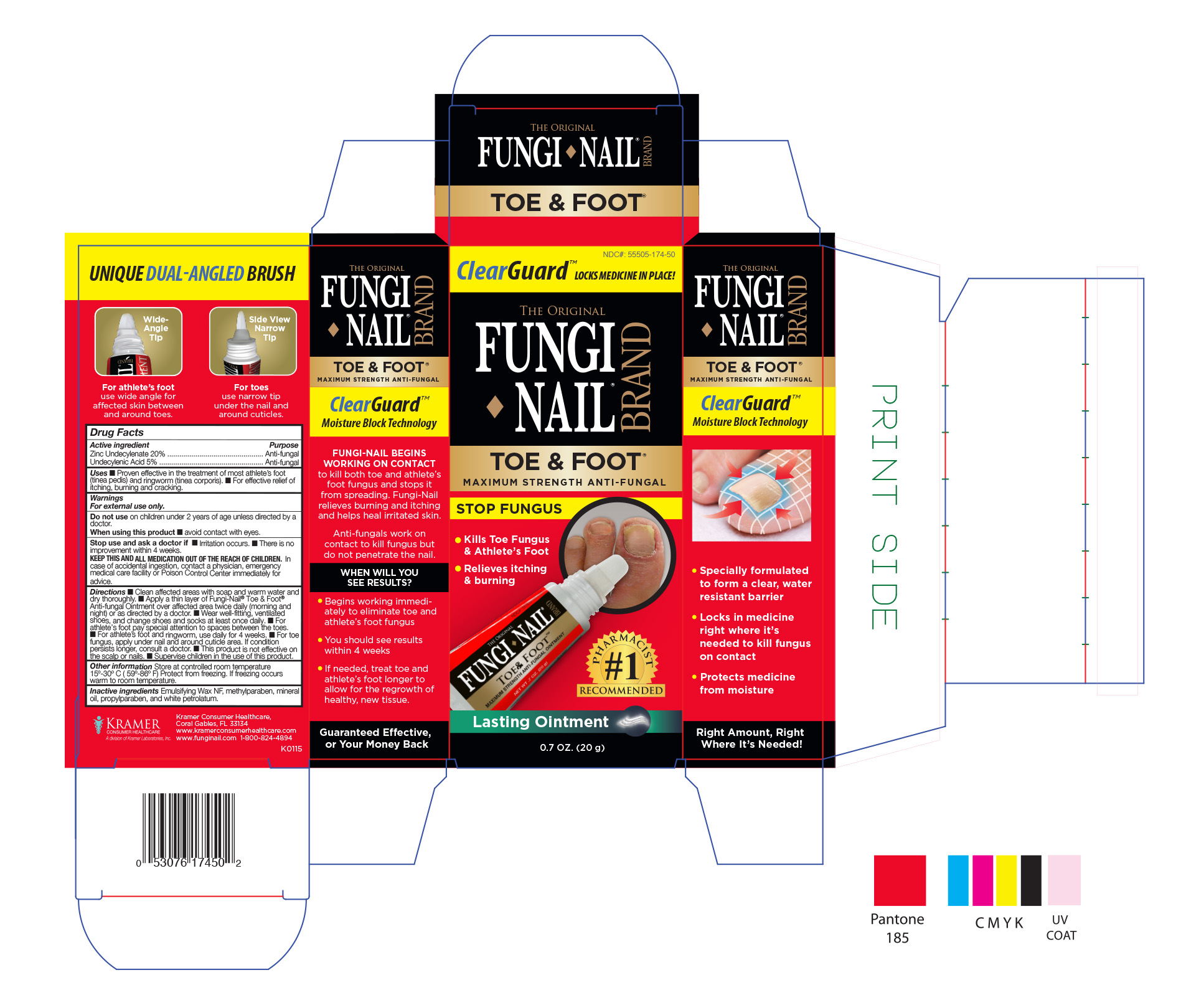
Fungi Nail Toe and Foot by Kramer Laboratories / Denison Pharmecuticals
Fungi Nail Toe and Foot by
Drug Labeling and Warnings
Fungi Nail Toe and Foot by is a Otc medication manufactured, distributed, or labeled by Kramer Laboratories, Denison Pharmecuticals. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
FUNGI NAIL TOE AND FOOT MS- zinc undecylenate undecylenic acid ointment
Kramer Laboratories
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
KEEP THIS AND ALL MEDICATION OUT OF THE REACH OF CHILDREN. In
case of accidental ingestion, contact a physician, emergency
medical care facility or Poison Control Center immediately for
advice.
Uses ■ Proven effective in the treatment of most athlete’s foot
(tinea pedis) and ringworm (tinea corporis). ■ For effective relief of
itching, burning and cracking.
Directions ■ Clean affected areas with soap and warm water and
dry thoroughly. ■ Apply a thin layer of Fungi-Nail® Toe & Foot™
Anti-fungal Ointment over affected area twice daily (morning and
night) or as directed by a doctor. ■ Wear well-fitting, ventilated
shoes, and change shoes and socks at least once daily. ■ For
athlete’s foot pay special attention to spaces between the toes.
■ For athlete’s foot and ringworm, use daily for 4 weeks. ■ For toe
fungus, apply under nail and around cuticle area. If condition
persists longer, consult a doctor. ■ This product is not effective on
the scalp or nails. ■ Supervise children in the use of this product.
| FUNGI NAIL TOE AND FOOT
MS
zinc undecylenate undecylenic acid ointment |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Kramer Laboratories (122720675) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Denison Pharmecuticals | 001207208 | manufacture(55505-174) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.