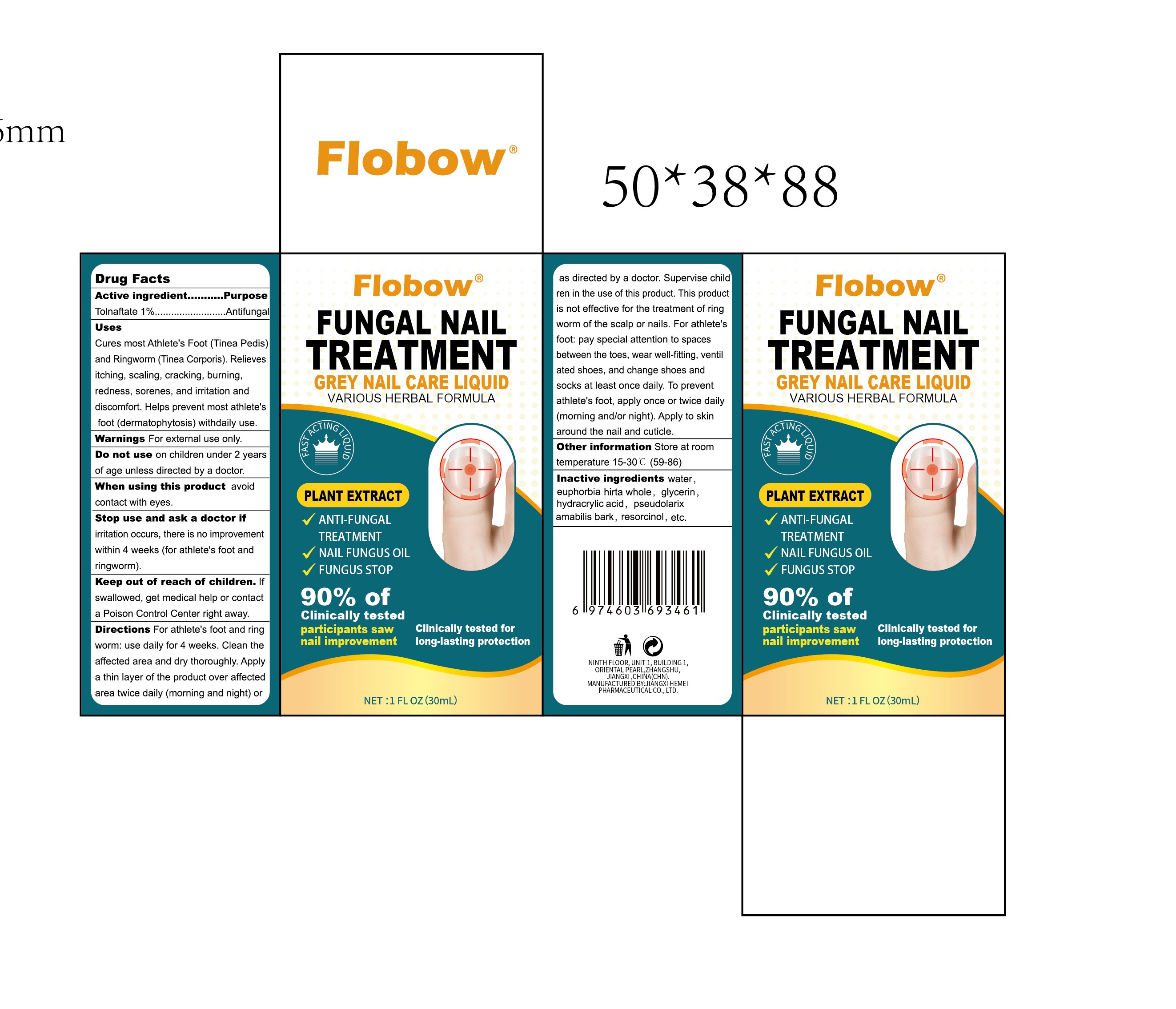
FIobow FUNGAL NAIL TREATMENT by Jiangxi Hemei Pharmaceutical Co., Ltd 84010-071 complete
FIobow FUNGAL NAIL TREATMENT by
Drug Labeling and Warnings
FIobow FUNGAL NAIL TREATMENT by is a Otc medication manufactured, distributed, or labeled by Jiangxi Hemei Pharmaceutical Co., Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
FIOBOW FUNGAL NAIL TREATMENT- tolnaftate 1% fungal nail treatment liquid
Jiangxi Hemei Pharmaceutical Co., Ltd
----------
84010-071 complete
Use
Cures most Athlete's Foot (Tinea Pedis) and Ringworm (Tinea Corporis). Relieves itching, scaling, cracking, burning, redness, sorenes, and irritation and discomfort. Helps prevent most athlete's foot (dermatophytosis) with daily use.
Stop Use
irritation occurs, there is no improvement within 4 weeks (for athlete's foot and ringworm).
Ask Doctor
irritation occurs, there is no improvement within 4 weeks (for athlete's foot and ringworm).
Keep Out Of Reach Of Children
If swallowed, get medical help or contact a Poison Control Center right away.
Directions
For athlete's foot and ring
worm: use daily for 4 weeks. Clean the affected area and dry thoroughly. Apply a thin layer of the product over affected
area twice daily (morning and night) or as directed by a doctor. Supervise children in the use of this product. This product is not effective for the treatment of ring worm of the scalp or nails.
For athlete's foot: pay special attention to spaces between the toes, wear well-fitting, ventil ated shoes, and change shoes and socks at least once daily. To prevent athlete's foot, apply once or twice daily (morning and/or night). Apply to skin around the nail and cuticle.
| FIOBOW FUNGAL NAIL TREATMENT
tolnaftate 1% fungal nail treatment liquid |
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
| Labeler - Jiangxi Hemei Pharmaceutical Co., Ltd (724892056) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Jiangxi Hemei Pharmaceutical Co., Ltd | 724892056 | manufacture(84010-071) | |