ASPIRIN tablet, delayed release
Aspirin by
Drug Labeling and Warnings
Aspirin by is a Otc medication manufactured, distributed, or labeled by Topco Associates, LLC, LNK International, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
Reye’s syndrome: Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye's syndrome, a rare but serious illness.
Allergy alert: Aspirin may cause a severe allergic reaction, which may include:
- hives
- facial swelling
- shock
- asthma (wheezing)
Stomach bleeding warning: This product contains an NSAID, which may cause severe stomach bleeding. The chance is higher if you
- take other drugs containing prescription or nonprescription NSAIDs [aspirin, ibuprofen, naproxen, or others]
- have had stomach ulcers or bleeding problems
- take a blood thinning (anticoagulant) or steroid drug
- are age 60 or older
- have 3 or more alcoholic drinks every day while using this product
- take more or for a longer time than directed
Do not use
- if you are allergic to aspirin or any other pain reliever/fever reducer
- if you have ever had an allergic reaction to this product or any of its ingredients
Ask a doctor before use if
- stomach bleeding warning applies to you
- you have a history of stomach problems, such as heartburn
- you have high blood pressure, heart disease, liver cirrhosis, or kidney disease
- you have asthma
- you are taking a diuretic
Ask a doctor or pharmacist before use if you are
taking a prescription drug for
- gout
- diabetes
- arthritis
Stop use and ask a doctor if
- an allergic reaction occurs. Seek medical help right away.
- you experience any of the following signs of stomach bleeding:
- feel faint
- vomit blood
- have bloody or black stools
- have stomach pain that does not get better
- ringing in the ears or a loss of hearing occurs
- new symptoms occur
- pain gets worse or lasts more than 10 days
- redness or swelling is present
These could be signs of a serious condition.
- Directions
- Other information
-
Inactive ingredients
black iron oxide, cellulose, colloidal anhydrous silica, corn starch, D&C yellow #10 aluminum lake, FD&C yellow #6 aluminum lake, hypromellose, polydextrose, polyethylene glycol, polyvinyl acetate phthalate, propylene glycol, shellac wax, simethicone, sodium alginate, sodium bicarbonate, stearic acid, talc, titanium dioxide, triacetin, triethyl citrate
- Questions or comments?
-
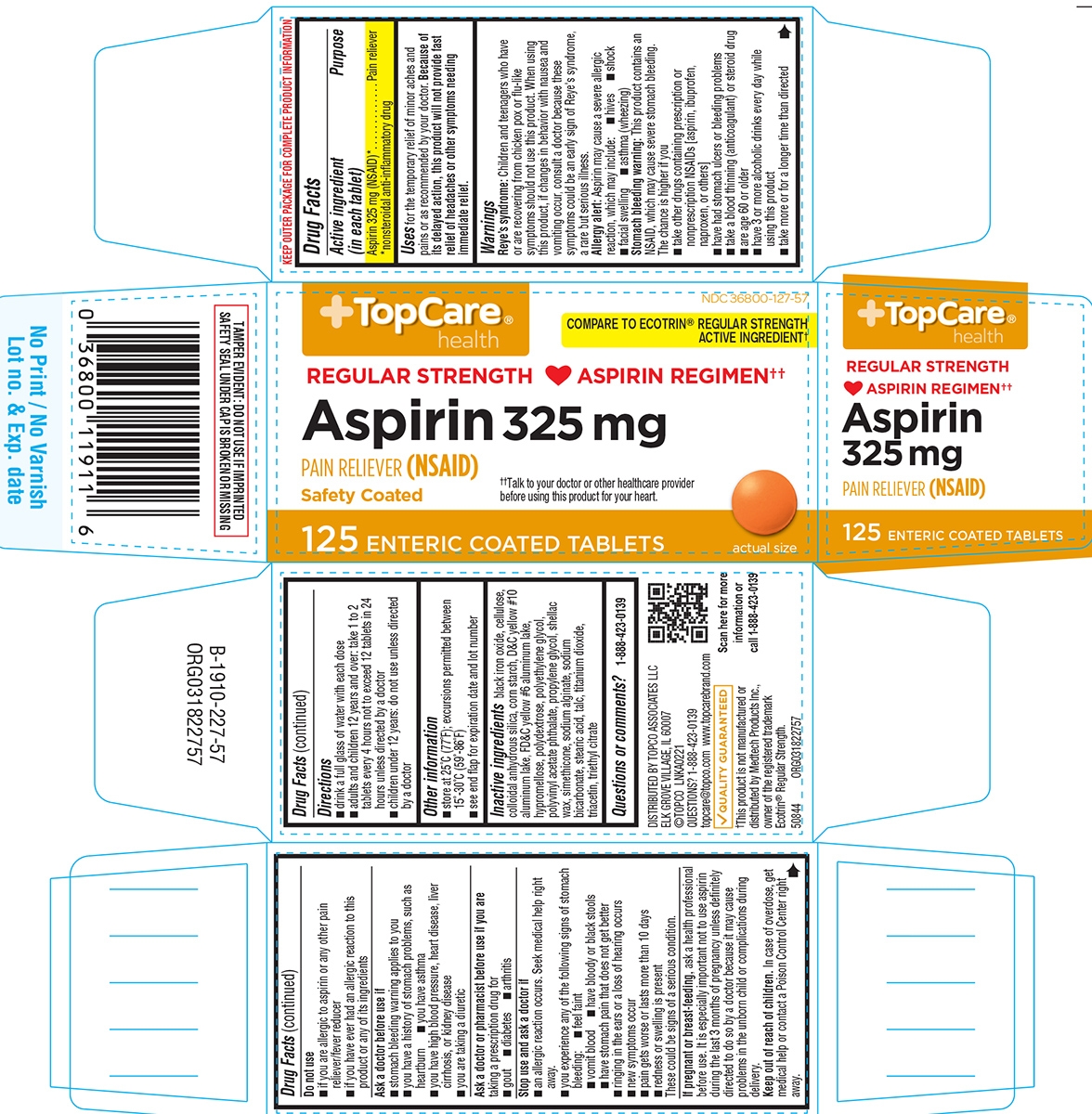
Principal Display Panel
TopCare®
healthNDC: 36800-127-57
COMPARE TO ECOTRIN® REGULAR STRENGTH
ACTIVE INGREDIENT†REGULAR STRENGTH ASPIRIN REGIMEN††
Aspirin 325 mg
Pain reliever (NSAID)Safety Coated
††Talk to your doctor or other healthcare provider before using this product for your heart.
125 ENTERIC COATED TABLETS
Actual Size
TAMPER EVIDENT: DO NOT USE IF IMPRINTED
SAFETY SEAL UNDER CAP IS BROKEN OR MISSINGDISTRIBUTED BY TOPCO ASSOCIATES LLC
ELK GROVE VILLAGE, IL 60007
©TOPCO LNKA0221
QUESTIONS? 1-888-423-0139
topcare@topco.com www.topcarebrand.comQUALITY GUARANTEED
†This product is not manufactured or
distributed by Medtech Products Inc.,
owner of the registered trademark
Ecotrin® Regular Strength.50844 ORG031822757
44-227
-
INGREDIENTS AND APPEARANCE
ASPIRIN
aspirin tablet, delayed releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 36800-127 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ASPIRIN (UNII: R16CO5Y76E) (ASPIRIN - UNII:R16CO5Y76E) ASPIRIN 325 mg Inactive Ingredients Ingredient Name Strength FERROSOFERRIC OXIDE (UNII: XM0M87F357) POWDERED CELLULOSE (UNII: SMD1X3XO9M) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) STARCH, CORN (UNII: O8232NY3SJ) D&C YELLOW NO. 10 ALUMINUM LAKE (UNII: CQ3XH3DET6) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) POLYDEXTROSE (UNII: VH2XOU12IE) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) Polyvinyl Acetate Phthalate (UNII: 58QVG85GW3) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SHELLAC (UNII: 46N107B71O) DIMETHICONE (UNII: 92RU3N3Y1O) WATER (UNII: 059QF0KO0R) SODIUM ALGINATE (UNII: C269C4G2ZQ) SODIUM BICARBONATE (UNII: 8MDF5V39QO) STEARIC ACID (UNII: 4ELV7Z65AP) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIACETIN (UNII: XHX3C3X673) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) Product Characteristics Color orange Score no score Shape ROUND Size 10mm Flavor Imprint Code 44;227 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 36800-127-57 1 in 1 CARTON 04/13/2021 04/05/2025 1 125 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M013 04/13/2021 04/05/2025 Labeler - Topco Associates, LLC (006935977) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 038154464 manufacture(36800-127) , pack(36800-127) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867837 manufacture(36800-127) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867894 manufacture(36800-127) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 868734088 manufacture(36800-127) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 967626305 pack(36800-127) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 117025878 manufacture(36800-127)
Trademark Results [Aspirin]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 ASPIRIN 76481358 2788617 Dead/Cancelled |
Simon Carter Accessories, Ltd. 2003-01-10 |
 ASPIRIN 75209895 not registered Dead/Abandoned |
Bayer Aktiengesellschaft 1996-12-09 |
 ASPIRIN 73234351 1171777 Dead/Cancelled |
McIntyre; William A. 1979-10-09 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.