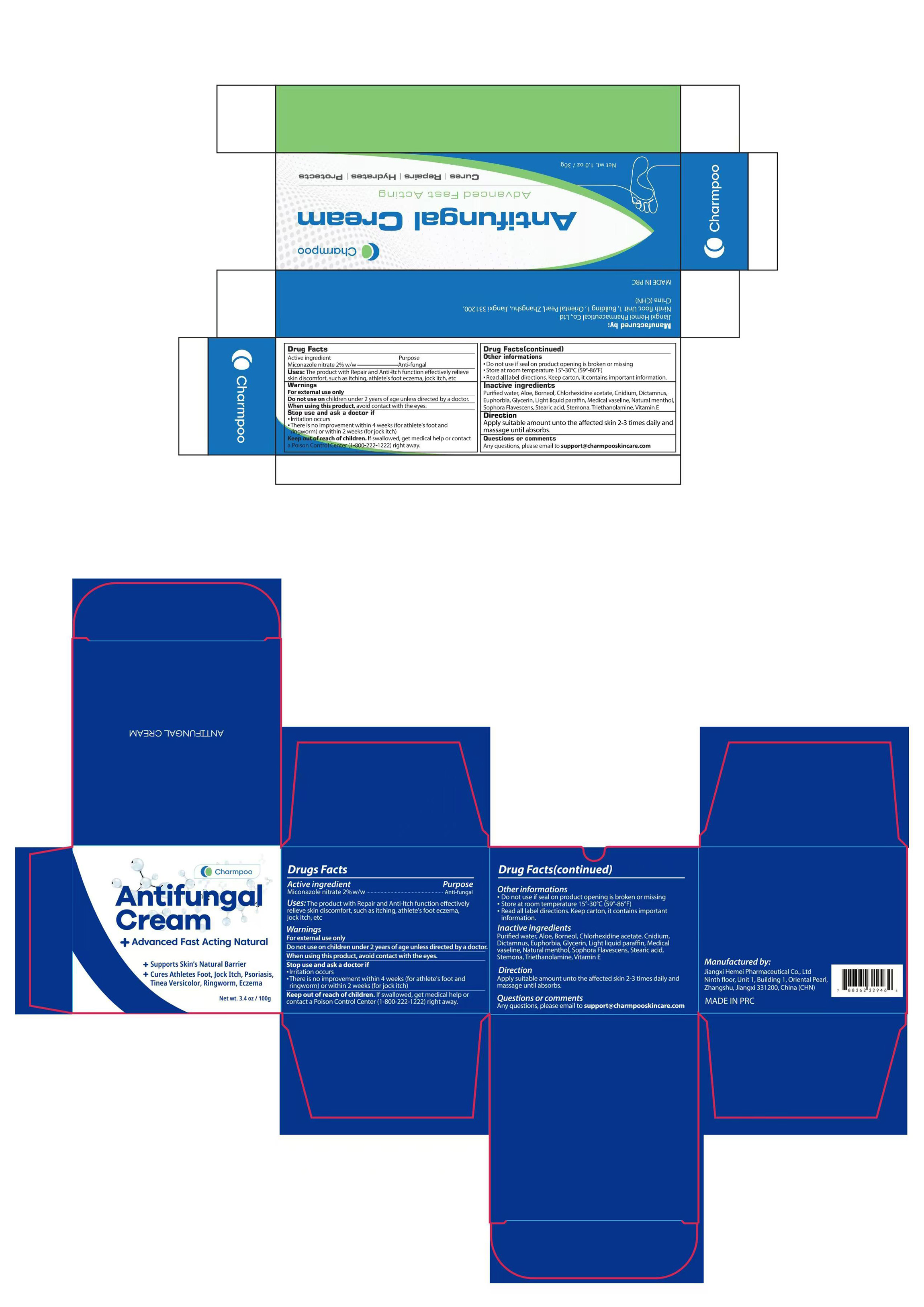
Charmpoo Antifungal Cream by Jiangxi Hemei Pharmaceutical Co., Ltd 84010-046 complete
Charmpoo Antifungal Cream by
Drug Labeling and Warnings
Charmpoo Antifungal Cream by is a Otc medication manufactured, distributed, or labeled by Jiangxi Hemei Pharmaceutical Co., Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
CHARMPOO ANTIFUNGAL CREAM- antifungal cream cream
Jiangxi Hemei Pharmaceutical Co., Ltd
----------
84010-046 complete
Use
The product with Repair and Anti-ltch function effectively relieve skin discomfort, such as itching, athlete's foot eczema, jock itch, etc
Stop Use
·Irritation occurs
·There is no improvement within 4 weeks (for athlete's foot and fingworm) or within 2 weeks (for jock itch)
Ask Doctor
·Irritation occurs
·There is no improvement within 4 weeks (for athlete's foot and fingworm) or within 2 weeks (for jock itch)
Keep Oot Of Reach Of Children
lf swallowed, get medical help or contact a Poison controlcenter(1-800-222-1222)right away.
Other information
·Do not use if seal on product opening is broken or missing
·Store at room temperature 15°-30°C(59°-86°F)
·Read all label directions. Keep carton, it contains important information.
| CHARMPOO ANTIFUNGAL CREAM
antifungal cream cream |
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||
| Labeler - Jiangxi Hemei Pharmaceutical Co., Ltd (724892056) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Jiangxi Hemei Pharmaceutical Co., Ltd | 724892056 | manufacture(84010-046) | |