Sea-Laminaria Kelp Day Cream SPF 30
Sea-Laminaria Kelp Day Cream SPF 30 by
Drug Labeling and Warnings
Sea-Laminaria Kelp Day Cream SPF 30 by is a Otc medication manufactured, distributed, or labeled by CA-Botana International INC, CA- Botana International INC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
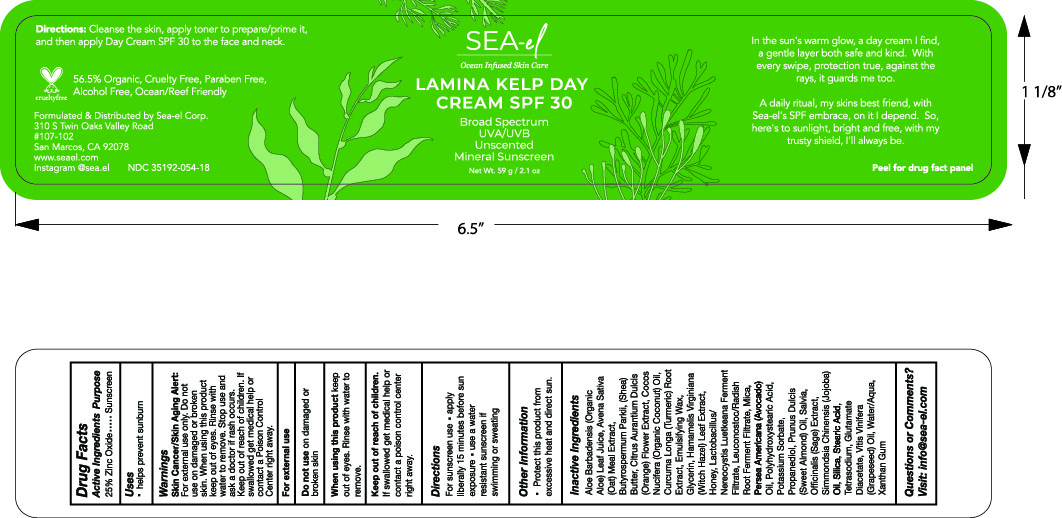
SEA-LAMINARIA KELP DAY CREAM SPF 30- sunscreen cream
CA-Botana International INC
----------
Sea-Laminaria Kelp Day Cream SPF 30
Dosage & Administration
Helps prevent sunburn. If used as directed with other sun protection measured decreases the risk of skin cancer and early skin aging caused by the sun. Apply liberally 15 minutes before sun exposure. Usea water resistant sunscreen if swimming or sweating. Reaply : at least 2 hours. Children under 6 months: Ask a doctor. Sun protections measurements. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a broad spectrum SPF of 15 or higher and othe sun protection measures including: limit time in the sun, specially from 10 a.m.- 2 p.m Wear long-sleeveshirts, pants, hats, and sunglasses. Protect this product from excessive heat and direct sun.
Warning section
For external use only Do not use on damaged or broken skin . When using this product keep out of eyes . Rinse with water to remove
If product is swallowed get medical help or contact a poison Center right away
Indications & Usage
Apply liberally 15 minutes before exposure, use waterresistant sunscreen, if swimming or sweating reaply at least every 2 hours Children under 6 months: ask a doctor
Inactive ingredients
Silica, Sage oil, Mica, Avocado oil, Witch Hazel,Potassium Sorbate, Prunus amigdalus dulcis (Sweet almond) oil, Citrus aurantium dulcis (orange) Flower, Chia seed oil, Stearic Acid, Curcuma longa (Tumeric) root, Polawax (Emulsifying wax), Polyhydroxystearic acid, Propanediol, Tetrasodium Glutamate Diacetate, Coconut oil, Glycerin, Nereocystis Luetkeana, Leuconostoc/Radish root ferment, Avena Sativa, Butyrospermum Parkii (Shea) Butter, Honey, Grapee seed oil, Water, Xanthan Gum, Jojoba oil, aloe
| SEA-LAMINARIA KELP DAY CREAM SPF 30
sunscreen cream |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Labeler - CA-Botana International INC (106276728) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| CA- Botana International INC | 106276728 | manufacture(35192-054) | |