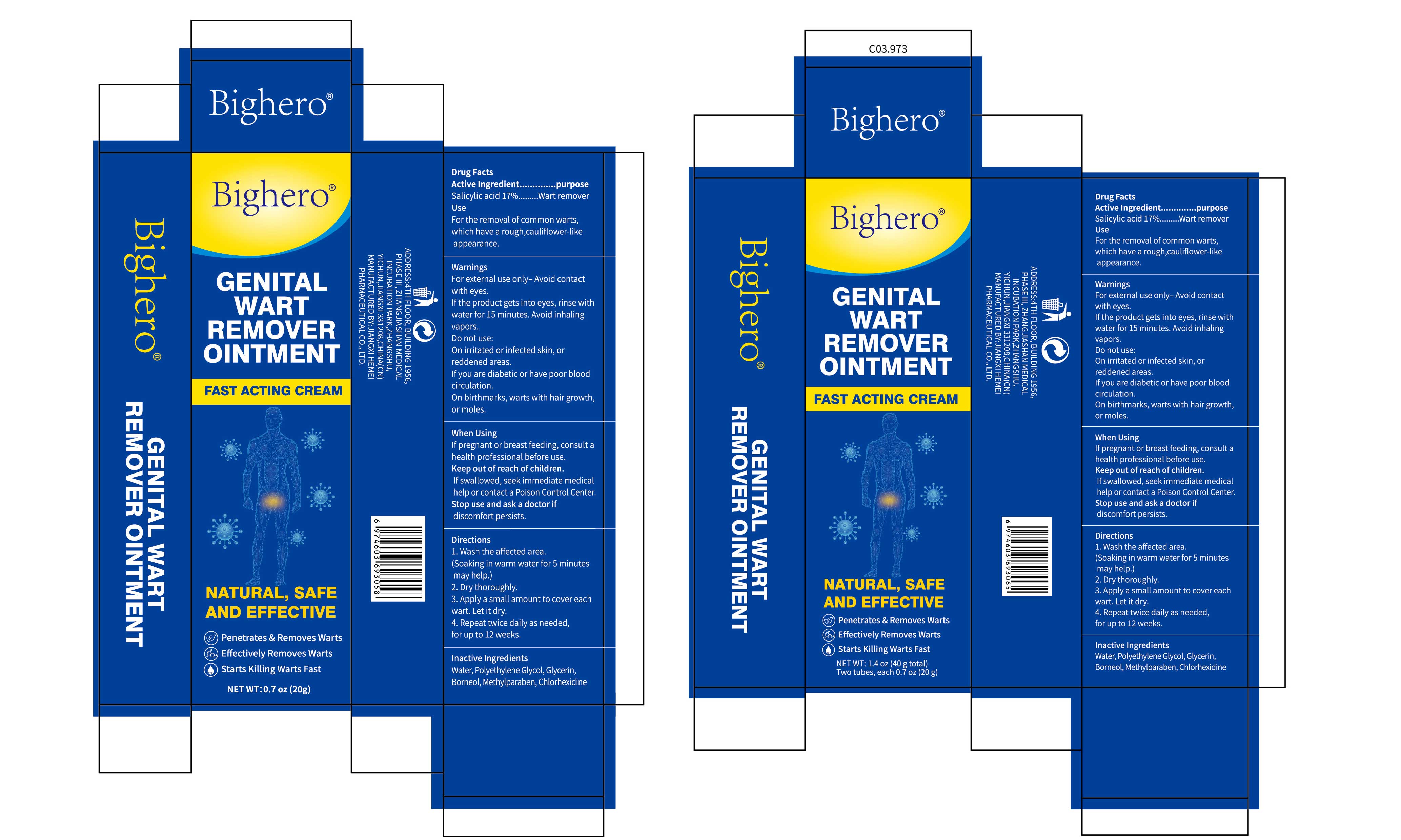
Bighero GENITAL WART REMOVER by Jiangxi Hemei Pharmaceutical Co., Ltd 84010-047 complete
Bighero GENITAL WART REMOVER by
Drug Labeling and Warnings
Bighero GENITAL WART REMOVER by is a Otc medication manufactured, distributed, or labeled by Jiangxi Hemei Pharmaceutical Co., Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
BIGHERO GENITAL WART REMOVER- salicylic acid 17%genital wart remover ointment
Jiangxi Hemei Pharmaceutical Co., Ltd
----------
84010-047 complete
Warnings
For external use only- Avoid contact with eyes.
If the product gets into eyes, rinse with water for 15 minutes. Avoid inhaling vapors.
Do not use
On irritated or infected skin, or reddened areas.
If you are diabetic or have poor blood circulation. On birthmarks, warts with hair growth,or moles.
Keep Oot Of Reach Of Children
If swallowed, seek immediate medicalhelp or contact a Poison Control Center.
Directions
1.Wash the affected area. (Soaking in warm water for 5 minutes may help.)
2. Dry thoroughly.
3. Apply a small amount to cover each wart. Let it dry
4. Repeat twice daily as needed, for up to 12 weeks.
| BIGHERO GENITAL WART REMOVER
salicylic acid 17%genital wart remover ointment |
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
|
|||||||||||||||||||||||||
| Labeler - Jiangxi Hemei Pharmaceutical Co., Ltd (724892056) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Jiangxi Hemei Pharmaceutical Co., Ltd | 724892056 | manufacture(84010-047) | |