Super Instaready BB Powder_PF-Art
Super BB InstaReady Filter Trio BB Powder by
Drug Labeling and Warnings
Super BB InstaReady Filter Trio BB Powder by is a Otc medication manufactured, distributed, or labeled by Physicians Formula, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
SUPER BB INSTAREADY FILTER TRIO BB POWDER- titanium dioxide powder
Physicians Formula, Inc.
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
Super Instaready BB Powder_PF-Art
| SUPER BB INSTAREADY FILTER TRIO BB POWDER
titanium dioxide powder |
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||
| Labeler - Physicians Formula, Inc. (021261805) |
Revised: 12/2019
Document Id: 9a04d236-4fb5-7c3b-e053-2a95a90abc8a
Set id: 2a32ad22-ea75-128e-e054-00144ff88e88
Version: 4
Effective Time: 20191218
Physicians Formula, Inc.
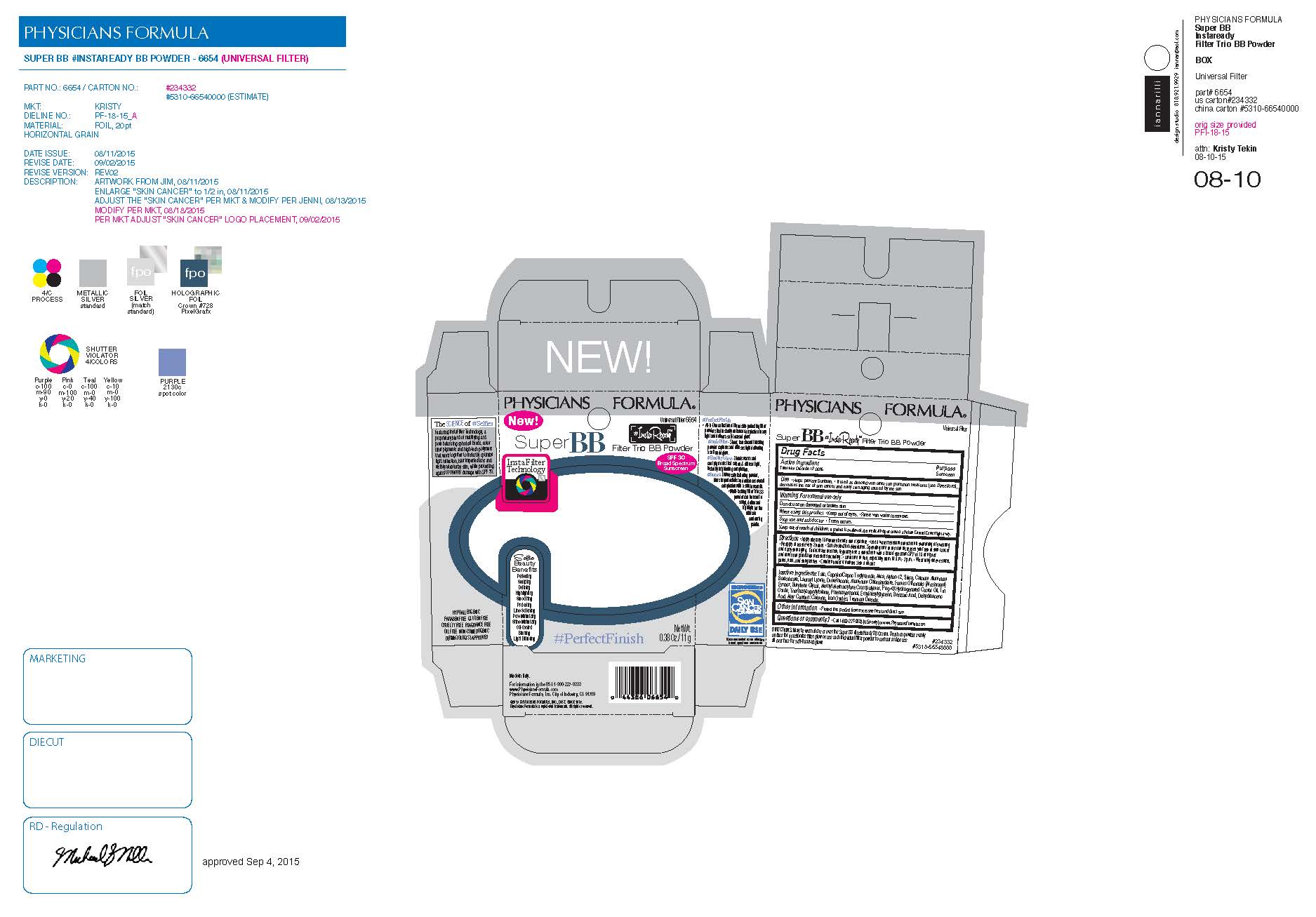 Principle Display Panel
Principle Display Panel