Allevia by Florrax Pharmaceutical Corp. Allevia
Allevia by
Drug Labeling and Warnings
Allevia by is a Prescription medication manufactured, distributed, or labeled by Florrax Pharmaceutical Corp.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
ALLEVIA- vitamin c, d3, b1, b6, b12, folate, nadh, coenzyme q-10 tablet
Florrax Pharmaceutical Corp.
Disclaimer: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
----------
Allevia
| ALLEVIA
vitamin c, d3, b1, b6, b12, folate, nadh, coenzyme q-10 tablet |
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||
| Labeler - Florrax Pharmaceutical Corp. (119257612) |
| Registrant - Florrax Pharmaceutical Corp. (119257612) |
Revised: 12/2025
Document Id: 460509fe-ef09-148d-e063-6394a90a2818
Set id: 2f235b67-e688-a377-e063-6394a90a6fee
Version: 3
Effective Time: 20251215
Trademark Results [Allevia]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
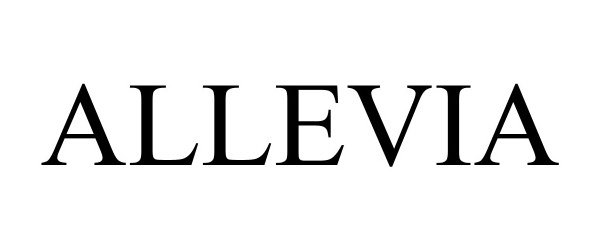 ALLEVIA 98104420 not registered Live/Pending |
Erbe Elektromedizin GmbH 2023-07-27 |
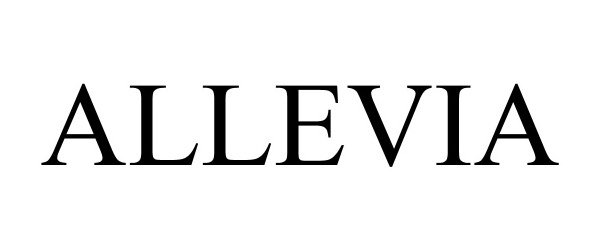 ALLEVIA 90006193 not registered Live/Pending |
BIOTRONIK SE & Co. KG 2020-06-17 |
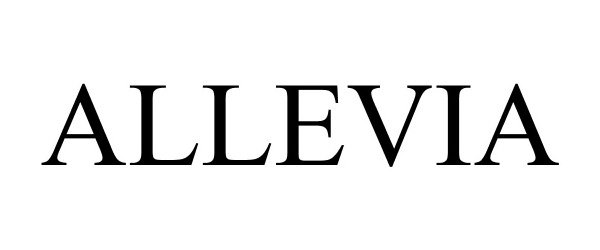 ALLEVIA 88367596 not registered Live/Pending |
Accora Limited 2019-04-02 |
 ALLEVIA 88152241 not registered Dead/Abandoned |
Keeco, LLC 2018-10-11 |
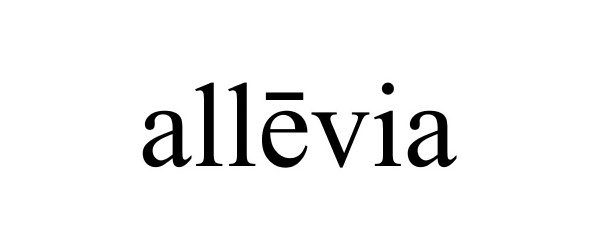 ALLEVIA 87340756 not registered Dead/Abandoned |
Texas Ear Nose & Throat Specialists, P.A. 2017-02-17 |
 ALLEVIA 87340743 not registered Dead/Abandoned |
Texas Ear Nose & Throat Specialists, P.A. 2017-02-17 |
 ALLEVIA 87340707 not registered Dead/Abandoned |
Texas Ear Nose & Throat Specialists, P.A. 2017-02-17 |
 ALLEVIA 87168956 5195064 Live/Registered |
Innvictis Crop Care, LLC 2016-09-13 |
 ALLEVIA 86973511 5073318 Live/Registered |
L&P Property Management Company 2016-04-12 |
 ALLEVIA 81003689 1003689 Dead/Cancelled |
Alexandra De Markoff Sales Corporation 0000-00-00 |
 ALLEVIA 77866686 not registered Dead/Abandoned |
Exquim S.A. 2009-11-06 |
 ALLEVIA 76666009 not registered Dead/Abandoned |
NEWTON-EVERETT, LLC 2006-09-14 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
