CARISOPRODOL by DIRECT RX CARISOPRODOL tablet
CARISOPRODOL by
Drug Labeling and Warnings
CARISOPRODOL by is a Prescription medication manufactured, distributed, or labeled by DIRECT RX. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- 1 INDICATIONS AND USAGE
- 2 DOSAGE AND ADMINISTRATION
- 3 DOSAGE FORM AND STRENGTH
- 4 CONTRAINDICATIONS
- 5 WARNINGS AND PRECAUTIONS
- 6 ADVERSE REACTIONS
- 7 DRUG INTERACTIONS
- 8 USE IN SPECIFIC POPULATION
- 9 DRUG ABUSE AND DEPENDENCE
- 10 OVERDOSAGE
- 11 DESCRIPTION
- 12 CLINCIAL PHARMACOLOGY
- 13 NONCLINICAL TOXICOLOGY
- 14 CLINICAL STUDIES
- 15 STORAGE AND HANDLING
- 17 PATIENT COUNSELING INFORMATION
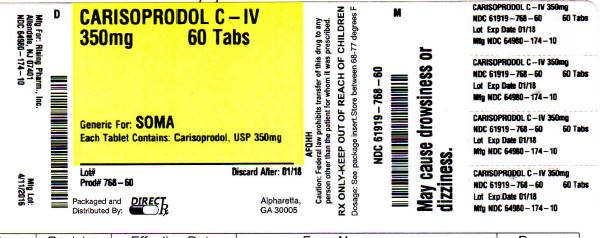
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CARISOPRODOL
carisoprodol tabletProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 61919-768(NDC:64980-174) Route of Administration ORAL DEA Schedule CIV Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CARISOPRODOL (UNII: 21925K482H) (CARISOPRODOL - UNII:21925K482H) CARISOPRODOL 350 mg Inactive Ingredients Ingredient Name Strength HYDROXYPROPYL CELLULOSE (TYPE H) (UNII: RFW2ET671P) ANHYDROUS LACTOSE (UNII: 3SY5LH9PMK) STARCH, CORN (UNII: O8232NY3SJ) MAGNESIUM STEARATE (UNII: 70097M6I30) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) SODIUM LAURYL SULFATE (UNII: 368GB5141J) Product Characteristics Color white Score no score Shape ROUND Size 5mm Flavor Imprint Code CL;022 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 61919-768-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 04/01/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA040823 04/01/2016 Labeler - DIRECT RX (079254320) Establishment Name Address ID/FEI Business Operations DIRECT RX 079254320 repack(61919-768)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.