ADASUVE- loxapine aerosol, powder
Adasuve by
Drug Labeling and Warnings
Adasuve by is a Prescription medication manufactured, distributed, or labeled by Galen US Incorporated. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use ADASUVE safely and effectively. See full prescribing information for ADASUVE.
ADASUVE® (loxapine) inhalation powder, for oral inhalation use
Initial U.S. Approval: 1975WARNING: BRONCHOSPASM and INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS
See full prescribing information for complete boxed warning.
Bronchospasm
-
ADASUVE can cause bronchospasm that has the potential to lead to respiratory distress and respiratory arrest (4, 5.1)
-
ADASUVE is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the ADASUVE REMS (5.2)
- Administer ADASUVE only in an enrolled healthcare facility that has immediate access on site to supplies and personnel trained to manage acute bronchospasm and ready access to emergency response services. Facilities must have a short-acting bronchodilator, including a nebulizer and inhalation solution, for the immediate treatment of bronchospasm (5.1, 5.2)
Increased Mortality in Elderly Patients with Dementia-Related Psychosis
- Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. ADASUVE is not approved for the treatment of patients with dementia-related psychosis (5.3)
RECENT MAJOR CHANGES
INDICATIONS AND USAGE
ADASUVE is a typical antipsychotic indicated for the acute treatment of agitation associated with schizophrenia or bipolar I disorder in adults.
Efficacy was demonstrated in 2 trials in acute agitation: one in schizophrenia and one in bipolar I disorder ( 1, 14)
Limitations of Use:
ADASUVE must be administered only in an enrolled healthcare facility ( 1)
DOSAGE AND ADMINISTRATION
- Must be administered only by a healthcare professional (
2.1)
- 10 mg by oral inhalation using an inhaler (
2.1)
- Administer only a single dose within any 24-hour period (
2.1)
- Prior to administering, screen all patients for a history of pulmonary disease, and examine patients (including chest auscultation) for respiratory abnormalities (e.g. wheezing) (
2.2)
- Refer to Full Prescribing Information for important instructions on use of the ADASUVE inhaler (
2.3)
- After administration, monitor patients for signs and symptoms of bronchospasm at least every 15 minutes for at least one hour ( 2.4)
DOSAGE FORMS AND STRENGTHS
- Inhalation powder: 10 mg unit in a single-use inhaler ( 3)
CONTRAINDICATIONS
- Current diagnosis or history of asthma, chronic obstructive pulmonary disease (COPD), or other lung disease associated with bronchospasm (
4)
- Acute respiratory signs/symptoms (e.g., wheezing) (
4)
- Current use of medications to treat airways disease, such as asthma or COPD (
4)
- History of bronchospasm following ADASUVE treatment (
4)
- Known hypersensitivity to loxapine or amoxapine ( 4)
WARNINGS AND PRECAUTIONS
-
Neuroleptic Malignant Syndrome: May develop in patients treated with antipsychotic drugs. Discontinue treatment (
5.4)
-
Hypotension and Syncope: Use with caution in patients with known cardiovascular or cerebrovascular disease (
5.5)
-
Seizure: Use with caution in patients with a history of seizures or with conditions that lower the seizure threshold (
5.7)
-
Potential for Cognitive and Motor Impairment: Use caution when driving or operating machinery (
5.8)
- Cerebrovascular Adverse Reactions: Increased incidence of stroke and transient ischemic attack in elderly patients with dementia-related psychosis treated with antipsychotic drugs ( 5.9)
ADVERSE REACTIONS
Most common adverse reactions (incidence ≥ 2% and greater than placebo) were dysgeusia, sedation, and throat irritation ( 6.1)
To report SUSPECTED ADVERSE REACTIONS contact Galen US Inc. at 1-800-284-0062 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatchUSE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and Medication Guide.
Revised: 8/2017
-
ADASUVE can cause bronchospasm that has the potential to lead to respiratory distress and respiratory arrest (4, 5.1)
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
WARNING: BRONCHOSPASM and INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Dosing Information
2.2 Required Examination Prior to Dosing
2.3 Important Administration Instructions
2.4 Monitoring to Assess Safety
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Bronchospasm
5.2 ADASUVE REMS to Mitigate Bronchospasm
5.3 Increased Mortality in Elderly Patients with Dementia-Related Psychosis
5.4 Neuroleptic Malignant Syndrome
5.5 Hypotension and Syncope
5.6 Falls
5.7 Seizures
5.8 Potential for Cognitive and Motor Impairment
5.9 Cerebrovascular Reactions, Including Stroke, in Elderly Patients with Dementia-Related Psychosis
5.10 Anticholinergic Reactions Including Exacerbation of Glaucoma and Urinary Retention
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
7 DRUG INTERACTIONS
7.1 CNS Depressants
7.2 Anticholinergic Drugs
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
16.2 Restricted Access
16.3 Storage and Handling
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
-
BOXED WARNING
(What is this?)
WARNING: BRONCHOSPASM and INCREASED MORTALITY IN ELDERLY PATIENTS WITH DEMENTIA-RELATED PSYCHOSIS
Bronchospasm
ADASUVE can cause bronchospasm that has the potential to lead to respiratory distress and respiratory arrest. Administer ADASUVE only in an enrolled healthcare facility that has immediate access on site to supplies and personnel trained to manage acute bronchospasm and ready access to emergency response services [see Warnings and Precautions (5.1, 5.2)]. Facilities must have a short-acting bronchodilator (e.g. albuterol), including a nebulizer and inhalation solution, for the immediate treatment of bronchospasm. Prior to administering ADASUVE, screen patients regarding a current diagnosis, history, or symptoms of asthma, COPD and other lung diseases, and examine (including chest auscultation) patients for respiratory signs. Monitor for signs and symptoms of bronchospasm following treatment with ADASUVE [see Dosage and Administration (2.2, 2.4) and Contraindications (4)].
Because of the risk of bronchospasm, ADASUVE is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the ADASUVE REMS [see Warnings and Precautions (5.2)].
Increased Mortality in Elderly Patients with Dementia-Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. ADASUVE is not approved for the treatment of patients with dementia-related psychosis [see Warnings and Precautions (5.3)].
-
1
INDICATIONS AND USAGE
ADASUVE is a typical antipsychotic indicated for the acute treatment of agitation associated with schizophrenia or bipolar I disorder in adults.
“Psychomotor agitation” is defined in DSM-IV as “excessive motor activity associated with a feeling of inner tension.” Patients experiencing agitation often manifest behaviors that interfere with their care (e.g., threatening behaviors, escalating or urgently distressing behavior, self-exhausting behavior), leading clinicians to the use of rapidly absorbed antipsychotic medications to achieve immediate control of the agitation [see Clinical Studies (14)].
The efficacy of ADASUVE was established in one study of acute agitation in patients with schizophrenia and one study of acute agitation in patients with bipolar I disorder [see Clinical Studies (14)].
Limitations of Use:
As part of the ADASUVE REMS Program to mitigate the risk of bronchospasm, ADASUVE must be administered only in an enrolled healthcare facility [see Warnings and Precautions (5.2)].
-
2
DOSAGE AND ADMINISTRATION
2.1 Dosing Information
ADASUVE must be administered only by a healthcare professional. ADASUVE is administered by oral inhalation only. The recommended dose for acute agitation is 10 mg administered by oral inhalation, using a single-use inhaler. Administer only a single dose within a 24-hour period [see Warnings and Precautions (5.1)].
2.2 Required Examination Prior to Dosing
Prior to administering ADASUVE, screen all patients for a history of asthma, COPD, or other pulmonary disease, and examine patients (including chest auscultation) for respiratory signs (e.g. wheezing) [see Warnings and Precautions (5.1)].
2.3 Important Administration Instructions
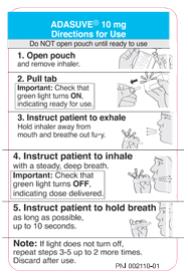
Read all of these instructions prior to administering ADASUVE.
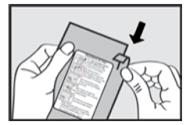
Step 1. Open the Pouch
When ready to use, tear open the foil pouch and remove the inhaler from the package (see Figure 1).
When the ADASUVE inhaler is removed from the pouch, the indicator light is off (see Figure 2).
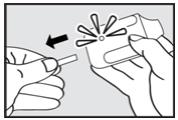
Step 2. Pull Tab
Firmly pull the plastic tab from the rear of the inhaler (see Figure 3). Check that the green light turns on. This indicates that the inhaler is ready for use. Use the inhaler within 15 minutes after removing the tab to prevent automatic deactivation of the inhaler. The green light will turn off, indicating that the inhaler is not usable. Discard the inhaler after one use.
Step 3. Explain Procedures to the Patient
Explain the administration procedures to the patient prior to use, and advise the patient that it is important to follow the instructions. Inform the patient that the inhaler may produce a flash of light and a clicking sound, and it may become warm during use. These are normal.
Step 4. Instruct the Patient to Exhale
Instruct the patient to hold the inhaler away from the mouth and breathe out fully to empty the lungs (see Figure 4).
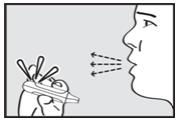
Step 5. Instruct the Patient to Inhale
Instruct the patient to put the mouthpiece of the inhaler between the lips, close the lips, and inhale through the mouthpiece with a steady deep breath (see Figure 5). Check that the green light turns off indicating that the dose has been delivered.
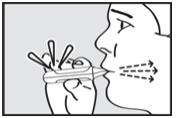
Step 6. Instruct the Patient to Hold Breath
Instruct the patient to remove the mouthpiece from the mouth and hold the breath for as long as possible, up to 10 seconds (see Figure 6).
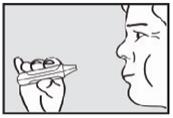
Important: If the green light remains on after the patient inhales, the dose of ADASUVE has NOT been delivered. Instruct the patient to repeat Step 4, Step 5, and Step 6 up to 2 additional times. If the green light still does not turn off, discard the inhaler and use a new one.
2.4 Monitoring to Assess Safety
Monitor the patient for signs and symptoms of bronchospasm after ADASUVE administration. Perform a physical examination, including chest auscultation, at least every 15 minutes for at least one hour after ADASUVE administration [see Warnings and Precautions (5.1)].
- 3 DOSAGE FORMS AND STRENGTHS
-
4
CONTRAINDICATIONS
ADASUVE is contraindicated in patients with the following:
- Current diagnosis or history of asthma, COPD, or other lung disease associated with bronchospasm
[see Warnings and Precautions (5.1)]
- Acute respiratory symptoms or signs (e.g., wheezing)
[see Warnings and Precautions (5.1)]
- Current use of medications to treat airways disease, such as asthma or COPD
[see Warnings and Precautions (5.1)]
- History of bronchospasm following ADASUVE treatment
[see Warnings and Precautions (5.1)]
- Known hypersensitivity to loxapine or amoxapine. Serious skin reactions have occurred with oral loxapine and amoxapine.
- Current diagnosis or history of asthma, COPD, or other lung disease associated with bronchospasm
[see Warnings and Precautions (5.1)]
-
5
WARNINGS AND PRECAUTIONS
5.1 Bronchospasm
ADASUVE can cause bronchospasm that has the potential to lead to respiratory distress and respiratory arrest [see Adverse Reactions (6.1)]. Administer ADASUVE only in an enrolled healthcare facility that has immediate access on site to supplies and personnel trained to manage acute bronchospasm and ready access to emergency response services. Facilities must have a short-acting bronchodilator (e.g. albuterol), including a nebulizer and inhalation solution, for the immediate treatment of bronchospasm [see Boxed Warning and Warnings and Precautions (5.2)].
Prior to administering ADASUVE, screen patients regarding a current diagnosis or history of asthma, COPD, and other lung disease associated with bronchospasm, acute respiratory symptoms or signs, current use of medications to treat airways disease, such as asthma or COPD; and examine patients (including chest auscultation) for respiratory abnormalities (e.g., wheezing) . Monitor patients for symptoms and signs of bronchospasm (i.e., vital signs and chest auscultation) at least every 15 minutes for a minimum of one hour following treatment with ADASUVE . ADASUVE can cause sedation, which can mask the symptoms of bronchospasm. Prior to administering ADASUVE, screen patients regarding a current diagnosis or history of asthma, COPD, and other lung disease associated with bronchospasm, acute respiratory symptoms or signs, current use of medications to treat airways disease, such as asthma or COPD; and examine patients (including chest auscultation) for respiratory abnormalities (e.g., wheezing) [See Dosage and Administration (2.2) and Contraindications (4)]. Monitor patients for symptoms and signs of bronchospasm (i.e., vital signs and chest auscultation) at least every 15 minutes for a minimum of one hour following treatment with ADASUVE [see Dosage and Administration (2.4)]. ADASUVE can cause sedation, which can mask the symptoms of bronchospasm.
Because clinical trials in patients with asthma or COPD demonstrated that the degree of bronchospasm, as indicated by changes in forced expiratory volume in 1 second (FEV1), was greater following a second dose of ADASUVE, limit ADASUVE use to a single dose within a 24 hour period. Because clinical trials in patients with asthma or COPD demonstrated that the degree of bronchospasm, as indicated by changes in forced expiratory volume in 1 second (FEV1), was greater following a second dose of ADASUVE, limit ADASUVE use to a single dose within a 24 hour period.
Advise all patients of the risk of bronchospasm. Advise them to inform the healthcare professional if they develop any breathing problems such as wheezing, shortness of breath, chest tightness, or cough following treatment with ADASUVE.Advise all patients of the risk of bronchospasm. Advise them to inform the healthcare professional if they develop any breathing problems such as wheezing, shortness of breath, chest tightness, or cough following treatment with ADASUVE.
5.2 ADASUVE REMS to Mitigate Bronchospasm
Because of the risk of bronchospasm, ADASUVE is available only through a restricted program under a REMS called the ADASUVE REMS. Required components of the ADASUVE REMS are: Because of the risk of bronchospasm, ADASUVE is available only through a restricted program under a REMS called the ADASUVE REMS. [see Boxed Warning and Warnings and Precautions (5.1)] Required components of the ADASUVE REMS are:
Healthcare facilities that dispense and administer ADASUVE must be enrolled and comply with the REMS requirements. Certified healthcare facilities must be able to provide immediate access on site to supplies and personnel trained to manage acute bronchospasm and ready access to emergency response services. Facilities must have a short-acting bronchodilator (e.g. albuterol), including a nebulizer and inhalation solution, for the immediate treatment of bronchospasm.
Wholesalers and distributors that distribute ADASUVE must distribute only to enrolled healthcare facilities.
Further information is available at www.adasuverems.com or 1-855-755-0492.Further information is available at www.adasuverems.com or 1-855-755-0492.
5.3 Increased Mortality in Elderly Patients with Dementia-Related Psychosis
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at increased risk of death. Analyses of 17 placebo-controlled trials (modal duration of 10 weeks), largely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of 1.6 to 1.7 times the risk of death in placebo-treated patients. Over the course of a typical 10-week controlled trial, the rate of death in drug-treated patients was about 4.5%, compared to a rate of about 2.6% in the placebo group. Although the cases of death were varied, most of the deaths appeared to be either cardiovascular (e.g., heart failure, sudden death) or infectious (e.g., pneumonia) in nature. Observational studies suggest that, similar to atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality. The extent to which the findings of increased mortality in observational studies can be attributed to the antipsychotic drug as opposed to some characteristic(s) of the patients is not clear. ADASUVE is not approved for the treatment of elderly patients with dementia-related psychosis [see Boxed Warning].
5.4 Neuroleptic Malignant Syndrome
Antipsychotic drugs can cause a potentially fatal symptom complex termed Neuroleptic Malignant Syndrome (NMS). Clinical manifestations of NMS include hyperpyrexia, muscle rigidity, altered mental status, and autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis, and cardiac dysrhythmia). Associated features can include elevated serum creatine phosphokinase (CPK) concentration, rhabdomyolysis, elevated serum and urine myoglobin concentration, and renal failure. NMS did not occur in the ADASUVE clinical program.
The diagnostic evaluation of patients with this syndrome is complicated. It is important to consider the presence of other serious medical conditions (e.g., pneumonia, systemic infection, heat stroke, primary CNS pathology, central anticholinergic toxicity, extrapyramidal symptoms, or drug fever).
The management of NMS should include: 1) immediate discontinuation of antipsychotic drugs and other drugs that may contribute to the underlying disorder, 2) intensive symptomatic treatment and medical monitoring, and 3) treatment of any concomitant serious medical problems. There is no general agreement about specific pharmacological treatment regimens for NMS.
If a patient requires antipsychotic drug treatment after recovery from NMS, the potential reintroduction of drug therapy should be carefully considered. The patient should be carefully monitored, since recurrences of NMS have been reported.
5.5 Hypotension and Syncope
ADASUVE can cause hypotension, orthostatic hypotension, and syncope. Use ADASUVE with caution in patients with known cardiovascular disease (history of myocardial infarction or ischemic heart disease, heart failure or conduction abnormalities), cerebrovascular disease, or conditions that would predispose patients to hypotension (dehydration, hypovolemia, or treatment with antihypertensive medications or other drugs that affect blood pressure or reduce heart rate).
In the presence of severe hypotension requiring vasopressor therapy, the preferred drugs may be norepinephrine or phenylephrine. Epinephrine should not be used, because beta stimulation may worsen hypotension in the setting of ADASUVE-induced partial alpha blockade.
In short-term (24-hour) placebo-controlled trials of patients with agitation associated with schizophrenia or bipolar I disorder, hypotension occurred in 0.4% and 0.8% in the ADASUVE 10 mg and placebo groups, respectively. There were no cases of orthostatic hypotension, postural symptoms, presyncope or syncope. A systolic blood pressure ≤ 90 mm Hg with a decrease of ≥ 20 mm Hg occurred in 1.5% and 0.8% of the ADASUVE 10 mg and placebo groups, respectively. A diastolic blood pressure ≤ 50 mm Hg with a decrease of ≥15 mm Hg occurred in 0.8% and 0.4% of the ADASUVE 10 mg and placebo groups, respectively.
In 5 Phase 1 studies in normal volunteers, the incidence of hypotension was 3% and 0% in ADASUVE 10 mg and the placebo groups, respectively. The incidence of syncope or presyncope in normal volunteers was 2.3% and 0% in the ADASUVE and placebo groups, respectively. In normal volunteers, a systolic blood pressure ≤ 90 mm Hg with a decrease of ≥ 20 mm Hg occurred in 5.3% and 1.1% in the ADASUVE and placebo groups, respectively. A diastolic blood pressure ≤ 50 mm Hg with a decrease of ≥ 15 mm Hg occurred in 7.5% and 3.3% in the ADASUVE and placebo groups, respectively.
5.6 Falls
ADASUVE may cause somnolence, postural hypotension, motor and sensory instability, which may lead to falls and, consequently, fractures or other injuries. For patients with diseases, conditions, or medications that could exacerbate these effects, complete fall risk assessments when initiating antipsychotic treatment and recurrently for patients on long-term antipsychotic therapy.
5.7 Seizures
ADASUVE lowers the seizure threshold. Seizures have occurred in patients treated with oral loxapine. Seizures can occur in epileptic patients even during antiepileptic drug maintenance therapy. In short term (24 hour), placebo-controlled trials of ADASUVE, there were no reports of seizures.
5.8 Potential for Cognitive and Motor Impairment
ADASUVE can impair judgment, thinking, and motor skills. In short-term, placebo-controlled trials, sedation and/or somnolence were reported in 12% and 10% in the ADASUVE and placebo groups, respectively. No patients discontinued treatment because of sedation or somnolence.
The potential for cognitive and motor impairment is increased when ADASUVE is administered concurrently with other CNS depressants [see Drug Interactions (7.1)]. Caution patients about operating hazardous machinery, including automobiles, until they are reasonably certain that therapy with ADASUVE does not affect them adversely.
5.9 Cerebrovascular Reactions, Including Stroke, in Elderly Patients with Dementia-Related Psychosis
In placebo-controlled trials with atypical antipsychotics in elderly patients with dementia-related psychosis, there was a higher incidence of cerebrovascular adverse reactions (stroke and transient ischemic attacks), including fatalities, compared to placebo-treated patients. ADASUVE is not approved for the treatment of patients with dementia-related psychosis [see Boxed Warning and Warnings and Precautions (5.3)].
5.10 Anticholinergic Reactions Including Exacerbation of Glaucoma and Urinary Retention
ADASUVE has anticholinergic activity, and it has the potential to cause anticholinergic adverse reactions including exacerbation of glaucoma or urinary retention. The concomitant use of other anticholinergic drugs (e.g., antiparkinson drugs) with ADASUVE could have additive effects.
-
6
ADVERSE REACTIONS
The following adverse reactions are discussed in more detail in other sections of the labeling:
- Hypersensitivity (serious skin reactions)
[see Contraindications (4)]
- Bronchospasm
[see Warnings and Precautions (5.1)]
- Increased Mortality in Elderly Patients with Dementia-Related Psychosis
[see Warnings and Precautions (5.3)]
- Neuroleptic Malignant Syndrome
[see Warnings and Precautions (5.4)]
- Hypotension and syncope
[see Warnings and Precautions (5.5)]
- Seizure
[see Warnings and Precautions (5.7)]
- Potential for Cognitive and Motor Impairment
[see Warnings and Precautions (5.8)]
- Cerebrovascular Reactions, Including Stroke, in Elderly Patients with Dementia-Related Psychosis
[see Warnings and Precautions (5.9)]
- Anticholinergic Reactions Including Exacerbation of Glaucoma and Urinary Retention [see Warnings and Precautions (5.10)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The following findings are based on pooled data from three short-term (24-hour), randomized, double-blind, placebo-controlled clinical trials (Studies 1, 2, and 3) of ADASUVE 10 mg in the treatment of patients with acute agitation associated with schizophrenia or bipolar I disorder. In the 3 trials, 259 patients received ADASUVE 10 mg, and 263 received placebo [see Clinical Studies (14)].
Commonly Observed Adverse Reactions: In the 3 trials in acute agitation, the most common adverse reactions were dysgeusia, sedation, and throat irritation. These reactions occurred at a rate of at least 2% of the ADASUVE group and at a rate greater than in the placebo group. (Refer to Table 1).
Table 1. Adverse Reactions in 3 Pooled Short-Term, Placebo-Controlled Trials (Studies 1, 2, and 3) in Patients with Schizophrenia or Bipolar Disorder
Adverse Reaction
Placebo
(n = 263)
ADASUVE
(n = 259)Dysgeusia 5% 14% Sedation 10% 12% Throat Irritation 0% 3% Airway Adverse Reactions in the 3 Trials in Acute Agitation
Agitated patients with Schizophrenia or Bipolar Disorder: In the 3 short-term (24-hour), placebo-controlled trials in patients with agitation associated with schizophrenia or bipolar disorder (Studies 1, 2, and 3), bronchospasm (which includes reports of wheezing, shortness of breath and cough) occurred more frequently in the ADASUVE group, compared to the placebo group: 0% (0/263) in the placebo group and 0.8% (2/259) in the ADASUVE 10 mg group. One patient with schizophrenia, without a history of pulmonary disease, had significant bronchospasm requiring rescue treatment with a bronchodilator and oxygen.
Bronchospasm and Airway Adverse Reactions in Pulmonary Safety Trials
Clinical pulmonary safety trials demonstrated that ADASUVE can cause bronchospasm as measured by FEV1, and as indicated by respiratory signs and symptoms in the trials. In addition, the trials demonstrated that patients with asthma or other pulmonary diseases, such as COPD are at increased risk of bronchospasm. The effect of ADASUVE on pulmonary function was evaluated in 3 randomized, double-blind, placebo-controlled clinical pulmonary safety trials in healthy volunteers, patients with asthma, and patients with COPD. Pulmonary function was assessed by serial FEV1 tests, and respiratory signs and symptoms were assessed. In the asthma and COPD trials, patients with respiratory symptoms or FEV1 decrease of ≥ 20% were administered rescue treatment with albuterol (metered dose inhaler or nebulizer) as required. These patients were not eligible for a second dose; however, they had continued FEV1 monitoring in the trial.
Healthy Volunteers: In the healthy volunteer crossover trial, 30 subjects received 2 doses of either ADASUVE or placebo 8 hours apart, and 2 doses of the alternate treatment at least 4 days later. The results for maximum decrease in FEV1 are presented in Table 2. No subjects in this trial developed airway related adverse reactions (cough, wheezing, chest tightness, or dyspnea).
Asthma Patients: In the asthma trial, 52 patients with mild-moderate persistent asthma (with FEV1 ≥ 60% of predicted) were randomized to treatment with 2 doses of ADASUVE 10 mg or placebo. The second dose was to be administered 10 hours after the first dose. Approximately 67% of these patients had a baseline FEV1 ≥ 80% of predicted. The remaining patients had an FEV1 60-80% of predicted. Nine patients (17%) were former smokers. As shown in Table 2 and Figure 7, there was a marked decrease in FEV1 immediately following the first dose (maximum mean decreases in FEV1 and % predicted FEV1 were 303 mL and 9.1%, respectively). Furthermore, the effect on FEV1 was greater following the second dose (maximum mean decreases in FEV1 and % predicted FEV1 were 537 mL and 14.7 %, respectively). Respiratory-related adverse reactions (bronchospasm, chest discomfort, cough, dyspnea, throat tightness, and wheezing) occurred in 54% of ADASUVE-treated patients and 12% of placebo-treated patients. There were no serious adverse events. Nine of 26 (35%) patients in the ADASUVE group, compared to one of 26 (4%) in the placebo group, did not receive a second dose of study medication, because they had a ≥ 20% decrease in FEV1 or they developed respiratory symptoms after the first dose. Rescue medication (albuterol via metered dose inhaler or nebulizer) was administered to 54% of patients in the ADASUVE group [7 patients (27%) after the first dose and 7 of the remaining 17 patients (41%) after the second dose] and 12% in the placebo group (1 patient after the first dose and 2 patients after the second dose).
COPD Patients: In the COPD trial, 53 patients with mild to severe COPD (with FEV1 ≥ 40% of predicted) were randomized to treatment with 2 doses of ADASUVE 10 mg or placebo. The second dose was to be administered 10 hours after the first dose. Approximately 57% of these patients had moderate COPD [Global Initiative for Chronic Obstructive Lung Disease (GOLD) Stage II]; 32% had severe disease (GOLD Stage III); and 11% had mild disease (GOLD Stage I). As illustrated in Table 2 there was a decrease in FEV1 soon after the first dose (maximum mean decreases in FEV1 and % predicted FEV1 were 96 mL and 3.5%, respectively), and the effect on FEV1 was greater following the second dose (maximum mean decreases in FEV1 and % predicted FEV1 were 125 mL and 4.5%, respectively). Respiratory adverse reactions occurred more frequently in the ADASUVE group (19%) than in the placebo group (11%). There were no serious adverse events. Seven of 25 (28%) patients in the ADASUVE group and 1of 27 (4%) in the placebo group did not receive a second dose of study medication because of a ≥ 20% decrease in FEV1 or the development of respiratory symptoms after the first dose. Rescue medication (albuterol via MDI or nebulizer) was administered to 23% of patients in the ADASUVE group: 8% of patients after the first dose and 21% of patients after the second dose, and to 15% of patients in the placebo group.
Table 2: Maximum Decrease in FEV1 from Baseline in the Healthy Volunteer, Asthma, and COPD Trials Healthy Volunteer Asthma COPD Maximum % FEV ↓ Placebo
n (%)ADASUVE 10 mg
n (%)Placebo
n (%)ADASUVE 10 mg
n (%)Placebo
n (%)ADASUVE 10 mg
n (%)After any Dose N=26 N=26 N=26 N=26 N=27 N=25 ≥10 7 (27) 7 (27) 3 (12) 22 (85) 18 (67) 20 (80) ≥15 1 (4) 5 (19) 1 (4) 16 (62) 9 (33) 14 (56) ≥20 0 1 (4) 1 (4) 11 (42) 3 (11) 10 (40) After Dose 1 N=26 N=26 N=26 N=26 N=27 N=25 ≥10 4 (15) 5 (19) 2 (8) 16 (62) 8 (30) 16 (64) ≥15 1 (4) 2 (8) 1 (4) 8 (31) 4 (15) 10 (40) ≥20 0 0 1 (4) 6 (23) 2 (7) 9 (36) After Dose 2 N=26 N=25 N=25 N=17 N=26 N=19 ≥10 5 (19) 6 (24) 3 (12) 12 (71) 15 (58) 12 (63) ≥15 0 5 (20) 1 (4) 9 (53) 6 (23) 10 (53) ≥20 0 1 (4) 1 (4) 5 (30) 1 (4) 5 (26) FEV1 categories are cumulative; i.e. a subject with a maximum decrease of 21% is included in all 3 categories. Patients with a ≥ 20% decrease in FEV1 did not receive a second dose of study drug.
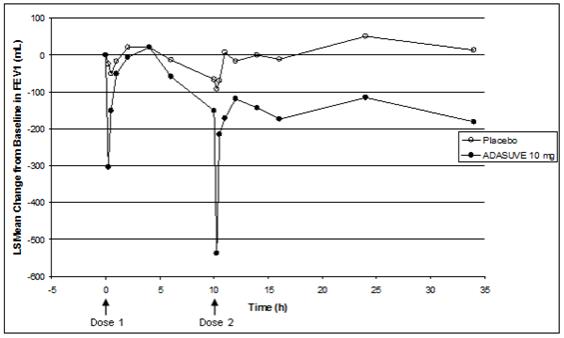
Patients with a ≥ 20% decrease in FEV1 did not receive a second dose of study drug and are not included in the curves beyond hour 10.
Extrapyramidal Symptoms (EPS): Extrapyramidal reactions have occurred during the administration of oral loxapine. In most patients, these reactions involved parkinsonian symptoms such as tremor, rigidity, and masked facies. Akathisia (motor restlessness) has also occurred.
In the 3 short-term (24-hour), placebo-controlled trials of ADASUVE in 259 patients with agitation associated with schizophrenia or bipolar disorder, extrapyramidal reactions occurred. One patient (0.4%) treated with ADASUVE developed neck dystonia and oculogyration. The incidence of akathisia was 0% and 0.4% in the placebo and ADASUVE groups, respectively.
Dystonia (Antipsychotic Class Effect): Symptoms of dystonia, prolonged abnormal contractions of muscle groups, may occur in susceptible individuals during treatment with ADASUVE. Dystonic symptoms include spasm of the neck muscles, sometimes progressing to tightness of the throat, difficulty swallowing or breathing, and/or protrusion of the tongue.
Acute dystonia tends to be dose-related, but can occur at low doses, and occurs more frequently with first generation antipsychotic drugs such as ADASUVE. The risk is greater in males and younger age groups.
Cardiovascular Reactions: Tachycardia, hypotension, hypertension, orthostatic hypotension, lightheadedness, and syncope have been reported with oral administration of loxapine.
- Hypersensitivity (serious skin reactions)
[see Contraindications (4)]
-
7
DRUG INTERACTIONS
7.1 CNS Depressants
ADASUVE is a central nervous system (CNS) depressant. The concurrent use of ADASUVE with other CNS depressants (e.g., alcohol, opioid analgesics, benzodiazepines, tricyclic antidepressants, general anesthetics, phenothiazines, sedative/hypnotics, muscle relaxants, and/or illicit CNS depressants) can increase the risk of respiratory depression, hypotension, profound sedation, and syncope. Therefore, consider reducing the dose of CNS depressants if used concomitantly with ADASUVE.
-
8
USE IN SPECIFIC POPULATIONS
In general, no dose adjustment for ADASUVE is required on the basis of a patient’s age, gender, race, smoking status, hepatic function, or renal function.
8.1 Pregnancy
Pregnancy Category C
Risk Summary
There are no adequate and well-controlled studies of ADASUVE use in pregnant women. Neonates exposed to antipsychotic drugs during the third trimester of pregnancy are at risk for extrapyramidal and/or withdrawal symptoms following delivery. Loxapine, the active ingredient in ADASUVE, has demonstrated increased embryofetal toxicity and death in rat fetuses and offspring exposed to doses approximately 0.5-fold the maximum recommended human dose (MRHD) on a mg/m 2 basis. ADASUVE should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Human Data
Neonates exposed to antipsychotic drugs during the third trimester of pregnancy are at risk for extrapyramidal and/or withdrawal symptoms following delivery. There have been reports of agitation, hypertonia, hypotonia, tremor, somnolence, respiratory distress, and feeding disorders in these neonates. These complications have varied in severity; in some cases symptoms have been self-limited, but in other cases neonates have required intensive care unit support and prolonged hospitalization.
Animal Data
In rats, embryofetal toxicity (increased fetal resorptions, reduced weights, and hydronephrosis with hydroureter) was observed following oral administration of loxapine during the period of organogenesis at a dose of 1 mg/kg/day. This dose is equivalent to the MRHD of 10 mg/day on a mg/m 2 basis. In addition, fetal toxicity (increased prenatal death, decreased postnatal survival, reduced fetal weights, delayed ossification, and/or distended renal pelvis with reduced or absent papillae) was observed following oral administration of loxapine from mid-pregnancy through weaning at doses of 0.6 mg/kg and higher. This dose is approximately half the MRHD of 10 mg/day on a mg/m 2 basis.
No teratogenicity was observed following oral administration of loxapine during the period of organogenesis in the rat, rabbit, or dog at doses up to 12, 60, and 10 mg/kg, respectively. These doses are approximately 12-, 120-, and 32-fold the MRHD of 10 mg/day on a mg/m 2 basis, respectively.
8.3 Nursing Mothers
It is not known whether ADASUVE is present in human milk. Loxapine and its metabolites are present in the milk of lactating dogs. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from ADASUVE, a decision should be made whether to discontinue nursing or discontinue ADASUVE, taking into account the importance of the drug to the mother.
8.4 Pediatric Use
The safety and effectiveness of ADASUVE in pediatric patients have not been established.
8.5 Geriatric Use
Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death [see Boxed Warning and Warnings and Precautions (5.3)]. ADASUVE is not approved for the treatment of dementia-related psychosis. Placebo-controlled studies of ADASUVE in patients with agitation associated with schizophrenia or bipolar disorder did not include patients over 65 years of age.
-
10
OVERDOSAGE
Signs and Symptoms of Overdosage
As would be expected from the pharmacologic actions of loxapine, the clinical findings may include CNS depression, unconsciousness, profound hypotension, respiratory depression, extrapyramidal symptoms, and seizure.
Management of Overdosage
For the most up to date information on the management of ADASUVE overdosage, contact a certified poison control center (1-800-222-1222 or www.poison.org). Provide supportive care including close medical supervision and monitoring. Treatment should consist of general measures employed in the management of overdosage with any drug. Consider the possibility of multiple drug overdosage. Ensure an adequate airway, oxygenation, and ventilation. Monitor cardiac rhythm and vital signs. Use supportive and symptomatic measures.
-
11
DESCRIPTION
ADASUVE, a typical antipsychotic, is an inhalation powder of loxapine supplied in a single-use, disposable inhaler containing 10 mg of loxapine base. ADASUVE is a drug-device combination product.
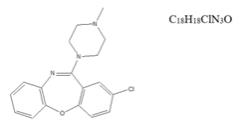
Active Ingredient: Loxapine (base). Loxapine, a dibenzoxazepine compound, represents a subclass of tricyclic antipsychotic agents, chemically distinct from the thioxanthenes, butyrophenones, and phenothiazines. Chemically, it is 2-Chloro-11-(4-methyl-1-piperazinyl) dibenz [b,f] [1,4] oxazepine.
ADASUVE is a single-use, drug-device combination product that provides rapid systemic delivery by inhalation of a thermally-generated aerosol of loxapine. Oral inhalation through the product initiates the controlled rapid heating of a thin film of excipient-free loxapine to form a thermally-generated drug vapor. The vapor condenses into aerosol particles that are dispersed into the airstream created by the patient inhaling through the mouthpiece.
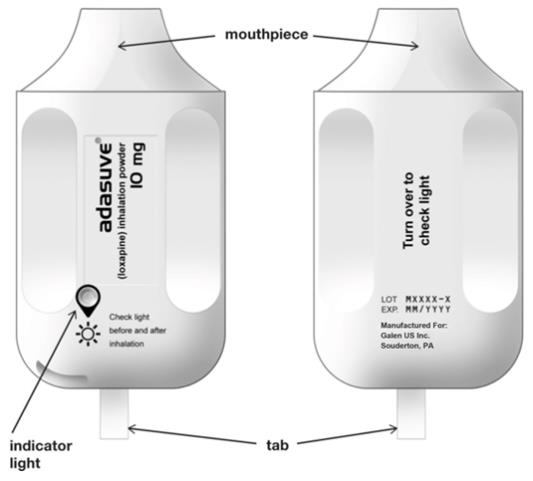
Each product is packaged inside a sealed foil pouch. The product is a white to off-white plastic unit, with a mouthpiece on one end and a pull-tab protruding from the other end.
Removal of a pull-tab from the product renders it ready for use, as indicated by illumination of a green light. After inhalation through the mouthpiece, successful dosing is signaled by the green light turning off.
Under standardized in vitro test conditions, ADASUVE, 10 mg delivers 9.1 mg of loxapine out of the mouthpiece.
-
12
CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The mechanism of action of loxapine in the treatment of agitation associated with schizophrenia is unknown. However, its efficacy could be mediated through a combination of antagonism of central dopamine D 2 and serotonin 5-HT 2A receptors. The mechanism of action of loxapine in the treatment of agitation associated with bipolar I disorder is unknown.
12.2 Pharmacodynamics
Loxapine acts as an antagonist at central serotonin and dopamine receptors, with high affinity for serotonin 5-HT 2A and dopamine D 1, D 2, D 3, and D 4 receptors (K i values of 2 nM, 18 nM, 10 nM, 21 nM, 9 nM, respectively). Some of the adverse effects of loxapine may be related to the antagonism of histamine H 1 (somnolence), muscarinic M1 (anticholinergic), and adrenergic α 2 (orthostatic hypotension) receptors (K i values of 15 nM, 117 nM and 250 nM, respectively).
Thorough QTc Study
ADASUVE did not prolong the QTc interval. The effect of ADASUVE on QTc prolongation was evaluated in a randomized, double-blinded, positive- (moxifloxacin 400 mg) and placebo-controlled parallel study in healthy subjects. A total of 48 healthy subjects were administered ADASUVE 10 mg. In this study with a demonstrated ability to detect small effects, the upper bound of the 90% confidence interval (CI) for the largest placebo-adjusted, baseline-corrected QTc based on individual correction method was below 10 milliseconds, the threshold for regulatory concern.
12.3 Pharmacokinetics
Absorption: The single-dose pharmacokinetic parameters of loxapine following administration of single doses of ADASUVE 10 mg in healthy adult subjects are presented in Table 3 and Figure 8.
Administration of ADASUVE resulted in rapid absorption of loxapine, with a median time of maximum plasma concentration (T max) of 2 minutes. Loxapine exposure in the first 2 hours after administration (AUC 0-2h) was 66.7 ngh/mL for the 10 mg dose. As a consequence of the very rapid absorption of loxapine after oral inhalation, there is substantial variability in the early plasma concentrations of loxapine. The mean plasma loxapine concentrations following administration of ADASUVE were linear over the clinical dose range. AUC 0-2h, AUC inf, and C max increased in a dose-dependent manner.
Table 3. Pharmacokinetics in Healthy Adult Subjects Administered a Single Dose of ADASUVE 10 mg Parameter Healthy Subjects ADASUVE 10 mg (N=114) AUC 0-2h (ngh/mL), mean ± SD 66.7 ± 18.2 AUC inf (ngh/mL), mean ± SD 188 ± 47 C max (ng/mL), mean ± SD 257 ± 219 T max (minutes), median (25%, 75%) 1.13 (1, 2) Half-life(h), mean ± SD 7.61 ± 1.87 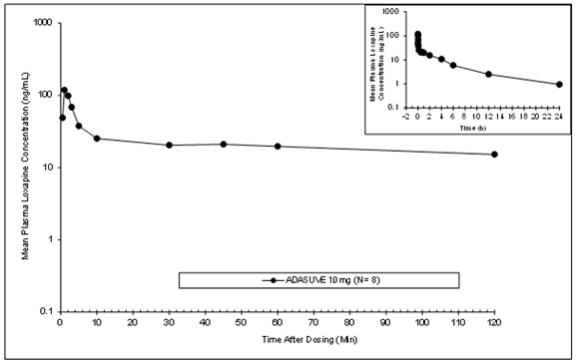
Distribution: Loxapine is removed rapidly from the plasma and distributed in tissues. Animal studies following oral administration suggest an initial preferential distribution in the lungs, brain, spleen, heart, and kidney. Loxapine is 96.6% bound to human plasma proteins.
Metabolism: Loxapine is metabolized extensively in the liver following oral administration, with multiple metabolites formed. The main metabolic pathways include: 1) hydroxylation to form 8-OH-loxapine by CYP1A2 and 7-OH-loxapine by CYP3A4 and CYP2D6, 2) N-oxidation to form loxapine N-oxide by flavanoid monoamine oxidases (FMOs), and 3) de-methylation to form amoxapine. Because there are multiple metabolic pathways, the risk of metabolic interactions caused by an effect on an individual isoform is minimal. For ADASUVE, the order of metabolites observed in humans (based on systemic exposure) was 8-OH-loxapine >> loxapine N-oxide, 7-OH-loxapine > amoxapine. Plasma levels of 8-OH-loxapine are similar to those of the parent compound.
Excretion: Excretion occurs mainly in the first 24 hours. Metabolites are excreted in the urine in the form of conjugates and in the feces unconjugated. The terminal elimination half-life (T ½) ranged from 6 to 8 hours.
Transporter Interaction: In vitro studies indicated that loxapine was not a substrate for p-glycoprotein (P-gp): however, loxapine inhibited P-gp.
Special Populations:
Pharmacokinetics in Smokers: Loxapine exposures in nonsmokers and smokers are similar, with geometric mean ratios of 92%, 85%, and 99% for AUC 0-2h, AUC inf, and C max respectively. No dosage adjustment is recommended based on smoking status.
Demographic Effects: There were no clinically significant differences in loxapine pharmacokinetics following administration of ADASUVE in subgroups based on age, weight, body mass index, gender, or race.
-
13
NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis: No adequate studies have been conducted.
Mutagenesis: Loxapine did not cause mutation or chromosomal aberration when tested in vitro and in vivo. Loxapine was negative in the Ames gene mutation assay, the human peripheral blood lymphocyte chromosomal aberration assay, and in the in vivo mouse bone marrow micronucleus assay up to 40 mg/kg (20-fold the MRHD on mg/m 2 basis).
Loxapine metabolite 8-OH-loxapine was not mutagenic in the in vitro Ames reverse mutation assay and was not clastogenic in the in vitro human peripheral blood lymphocyte chromosomal aberration assay.
Impairment of Fertility: Loxapine had no effects on fertility or early embryonic development in male rats or in male and female rabbits following oral administration. Mating was decreased in female rats because these animals were in persistent diestrus, an expected pharmacologic effect for this class of compounds. This occurred at doses approximately 0.2- and 1-fold the MRHD of 10 mg/day on a mg/m 2 basis.
13.2 Animal Toxicology and/or Pharmacology
In the rat, minimal and reversible squamous metaplasia of the larynx was observed after daily inhalation exposure of loxapine for 14 days at 1.7 to 13 mg/kg/day (approximately 2- to 13-fold the MRHD of 10 mg/day on a mg/m 2 basis, respectively). This finding was considered a nonspecific particle impaction effect. Mammary hyperplasia in males and females and ovarian follicular cysts and mucification of vaginal epithelium in female rats were observed at all doses, with partial or complete recovery at the end of 14 days of treatment. In the dog, no effects on the respiratory tract or reproductive tissues were observed after inhalation exposure to loxapine for 28 days at doses up to 1.8 mg/kg/day (approximately 6-fold the MRHD of 10 mg/day on a mg/m 2 basis).
-
14
CLINICAL STUDIES
The efficacy of ADASUVE 10 mg in the acute treatment of agitation associated with schizophrenia or bipolar I disorder was established in two short-term (24-hour), randomized, double-blind, placebo-controlled, fixed-dose trials. Study 1 included 344 patients who met DSM-IV criteria for schizophrenia. Study 2 included 314 patients who met DSM-IV criteria for bipolar I disorder, manic or mixed episodes with or without psychotic features.
Patients were judged by the clinical investigators to be clinically agitated, with a level of agitation that met or exceeded a specific severity threshold as measured by the Positive and Negative Syndrome Scale-Excited Component (PEC). The PEC is an investigator-rated instrument consisting of 5 items: poor impulse control, tension, hostility, uncooperativeness, and excitement. Each item is scored on a scale from 1 to 7 (1 = absent, 4 = moderate, 7 = extreme). Thus, the total PEC score can range from 5 to 35. For enrollment in the studies, patients had to have a PEC score of ≥ 14, with at least one individual item score > 4.
Patients whose agitation was related to acute alcohol or drug intoxication were excluded. Patients with clinically significant acute or chronic pulmonary disease (e.g., asthma, COPD, chronic bronchitis, and emphysema) were excluded from the trials [See Contraindications (4)].
The primary efficacy endpoint in both trials was the mean change from baseline in the PEC score, assessed 2 hours following dosing. The key secondary endpoint was the mean Clinical Global Impression Improvement (CGI-I) Scale score at two hours. The CGI-I is an investigator-rated global assessment of symptom improvement, scored on a scale of 1 to 7: 1 = very much improved; 4 = no change from baseline; 7 = very much worse.
In both studies, mean baseline PEC scores were similar in all treatment groups, averaging 17.3 to 17.7 (Table 4), with individual patient scores ranging from 14 to 31, indicating predominantly moderate levels of agitation. The mean baseline Clinical Global Impression Severity Scale (CGI-S) score in both studies was 4 (moderately ill). In Study 2, 69% of patients had a current manic episode, and 31% had a mixed/manic episode.
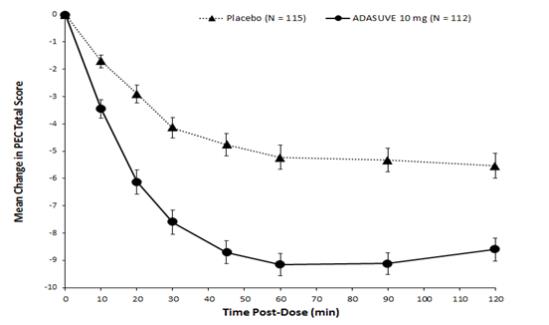
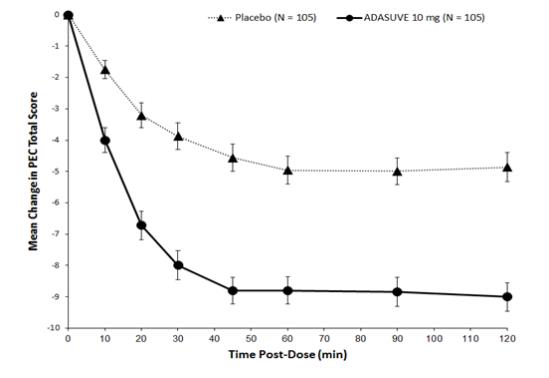
In Studies 1 and 2, treatment with ADASUVE was statistically significantly superior to placebo on the mean change in PEC score at 2 hours (Table 4). In both studies, the effect of ADASUVE was apparent at 10 minutes following dosing (Figures 9 and 10).
Table 4. Change from Baseline in the PEC Score at 2 Hours Post-Dose in the Schizophrenia (Study 1) and Bipolar I Disorder (Study 2) Trials Placebo
ADASUVE
Study 1 (Schizophrenia) N 115 112 PEC score Mean baseline 17.4 17.6 Change at 2 hours a -5.8 -8.7 Difference from placebo (95% CI) b -- -2.9 (-4.2, -1.6) p-value -- < 0.0001 Study 2 (Bipolar Disorder ) N 105 105 PEC score Mean baseline 17.7 17.3 Change at 2 hours a -4.7 -9.2 Difference from placebo (95% CI) b -- -4.5 (-5.8, -3.1) p-value -- < 0.0001 a Least squares mean for the difference defined as the change from baseline
b Least squares mean for the difference defined as the change from baseline at hour 2 in the drug group minus that in the placebo group.
Examination of population subsets (age, race, and gender) on the primary endpoint did not reveal any differential responsiveness on the basis of these subgroupings.
Figures 9 and 10 show the decreases in PEC score at each time point assessed in the trials. In both trials, the decrease in agitation with ADASUVE was apparent at each time point tested (10, 20, 30, 45, 60, 90, and 120 minutes post-dose).
The results of the secondary endpoint, CGI-I scores, are shown in Table 5.
Table 5. CGI-I Score at 2 Hours Post-Dose in the Schizophrenia and Bipolar I Disorder Trials Placebo
ADASUVE
Study 1 (Schizophrenia) N 115 112 CGI-I score at 2 hours a 2.8 2.1 Difference from placebo (95% CI) - -0.8 (-1.1, -0.4) p-value - <0.0001 Study 2 (Bipolar Disorder) N 105 105 CGI-I score at 2 hours a 3.0 1.9 Difference from placebo (95% CI) a - -1.1 (-1. 4, -0.8) p-value - <0.0001 a Least squares mean
-
16
HOW SUPPLIED/STORAGE AND HANDLING
16.1 How Supplied
ADASUVE ® (loxapine) inhalation powder is supplied as:
ADASUVE 10 mg (NDC: 10885-003-01) is a single-use, disposable inhaler containing 10 mg of loxapine, provided in a sealed foil pouch. ADASUVE, 10 mg is supplied in a carton of 5 units per carton (NDC: 10885-003-05).
16.2 Restricted Access
ADASUVE is only available through a restricted program called the ADASUVE REMS Program [see Warnings and Precautions (5.2)].
16.3 Storage and Handling
Store ADASUVE at room temperature, 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].
Keep out of reach of children.
Keep ADASUVE in pouch until time of use.
ADASUVE contains a lithium battery. Dispose of ADASUVE in accordance with all federal, state and local laws.
-
17
PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling ( Medication Guide)
Bronchospasm
Advise patients and caregivers that there is a risk of bronchospasm. Advise patients to inform their healthcare professional if they develop any breathing problems such as wheezing, shortness of breath, chest tightness, or cough following treatment with ADASUVE [see Boxed Warning and Warnings and Precautions (5.1)]
Interference with Cognitive and Motor Performance
Caution patients and caregivers about performing activities requiring mental alertness, such as operating hazardous machinery or operating a motor vehicle, until they are reasonably certain that ADASUVE has not affected them adversely [see Warnings and Precautions (5.8)].
Caution patients and caregivers about the potential for sedation, especially when used concurrently with other CNS depressants (e.g., alcohol, opioid analgesics, benzodiazepines, tricyclic antidepressants, general anesthetics, phenothiazines, sedative/hypnotics, muscle relaxants, and/or illicit CNS depressants).
Neuroleptic Malignant Syndrome
Patients and caregivers should be counseled that a potentially fatal symptom complex sometimes referred to as NMS has been reported in association with administration of antipsychotic drugs. Signs and symptoms of NMS include hyperpyrexia, muscle rigidity, altered mental status, and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis, and cardiac dysrhythmia) [see Warnings and Precautions (5.4)].
Hypotension and Syncope
Advise patients and caregivers of the risk of hypotension or orthostatic hypotension (symptoms include feeling dizzy or lightheaded upon standing) [see Warnings and Precautions (5.5)].
Anticholinergic Reactions
Counsel patients and caregivers about the potential risks of anticholinergic reactions, such as exacerbation of glaucoma and urinary retention [see Warnings and Precautions (5.10)].
Pregnancy
Counsel patients and caregivers regarding the potential risk to the fetus or neonate [see Use in Specific Populations (8.1)].
Nursing Mothers
Counsel patients and caregivers regarding the potential risk to the infant [see Use in Specific Populations (8.3)].
Manufactured for: Galen US INC., 25 Fretz Road, Souderton, PA 18964
-
MEDICATION GUIDE
MEDICATION GUIDE
ADASUVE® (AD-uh-soov)
(loxapine)
Inhalation Powder
Read this Medication Guide before you start taking ADASUVE and each time it is given to you. There may be new information. This Medication Guide does not take the place of talking to your healthcare provider about your medical condition or your treatment. You should share this information with your family members and caregivers.
What is the most important information I should know about ADASUVE?
ADASUVE is available only through the ADASUVE Risk Evaluation and Mitigation Strategy (REMS) Program. The healthcare facility must be enrolled in the ADASUVE REMS Program before you can be given ADASUVE.
ADASUVE may cause serious side effects, including:
-
Narrowing of the airways (bronchospasm) that can cause you to have problems breathing or to stop breathing. People who have asthma or other airway or lung problems, such as chronic obstructive pulmonary disease (COPD), have a higher risk of bronchospasm when taking ADASUVE. Symptoms of bronchospasm may include:
○ wheezing
○ coughing
○ chest tightness
○ shortness of breath
Tell your healthcare provider right away if you have any of these symptoms of bronchospasm after taking ADASUVE.
Your healthcare provider should check you for breathing problems before and after you take ADASUVE.
- Increased risk of death in elderly patients with dementia-related psychosis. Medicines like ADASUVE can raise the risk of death in elderly people who have lost touch with reality (psychosis) due to confusion and memory loss (dementia). ADASUVE is not approved for the treatment of patients with dementia-related psychosis.
What is ADASUVE?
ADASUVE is a prescription medicine that is inhaled through your mouth and is used to treat acute agitation in adults with schizophrenia or bipolar I disorder.
It is not known if ADASUVE is safe and effective in children.
Who should not take ADASUVE?
Do not take ADASUVE if you:
- have or have had asthma, chronic obstructive pulmonary disease (COPD), or other airway or lung problems that can cause bronchospasm
- are having problems with wheezing, coughing, chest tightness, or shortness of breath
- are taking medicines to treat asthma or COPD
- have taken ADASUVE before and had bronchospasm
- are allergic to loxapine or amoxapine
What should I tell my healthcare provider before taking ADASUVE?
Before you take ADASUVE, tell your healthcare provider if you:
- have high or low blood pressure
- have or have had heart problems or stroke
- have or have had seizures (convulsions)
- drink alcohol or use street drugs
- have any other medical conditions
- are pregnant or plan to become pregnant. It is not known if ADASUVE will harm your unborn baby.
- are breastfeeding or plan to breastfeed. It is not known if ADASUVE passes into your breast milk. You and your healthcare provider should decide if you will take ADASUVE if you are breastfeeding.
Tell your doctor about all medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements.
- ADASUVE and other medicines may affect each other causing side effects. ADASUVE may affect the way other medicines work, and other medicines may affect the way ADASUVE works.
Ask your healthcare provider or pharmacist for a list of these medicines if you are not sure.
Know the medicines you take. Keep a list of them to show to your healthcare provider and pharmacist when you get a new medicine.
How should I take ADASUVE?
- Your healthcare provider will show you how to take ADASUVE right before you take it.
- Take ADASUVE exactly as your healthcare provider shows you to take it.
- ADASUVE is for oral inhalation only.
What should I avoid while taking ADASUVE?
- Do not drive, operate heavy machinery, or do other dangerous activities until you know how ADASUVE affects you.
- Do not drink alcohol while taking ADASUVE. It can increase your chances of getting serious side effects.
What are the possible side effects of ADASUVE?
ADASUVE can cause serious side effects, including:
- See “What is the most important information I should know about ADASUVE?”
-
neuroleptic malignant syndrome (NMS). NMS is a rare but serious condition that may cause death. Symptoms of NMS may include:
○ high fever
○ stiff muscles
○ confusion
○ sweating
○ changes in pulse, heart rate, and blood pressure
Tell your healthcare provider right away if you have any of these symptoms of NMS after taking ADASUVE.
-
low blood pressure (hypotension), lightheadedness, or fainting
-
seizures (convulsions)
-
severe sleepiness and difficulty with potentially dangerous activities such as driving
-
worsening of glaucoma
- difficulty urinating
The most common side effects of ADASUVE include:
- bad, bitter, or metallic taste in your mouth (dysgeusia)
- sleepiness (especially when used with other drugs that cause sleepiness)
- sore throat
Tell your healthcare provider if you have any side effect that bothers you or that does not go away.
These are not all the possible side effects of ADASUVE. For more information ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of ADASUVE.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide.
This Medication Guide summarizes the most important information about ADASUVE. If you would like more information, talk to your healthcare provider. You can ask your pharmacist or healthcare provider for information about ADASUVE that is written for health professionals.
For more information, go to www.ADASUVE.com or call 1-800-284-0062.
What are the ingredients in ADASUVE?
Active Ingredient: loxapine
Inactive Ingredients: noneThis Medication Guide has been approved by the U.S. Food and Drug Administration
Manufactured for: Galen US Inc., 25 Fretz Road, Souderton, PA 18964
Rev. 08/2017
-
Narrowing of the airways (bronchospasm) that can cause you to have problems breathing or to stop breathing. People who have asthma or other airway or lung problems, such as chronic obstructive pulmonary disease (COPD), have a higher risk of bronchospasm when taking ADASUVE. Symptoms of bronchospasm may include:
-
PRINCIPAL DISPLAY PANEL
INSTRUCTIONS FOR USE
Becoming Familiar with ADASUVE Read all instructions before use.
ADASUVE is a single-dose, single-use inhaler for oral inhalation only. Discard inhaler after use.
Important Information: Refer to the Boxed Warning and Contraindications regarding Bronchospasm in Full Prescribing Information.
ADASUVE is contraindicated in:
- Patients with asthma, chronic obstructive pulmonary disease (COPD), or other lung diseases associated with bronchospasm, because they are at increased risk of bronchospasm.
- Patients with acute respiratory signs/symptoms (e.g., wheezing) or who are currently taking medications to treat airways disease, such as asthma or COPD.
- Patients with a history of bronchospasm following ADASUVE treatment .
Prior to administering ADASUVE, screen patients regarding a history or symptoms of asthma, COPD and other lung diseases, and examine (including chest auscultation) patients for respiratory signs.
The pictures below show the features of ADASUVE:
The indicator light conveys important information about dose delivery: - The indicator light is off when ADASUVE is removed from the pouch.
- The indicator light turns on (green) when the tab is pulled out. The inhaler is then ready for use.
- The indicator light turns off after the patient inhales. This indicates the dose has been delivered.
- If the indicator light does NOT turn off, the dose has NOT been delivered. See Instructions for Use.
ADASUVE Instructions for UseRead the following 6 steps before administering ADASUVE to a patient. Do not open the pouch until ready to use.
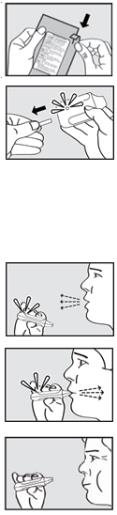
1. Open pouch
Tear open the foil pouch and remove the inhaler from the package.
2. Pull tab
Firmly pull the plastic tab from the rear of the inhaler.
IMPORTANT: Check that the green light turns on. This indicates that the inhaler is ready for use.
Use inhaler within 15 minutes after removing the tab or else the green light will turn off indicating the inhaler is not usable.
3. Explain Procedures to Patient
Explain the administration procedures to the patient prior to use and let patients know it is important to follow the instructions. Advise patients that the inhaler may produce a flash of light or a clicking sound, or become warm during use. These are normal.
Instruct the Patient to:
4. Exhale
Hold the inhaler away from the mouth and breathe out fully to empty lungs.
5. Inhale
Inhale through the mouthpiece with a steady deep breath.
IMPORTANT: Check that the green light turns off after the patient inhales. The light will turn off indicating that the dose has been delivered.6. Hold breath
Remove the mouthpiece from the mouth and hold breath for as long as possible, up to 10 seconds.Important: If the green light stays on after the patient inhales, the dose has NOT been delivered. Instruct the patient to repeat steps 4, 5, and 6 up to 2 additional times.
If the green light still does not turn off, discard the inhaler and use a new one.
Manufactured by: Alexza Pharmaceuticals, Inc., Mountain View, CA 94043
Manufactured for: Galen US Inc., 25 Fretz Road, Souderton, PA 18964
Iss. 08/2017
PRINCIPAL DISPLAY PANEL
NDC: 10885-003-01
adasuve
(loxapine) inhalation powder
1 single dose unit per pouch
10mg
Rx Only

PRINCIPAL DISPLAY PANEL
NDC: 10885-003-01
adasuve
(loxapine) inhalation powder
1 single dose unit per pouch
10mg
Rx Only
PRINCIPAL DISPLAY PANEL
NDC: 10885-003-05
adasuve
(loxapine) inhalation powder
5 single dose units per carton
10 mg
Rx Only - Patients with asthma, chronic obstructive pulmonary disease (COPD), or other lung diseases associated with bronchospasm, because they are at increased risk of bronchospasm.
-
PRINCIPAL DISPLAY PANEL

PRINCIPAL DISPLAY PANEL
NDC: 10885-003-05
adasuve
(loxapine) inhalation powder
5 single dose units per carton
10 mg
Rx Only
PRINCIPAL DISPLAY PANEL
NDC: 10885-003-05
adasuve
(loxapine) inhalation powder
5 single dose units per carton
10 mg
Rx Only -
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL
NDC: 10885-003-05
adasuve
(loxapine) inhalation powder
5 single dose units per carton
10 mg
Rx Only -
PRINCIPAL DISPLAY PANEL

PRINCIPAL DISPLAY PANEL
NDC: 10885-003-05
adasuve
(loxapine) inhalation powder
5 single dose units per carton
10 mg
Rx Only
-
INGREDIENTS AND APPEARANCE
ADASUVE
loxapine aerosol, powderProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 10885-003 Route of Administration RESPIRATORY (INHALATION) Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LOXAPINE (UNII: LER583670J) (LOXAPINE - UNII:LER583670J) LOXAPINE 10 mg Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 10885-003-05 5 in 1 CARTON 09/26/2017 09/30/2021 1 NDC: 10885-003-01 1 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA022549 09/26/2017 09/30/2021 Labeler - Galen US Incorporated (029491194)
Trademark Results [Adasuve]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 ADASUVE 77752342 4276060 Live/Registered |
Alexza Pharmaceuticals, Inc. 2009-06-04 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.