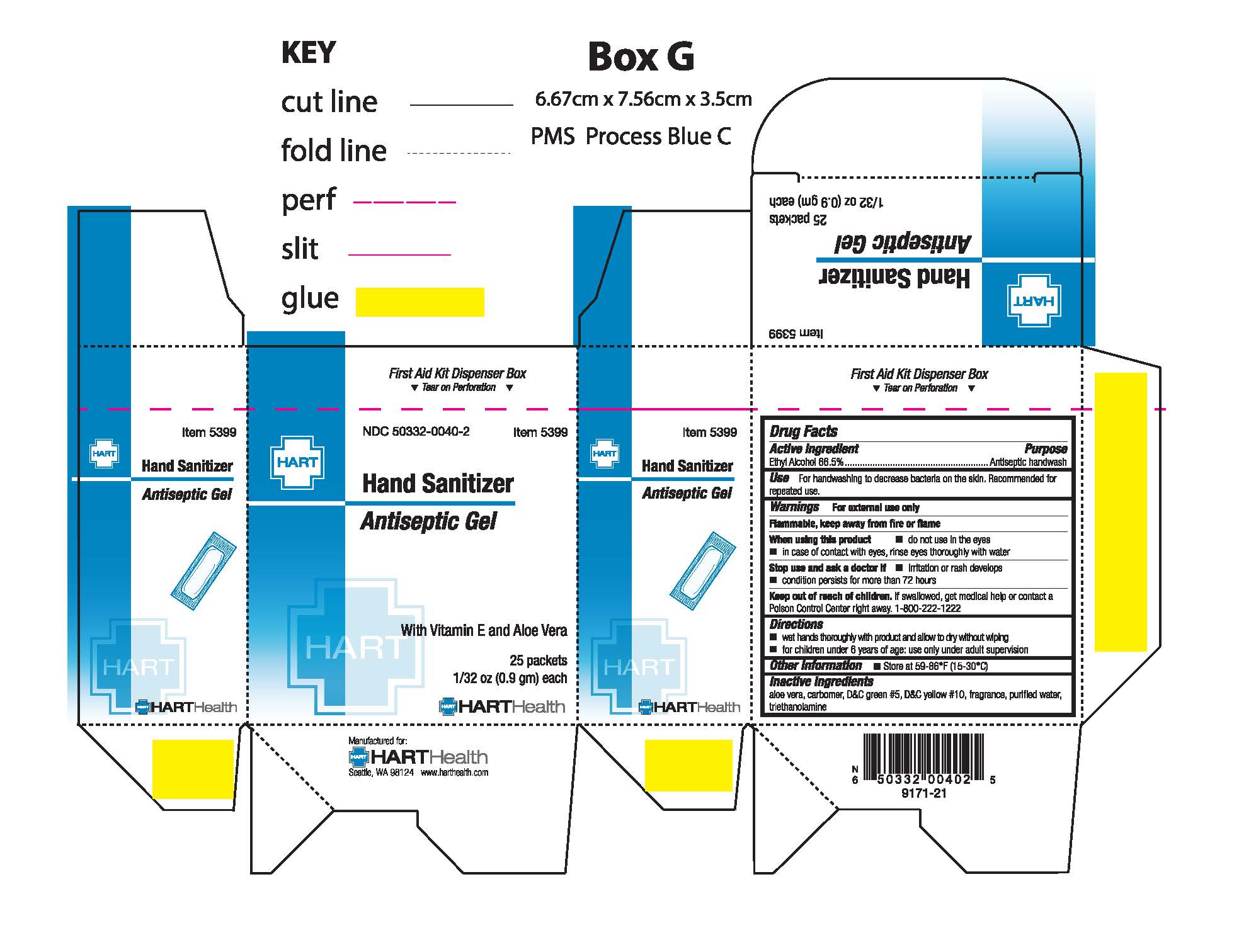
Hand Sanitizer Antiseptic Gel
Hand Sanitizer by
Drug Labeling and Warnings
Hand Sanitizer by is a Otc medication manufactured, distributed, or labeled by HART Health, WaterJel Technologies. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
HAND SANITIZER- antiseptic gel gel
HART Health
----------
Hand Sanitizer Antiseptic Gel
Stop use and ask a doctor if
- irritation and redness develop
- condition persists for more than 72 hours
Keep out of reach of children
If swallowed get medical help or contact a Poison Control Center right away. 1-800-222-1222
Directions
- wet hands thoroughly with product and allow to dry without wiping
- for children under 6 years of age; use only with adult supervision
| HAND SANITIZER
antiseptic gel gel |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - HART Health (069560969) |
| Registrant - HART Health (069560969) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| WaterJel Technologies | 155522589 | manufacture(50332-5394) | |
Revised: 1/2024
Document Id: 308b5263-070b-4da2-abbc-efe0358f73a8
Set id: 2c7110cd-e82d-442a-8738-f8d9c1ea4521
Version: 3
Effective Time: 20240127
Trademark Results [Hand Sanitizer]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 HAND SANITIZER 88958909 not registered Live/Pending |
MAISON BLANCHE, LLC 2020-06-10 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.