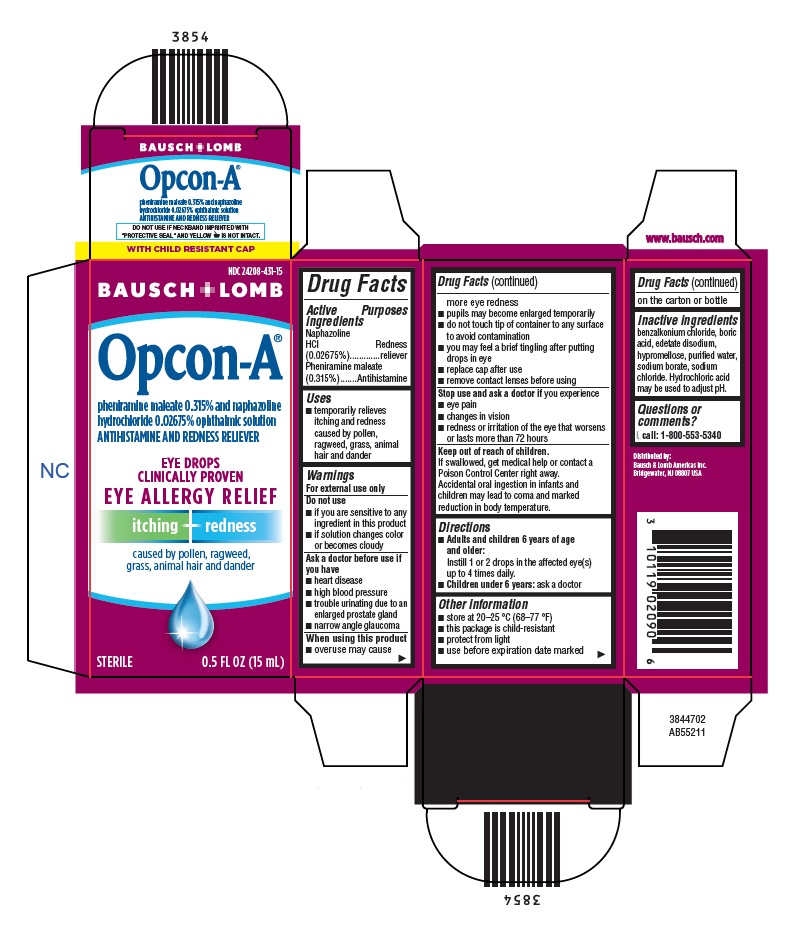
OPCON-A- naphazoline hydrochloride and pheniramine maleate solution/ drops
Opcon-A by
Drug Labeling and Warnings
Opcon-A by is a Otc medication manufactured, distributed, or labeled by Bausch & Lomb Incorporated. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active ingredients
- Purposes
- Uses
-
Warnings
For external use only
Do not use
- if you are sensitive to any ingredient in this product
- if solution changes color or becomes cloudy
Ask a doctor before use if you have
- heart disease
- high blood pressure
- trouble urinating due to an enlarged prostate gland
- narrow angle glaucoma
When using this product
- overuse may cause more eye redness
- pupils may become enlarged temporarily
- do not touch tip of container to any surface to avoid contamination
- you may feel a brief tingling after putting drops in eye
- replace cap after use
- remove contact lenses before using
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
Package/Label Principal Display Panel
NDC: 24208-431-15
BAUSCH + LOMB
Opcon-A ®
pheniramine maleate 0.315% and naphazoline
hydrochloride 0.02675% ophthalmic solution
ANTIHISTAMINE AND REDNESS RELIEVER
EYE DROPS
CLINICALLY PROVEN
EYE ALLERGY RELIEFitching + redness
caused by pollen, ragweed,
grass, animal hair and dander
[teardrop icon]
STERILE 0.5 FL OZ (15 mL)
3844702
AB55211 -
INGREDIENTS AND APPEARANCE
OPCON-A
naphazoline hydrochloride and pheniramine maleate solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 24208-431 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NAPHAZOLINE HYDROCHLORIDE (UNII: MZ1131787D) (NAPHAZOLINE - UNII:H231GF11BV) NAPHAZOLINE HYDROCHLORIDE 0.2675 mg in 1 mL PHENIRAMINE MALEATE (UNII: NYW905655B) (PHENIRAMINE - UNII:134FM9ZZ6M) PHENIRAMINE MALEATE 3.15 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) BORIC ACID (UNII: R57ZHV85D4) EDETATE DISODIUM (UNII: 7FLD91C86K) WATER (UNII: 059QF0KO0R) SODIUM BORATE (UNII: 91MBZ8H3QO) SODIUM CHLORIDE (UNII: 451W47IQ8X) HYDROCHLORIC ACID (UNII: QTT17582CB) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 24208-431-15 1 in 1 CARTON 03/19/2026 1 15 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product 2 NDC: 24208-431-45 2 in 1 CARTON 03/19/2026 2 15 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA020065 03/19/2026 Labeler - Bausch & Lomb Incorporated (196603781) Establishment Name Address ID/FEI Business Operations Bausch & Lomb Incorporated 079587625 manufacture(24208-431)
Trademark Results [Opcon-A]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 OPCON-A 74509424 1993877 Live/Registered |
BAUSCH & LOMB INCORPORATED 1994-04-06 |
 OPCON-A 73623284 1482990 Dead/Cancelled |
MURO PHARMACEUTICAL, INC. 1986-10-01 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.