestinal Effects
Risk of Ulceration, Bleeding, and Perforation
NSAIDs, including Salsalate tablets, can cause serious gastrointestinal (GI) adverse events including inflammation, bleeding, ulceration, and perforation of the stomach, small intestine, or large intestine, which can be fatal. These serious adverse events can occur at any time, with or without warning symptoms, in patients treated with NSAIDs. Only one in five patients, who develop a serious upper GI adverse event on NSAID therapy, is symptomatic. Upper GI ulcers, gross bleeding, or perforation caused by NSAIDs occur in approximately 1% of patients treated for 3 to 6 months, and in about 2% to 4% of patients treated for one year. These trends continue with longer duration of use, increasing the likelihood of developing a serious GI event at some time during the course of therapy. However, even short-term therapy is not without risk.
NSAIDs should be prescribed with extreme caution in those with a prior history of ulcer disease or gastrointestinal bleeding. Patients with a prior history of peptic ulcer disease and/or gastrointestinal bleeding who use NSAIDs have a greater than 10-fold increased risk for developing a GI bleed compared to patients with neither of these risk factors. Other factors that increase the risk for GI bleeding in patients treated with NSAIDs include concomitant use of oral corticosteroids or anticoagulants, longer duration of NSAID therapy, smoking, use of alcohol, older age, and poor general health status. Most spontaneous reports of fatal GI events are in elderly or debilitated patients and therefore, special care should be taken in treating this population.
To minimize the potential risk for an adverse GI event in patients treated with an NSAID, the lowest effective dose should be used for the shortest possible duration. Patients and physicians should remain alert for signs and symptoms of GI ulceration and bleeding during NSAID therapy and promptly initiate additional evaluation and treatment if a serious GI adverse event is suspected. This should include discontinuation of the NSAID until a serious GI adverse event is ruled out. For high risk patients, alternate therapies that do not involve NSAIDs should be considered.
Renal Effects
Long-term administration of NSAIDs has resulted in renal papillary necrosis and other renal injury. Renal toxicity has also been seen in patients in whom renal prostaglandins have a compensatory role in the maintenance of renal perfusion. In these patients, administration of a nonsteroidal anti-inflammatory drug may cause a dose-dependent reduction in prostaglandin formation and, secondarily, in renal blood flow, which may precipitate overt renal decompensation. Patients at greatest risk of this reaction are those with impaired renal function, heart failure, liver dysfunction, those taking diuretics and ACE inhibitors, and the elderly. Discontinuation of NSAID therapy is usually followed by recovery to the pretreatment state.
Advanced Renal Disease
No information is available from controlled clinical studies regarding the use of Salsalate tablets, USP in patients with advanced renal disease. Therefore, treatment with Salsalate tablets, USP are not recommended in these patients with advanced renal disease. If Salsalate tablets, USP therapy must be initiated, close monitoring of the patient's renal function is advisable.
Anaphylactoid Reactions
As with other NSAIDs, anaphylactoid reactions may occur in patients without known prior exposure to Salsalate tablets, USP. Salsalate tablets, USP should not be given to patients with the aspirin triad. This symptom complex typically occurs in asthmatic patients who experience rhinitis with or without nasal polyps, or who exhibit severe, potentially fatal bronchospasm after taking aspirin or other NSAIDs (see CONTRAINDICATIONS and PRECAUTIONS - Preexisting Asthma).
Emergency help should be sought in cases where an anaphylactoid reaction occurs.
Serious Skin Reactions
Drug Rash with Eosinophilia and Systemic Symptoms (DRESS)
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) has been reported in patients taking NSAIDs such as Salsalate tablets, USP. Some of these events have been fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling. Other clinical manifestations may include hepatitis, nephritis, hematological abnormalities, myocarditis, or myositis. Sometimes symptoms of DRESS may resemble an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its presentation, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, discontinue Salsalate tablets, USP and evaluate the patient immediately.
NSAIDs, including Salsalate tablets, USP, can cause serious skin adverse events such as exfoliative dermatitis, Stevens-Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. These serious events may occur without warning. Patients should be informed about the signs and symptoms of serious skin manifestations and use of the drug should be discontinued at the first appearance of skin rash or any other sign of hypersensitivity.
Fetal Toxicity
Premature Closure of Fetal Ductus Arteriosus
Avoid use of NSAIDs, including Salsalate tablets, USP, in pregnant women at about 30 weeks gestation and later. NSAIDs, including Salsalate tablets, USP, increase the risk of premature closure of the fetal ductus arteriosus at approximately this gestational age.
Oligohydramnios/Neonatal Renal Impairment
Use of NSAIDs, including Salsalate tablets, USP, at about 20 weeks gestation or later in pregnancy may cause fetal renal dysfunction leading to oligohydramnios and, in some cases, neonatal renal impairment. These adverse outcomes are seen, on average, after days to weeks of treatment, although oligohydramnios has been infrequently reported as soon as 48 hours after NSAID initiation. Oligohydramnios is often, but not always, reversible with treatment discontinuation. Complications of prolonged oligohydramnios may, for example, include limb contractures and delayed lung maturation.
In some postmarketing cases of impaired neonatal renal function, invasive procedures such as exchange transfusion or dialysis were required.
If NSAID treatment is necessary between about 20 weeks and 30 weeks gestation, limit Salsalate tablets, USP use to the lowest effective dose and shortest duration possible. Consider ultrasound monitoring of amniotic fluid if Salsalate tablets, USP treatment extends beyond 48 hours.
Discontinue Salsalate tablets, USP if oligohydramnios occurs and follow up according to clinical practice [see Use in Specific Populations].
-
PRECAUTIONS
General
Patients on treatment with salsalate should be warned not to take other salicylates so as to avoid potentially toxic concentrations. Great care should be exercised when salsalate is prescribed in the presence of chronic renal insufficiency or peptic ulcer disease. Protein binding of salicylic acid can be influenced by nutritional status, competitive binding of other drugs, and fluctuations in serum proteins caused by disease (rheumatoid arthritis, etc.).
Although cross reactivity, including bronchospasm, has been reported occasionally with non-acetylated salicylates, including salsalate, in aspirin-sensitive patients8,9, salsalate is less likely than aspirin to induce asthma in such patients10.
Salsalate tablets, USP cannot be expected to substitute for corticosteroids or to treat corticosteroid insufficiency. Abrupt discontinuation of corticosteroids may lead to disease exacerbation. Patients on prolonged corticosteroid therapy should have their therapy tapered slowly if a decision is made to discontinue corticosteroids.
The pharmacological activity of Salsalate tablets, USP in reducing [fever and] inflammation may diminish the utility of these diagnostic signs in detecting complications of presumed noninfectious, painful conditions.
Hepatic Effects
Borderline elevations of one or more liver tests may occur in up to 15% of patients taking NSAIDs including Salsalate tablets, USP. These laboratory abnormalities may progress, may remain unchanged, or may be transient with continuing therapy. Notable elevations of ALT or AST (approximately three or more times the upper limit of normal) have been reported in approximately 1% of patients in clinical trials with NSAIDs. In addition, rare cases of severe hepatic reactions, including jaundice and fatal fulminant hepatitis, liver necrosis and hepatic failure, some of them with fatal outcomes have been reported.
A patient with symptoms and/or signs suggesting liver dysfunction, or in whom an abnormal liver test has occurred, should be evaluated for evidence of the development of a more severe hepatic reaction while on therapy with Salsalate tablets, USP. If clinical signs and symptoms consistent with liver disease develop, or if systemic manifestations occur (e.g., eosinophilia, rash, etc.), Salsalate tablets, USP should be discontinued.
Hematological Effects
Anemia is sometimes seen in patients receiving NSAIDs, including Salsalate tablets, USP. This may be due to fluid retention, occult or gross GI blood loss, or an incompletely described effect upon erythropoiesis. Patients on long-term treatment with NSAIDs, including Salsalate tablets, USP, should have their hemoglobin or hematocrit checked if they exhibit any signs or symptoms of anemia.
NSAIDs inhibit platelet aggregation and have been shown to prolong bleeding time in some patients. Unlike aspirin, their effect on platelet function is quantitatively less, of shorter duration, and reversible. Patients receiving Salsalate tablets, USP, who may be adversely affected by alterations in platelet function, such as those with coagulation disorders or patients receiving anticoagulants, should be carefully monitored.
Preexisting Asthma
Patients with asthma may have aspirin-sensitive asthma. The use of aspirin in patients with aspirin-sensitive asthma has been associated with severe bronchospasm which can be fatal. Since cross reactivity, including bronchospasm, between aspirin and other nonsteroidal anti-inflammatory drugs has been reported in such aspirin-sensitive patients, Salsalate tablets, USP should not be administered to patients with this form of aspirin sensitivity and should be used with caution in patients with preexisting asthma.
INFORMATION FOR PATIENTS
Patients should be informed of the following information before initiating therapy with an NSAID and periodically during the course of ongoing therapy. Patients should also be encouraged to read the NSAID Medication Guide that accompanies each prescription dispensed.
1. Salsalate tablets, USP, like other NSAIDs, may cause serious CV side effects, such as MI or stroke, which may result in hospitalization and even death. Although serious CV events can occur without warning symptoms, patients should be alert for the signs and symptoms of chest pain, shortness of breath, weakness, slurring of speech, and should ask for medical advice when observing any indicative sign or symptoms. Patients should be apprised of the importance of this follow-up (see WARNINGS, Cardiovascular Effects).
2. Salsalate tablets, USP, like other NSAIDs, can cause GI discomfort and, rarely, serious GI side effects, such as ulcers and bleeding, which may result in hospitalization and even death. Although serious GI tract ulcerations and bleeding can occur without warning symptoms, patients should be alert for the signs and symptoms of ulcerations and bleeding, and should ask for medical advice when observing any indicative sign or symptoms including epigastric pain, dyspepsia, melena, and hematemesis. Patients should be apprised of the importance of this follow-up (see WARNINGS, Gastrointestinal Effects: Risk of Ulceration, Bleeding, and Perforation).
3. Salsalate tablets, USP like other NSAIDs, can cause serious skin side effects such as exfoliative dermatitis, SJS, and TEN, which may result in hospitalizations and even death. Although serious skin reactions may occur without warning, patients should be alert for the signs and symptoms of skin rash and blisters, fever, or other signs of hypersensitivity such as itching, and should ask for medical advice when observing any indicative signs or symptoms. Patients should be advised to stop the drug immediately if they develop any type of rash and contact their physicians as soon as possible.
4. Patients should promptly report signs or symptoms of unexplained weight gain or edema to their physicians.
5. Patients should be informed of the warning signs and symptoms of hepatotoxicity (e.g., nausea, fatigue, lethargy, pruritus, jaundice, right upper quadrant tenderness, and "flu-like" symptoms). If these occur, patients should be instructed to stop therapy and seek immediate medical therapy.
6. Patients should be informed of the signs of an anaphylactoid reaction (e.g. difficulty breathing, swelling of the face or throat). If these occur, patients should be instructed to seek immediate emergency help (see WARNINGS).
7. In late pregnancy, as with other NSAIDs, Salsalate tablets, USP should be avoided because it will cause premature closure of the ductus arteriosus.
LABORATORY TESTS
Plasma salicylic acid concentrations should be periodically monitored during long-term treatment with salsalate to aid maintenance of therapeutically effective levels: 10 to 30 mg/100 mL. Toxic manifestations are not usually seen until plasma concentrations exceed 30 mg/100 mL (see OVERDOSAGE). Urinary pH should also be regularly monitored: sudden acidification, as from pH 6.5 to 5.5, can double the plasma level, resulting in toxicity.
Because serious GI tract ulcerations and bleeding can occur without warning symptoms, physicians should monitor for signs or symptoms of GI bleeding. Patients on long-term treatment with NSAIDs, should have their CBC and a chemistry profile checked periodically. If clinical signs and symptoms consistent with liver or renal disease develop, systemic manifestations occur (e.g., eosinophilia, rash, etc.) or if abnormal liver tests persist or worsen, Salsalate tablets, USP should be discontinued.
DRUG INTERACTIONS
ACE-inhibitors
Reports suggest that NSAIDs may diminish the antihypertensive effect of ACE-inhibitors. This interaction should be given consideration in patients taking NSAIDs concomitantly with ACE-inhibitors.
Aspirin
Salicylates antagonize the uricosuric action of drugs used to treat gout. ASPIRIN AND OTHER SALICYLATE DRUGS WILL BE ADDITIVE TO SALSALATE AND MAY INCREASE PLASMA CONCENTRATIONS OF SALICYLIC ACID TO TOXIC LEVELS. Drugs and foods that raise urine pH will increase renal clearance and urinary excretion of salicylic acid, thus lowering plasma levels: acidifying drugs or foods will decrease urinary excretion and increase plasma levels. Salicylates given concomitantly with anticoagulant drugs may predispose to systemic bleeding. Salicylates may enhance the hypoglycemic effect of oral antidiabetic drugs of the sulfonylurea class. Salicylate competes with a number of drugs for protein binding sites, notably penicillin, thiopental, thyroxine, triiodothyronine, phenytoin, sulfinpyrazone, naproxen, warfarin, methotrexate, and possibly corticosteroids.
[When Salsalate tablets, USP are administered with aspirin, its protein binding is reduced, although the clearance of free Salsalate tablets, USP are not altered. The clinical significance of this interaction is not known; however,] as with other NSAIDs, concomitant administration of salsalate and aspirin is not generally recommended because of the potential of increased adverse effects.
Furosemide
Clinical studies, as well as post marketing observations, have shown that Salsalate tablets, USP can reduce the natriuretic effect-of furosemide and thiazides in some patients. This response has been attributed to inhibition of renal prostaglandin synthesis. During concomitant therapy with NSAIDs, the patient should be observed closely for signs of renal failure (see PRECAUTIONS, Renal Effects), as well as to assure diuretic efficacy.
Lithium
NSAIDs have produced an elevation of plasma lithium levels and a reduction in renal lithium clearance. The mean minimum lithium concentration increased 15% and the renal clearance was decreased by approximately 20%. These effects have been attributed to inhibition of renal prostaglandin synthesis by the NSAID. Thus, when NSAIDs and lithium are administered concurrently, subjects should be observed carefully for signs of lithium toxicity.
Drug/Laboratory Test Interactions
Salicylate competes with thyroid hormone for binding to plasma proteins, which may be reflected in a depressed plasma T4 value in some patients; thyroid function and basal metabolism are unaffected.
Carcinogenesis
No long-term animal studies have been performed with salsalate to evaluate its carcinogenic potential.
Pregnancy Category C
Risk Summary
Use of NSAIDs, including Salsalate tablets, USP, can cause premature closure of the fetal ductus arteriosus and fetal renal dysfunction leading to oligohydramnios and, in some cases, neonatal renal impairment. Because of these risks, limit dose and duration of Salsalate tablets, USP use between about 20 and 30 weeks of gestation, and avoid Salsalate tablets, USP use at about 30 weeks of gestation and later in pregnancy.
Premature Closure of Fetal Ductus Arteriosus
Use of NSAIDs, including Salsalate tablets, USP, at about 30 weeks gestation or later in pregnancy increases the risk of premature closure of the fetal ductus arteriosus.
Oligohydramnios/Neonatal Renal Impairment
Use of NSAIDs at about 20 weeks gestation or later in pregnancy has been associated with cases of fetal renal dysfunction leading to oligohydramnios, and in some cases, neonatal renal impairment.
Data from observational studies regarding other potential embryofetal risks of NSAID use in women in the first or second trimesters of pregnancy are inconclusive. In the general U.S. population, all clinically recognized pregnancies, regardless of drug exposure, have a background rate of 2-4% for major malformations, and 15-20% for pregnancy loss. Animal reproduction studies conducted in rats and rabbits have not demonstrated evidence of developmental abnormalities. However, animal reproduction studies are not always predictive of human response.
There are no adequate and well-controlled studies in pregnant women. Because of the known effects of nonsteroidal anti-inflammatory drugs on the fetal cardiovascular system (closure of ductus arteriosus), use during pregnancy (particularly late pregnancy) should be avoided. Based on animal data, prostaglandins have been shown to have an important role in endometrial vascular permeability, blastocyst implantation, and decidualization. In animal studies, administration of prostaglandin synthesis inhibitors such as [active moiety], resulted in increased pre- and post-implantation loss. Prostaglandins also have been shown to have an important role in fetal kidney development. In published animal studies, prostaglandin synthesis inhibitors have been reported to impair kidney development when administered at clinically relevant doses.
Clinical Considerations
Fetal/Neonatal Adverse Reactions
Premature Closure of Fetal Ductus Arteriosus
Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy, because NSAIDs, including Salsalate tablets, USP, can cause premature closure of the fetal ductus arteriosus (see Data).
Oligohydramnios/Neonatal Renal Impairment
If an NSAID is necessary at about 20 weeks gestation or later in pregnancy, limit the use to the lowest effective dose and shortest duration possible. If Salsalate tablets, USP treatment extends beyond 48 hours, consider monitoring with ultrasound for oligohydramnios. If oligohydramnios occurs, discontinue Salsalate tablets, USP and follow up according to clinical practice (see Data).
Data
Human Data
Premature Closure of Fetal Ductus Arteriosus
Published literature reports that the use of NSAIDs at about 30 weeks of gestation and later in pregnancy may cause premature closure of the fetal ductus arteriosus.
Oligohydramnios/Neonatal Renal Impairment
Published studies and postmarketing reports describe maternal NSAID use at about 20 weeks gestation or later in pregnancy associated with fetal renal dysfunction leading to oligohydramnios, and in some cases, neonatal renal impairment. These adverse outcomes are seen, on average, after days to weeks of treatment, although oligohydramnios has been infrequently reported as soon as 48 hours after NSAID initiation. In many cases, but not all, the decrease in amniotic fluid was transient and reversible with cessation of the drug. There have been a limited number of case reports of maternal NSAID use and neonatal renal dysfunction without oligohydramnios, some of which were irreversible. Some cases of neonatal renal dysfunction required treatment with invasive procedures, such as exchange transfusion or dialysis.
Methodological limitations of these postmarketing studies and reports include lack of a control group; limited information regarding dose, duration, and timing of drug exposure; and concomitant use of other medications. These limitations preclude establishing a reliable estimate of the risk of adverse fetal and neonatal outcomes with maternal NSAID use. Because the published safety data on neonatal outcomes involved mostly preterm infants, the generalizability of certain reported risks to the full-term infant exposed to NSAIDs through maternal use is uncertain.
Labor and Delivery
There exist no adequate and well-controlled studies in pregnant women. Although adverse effects on mother or infant have not been reported with salsalate use during labor, caution is advised when anti-inflammatory dosage is involved. However, other salicylates have been associated with prolonged gestation and labor, maternal and neonatal bleeding sequelae, potentiation of narcotic and barbiturate effects (respiratory or cardiac arrest in the mother), delivery problems and stillbirth. In rat studies with NSAIDs, as with other drugs known to inhibit prostaglandin synthesis, an increased incidence of dystocia, delayed parturition, and decreased pup survival occurred. The effects of Salsalate tablets, USP on labor and delivery in pregnant women are unknown.
Nursing Mothers
It is not known whether salsalate per se is excreted in human milk; salicylic acid, the primary metabolite of salsalate, has been shown to appear in human milk in concentrations approximating the maternal blood level. Thus, the infant of a mother on salsalate therapy might ingest in mother's milk 30 to 80% as much salicylate per kg body weight as the mother is taking. Accordingly, caution should be exercised when salsalate is administered to a nursing woman.
Pediatric Use
Safety and effectiveness of salsalate use in children have not been established (see WARNINGS section).
-
ADVERSE REACTIONS
In two well-controlled clinical trials, the following reversible adverse experiences characteristic of salicylates were most commonly reported with salsalate (n-280 pts; listed in descending order of frequency): tinnitus, nausea, hearing impairment, rash, and vertigo. These common symptoms of salicylates, i.e., tinnitus or reversible hearing impairment, are often used as a guide to therapy.
Although cause-and-effect relationships have not been established, spontaneous reports over a ten-year period have included the following additional medically significant adverse experiences: abdominal pain, abnormal hepatic function, anaphylactic shock, angioedema, bronchospasm, decreased creatinine clearance, diarrhea, G.I. bleeding, hepatitis, hypotension, nephritis and urticaria.
To report SUSPECTED ADVERSE REACTIONS, contact Westminster Pharmaceuticals, LLC at 1-844-221-7294 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
- DRUG ABUSE AND DEPENDENCE
-
OVERDOSAGE
Death has followed ingestion of 10 to 30 g of salicylates in adults, but much larger amounts have been ingested without fatal outcome.
Symptoms
The usual symptoms of salicylism - tinnitus, vertigo, headache, confusion, drowsiness, sweating, hyperventilation, vomiting and diarrhea - will occur. More severe intoxication will lead to disruption of electrolyte balance and blood pH, and hyperthermia and dehydration.
Treatment
Further absorption of salsalate from the G.I. tract should be prevented by emesis (syrup of ipecac), and, if necessary, by gastric lavage.
Fluid and electrolyte imbalance should be corrected by the administration of appropriate I.V. therapy. Adequate renal function should be maintained. Hemodialysis or peritoneal dialysis may be required in extreme cases.
-
DOSAGE AND ADMINISTRATION
Carefully consider the potential benefits and risks of Salsalate tablets, USP and other treatment options before deciding to use Salsalate tablets, USP. Use the lowest effective dose for the shortest duration consistent with individual patient treatment goals (see WARNINGS).
After observing the response to initial therapy with Salsalate tablets, USP, the dose and frequency should be adjusted to suit an individual patient's needs. Salsalate, USP is indicated for relief of the signs and symptoms of rheumatoid arthritis, osteoarthritis and related rheumatic disorder.
Adults
The usual dosage is 3000 mg daily, given in divided doses as follows: 1) two doses of two 750 mg tablets; 2) two doses of three 500 mg tablets; or 3) three doses of two 500 mg tablets. Some patients, e.g., the elderly, may require a lower dosage to achieve therapeutic blood concentrations and to avoid the more common side effects such as auditory.
Alleviation of symptoms is gradual, and full benefit may not be evident for 3 to 4 days, when plasma salicylate levels have achieved steady-state. There is no evidence for development of tissue tolerance (tachyphylaxis), but salicylate therapy may induce increased activity of metabolizing liver enzymes, causing a greater rate of salicyluric acid production and excretion, with a resultant increase in dosage requirement for maintenance of therapeutic serum salicylate levels.
-
HOW SUPPLIED
500 mg Tablets: Supplied in bottles of 100ct (NDC: 69367-615-01)
Appearance: Yellow, round, Biconvex tablet, debossed "615" on one side and plain on the other side.
750 mg Tablets: Supplied in bottles of 100ct (NDC: 69367-616-01)
Appearance: Yellow, capsule-shaped, Biconvex tablet, debossed "616" on one side and plain on the other side.
Dispense contents with a child-resistant closure (as required) and in a tight container as defined in the USP.
-
REFERENCES
- 1. Morris HG, Sherman NA, McQuain C, et al: Effects of Salsalate (Non- Acetylated Salicylate) and Aspirin (ASA) on Serum Prostaglandins in Humans. Ther. Drug Monit. 7:435-438, 1985.
- 2. April PA, Curran NJ, Ekohlm BP, et al: Multicenter Comparative Study of Salsalate (SSA) vs Aspirin (ASA) in Rheumatoid Arthritis (RA), Arthritis Rheumatism 30 (4 supplement):S93, 1987.
- 3. Deodhar SD, McLeod MM, Dick WC, et al: A Short-Term Comparative Trial of Salsalate and Indomethacin in Rheumatoid Arthritis. Curr. Med. Res. Opin., 5:185-188, 1977.
- 4. Estes D, Kaplan K: Lack of Platelet Effect With the Aspirin Analog, Salsalate, Arthritis and Rheumatism, 23:1303-1307, 1980.
- 5. Dick C, Dick PH, Nuki G, et al: Effect of Anti-inflammatory Drug Therapy on Clearance of 133Xe from Knee Joints of Patients with Rheumatoid Arthritis. British Med. J. 3:278-280, 1969.
- 6. Dick WC, Grayson MF, Woodburn A, et al: Indices of Inflammatory Activity. Ann. of the Rheum. Dis. 29:643-648, 1970.
- 7. Cohen A: Fecal Blood Loss and Plasma Salicylate Study of Salicylsalicylic Acid and Aspirin. J. Clin. Pharmacol. 19:242-247, 1979.
- 8. Chudwin DS, Strub M. Golden HE, et al: Sensitivity to Non-Acetylated Salicylates in a Patient with Asthma, Nasal Polyps, and Rheumatoid Arthritis. Annals of Allergy 57:133-134, 1986.
- 9. Spector SL, Wangaard CH, Farr RS: Aspirin and Concomitant Idiosyncrasies in Adult Asthmatic Patients. J. Allergy Clin. Immunol 64:500-506, 1979.
- 10. Stevenson DD, Schrank PJ, Hougham AJ, et al: Salsalate Cross Sensitivity in Aspirin-Sensitive Asthmatics. J. Allergy Clin. Immunol 81:181, 1988.
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 500 mg Tablet Bottle Label
- PRINCIPAL DISPLAY PANEL - 750 mg Tablet Bottle Label
-
INGREDIENTS AND APPEARANCE
SALSALATE
salsalate tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 69367-615 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALSALATE (UNII: V9MO595C9I) (SALSALATE - UNII:V9MO595C9I) SALSALATE 500 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SODIUM STARCH GLYCOLATE TYPE A (UNII: H8AV0SQX4D) STEARIC ACID (UNII: 4ELV7Z65AP) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIACETIN (UNII: XHX3C3X673) Product Characteristics Color YELLOW Score no score Shape ROUND Size 11mm Flavor Imprint Code 615 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 69367-615-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 11/15/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED DRUG OTHER 11/15/2023 SALSALATE
salsalate tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 69367-616 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALSALATE (UNII: V9MO595C9I) (SALSALATE - UNII:V9MO595C9I) SALSALATE 750 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SODIUM STARCH GLYCOLATE TYPE A (UNII: H8AV0SQX4D) STEARIC ACID (UNII: 4ELV7Z65AP) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIACETIN (UNII: XHX3C3X673) Product Characteristics Color YELLOW Score no score Shape OVAL Size 19mm Flavor Imprint Code 616 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 69367-616-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 11/15/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED DRUG OTHER 11/15/2023 Labeler - Westminster Pharmaceuticals, LLC (079516651)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.

