NASOCALM- sodium chloride spray
Nasocalm by
Drug Labeling and Warnings
Nasocalm by is a Otc medication manufactured, distributed, or labeled by Virexion Health Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- WARNINGS
- PURPOSE
- ACTIVE INGREDIENT
-
INDICATIONS & USAGE
Gently hydrates and soothes dry nasal passages.
Flushes out allergens, dust, and airborne pollutants.
Relieves mild nasal congestion caused by colds, flu, allergies, or dry environments.
Ideal for daily nasal hygiene, suitable for infants, pregnant women, and all ages.
Also used for nasal irrigation in patients with acute and chronic rhinitis, allergic rhinitis, nasal polyps, sinusitis, and other nasal diseases.
For use after rhinitis surgery or chemotherapy to help clear nasal passages. - DO NOT USE
- KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
Directions
1. Shake the bottle gently before use.
2. Adults & Children (6+ years): Spray 1-2 times per nostril, up to 6 times daily.
3. Infants & Young Children (under 6 years): Use 1 spray per nostril, up to 4 times daily, or as directed by a healthcare professional.
4. To apply, gently tilt head forward, insert nozzle into nostril, and press the pump while inhaling lightly.
5. Wipe the nozzle after use and close the cap securely.
6. Children and infants must be used under adult supervision. For children under 5 years old, please follow the advice of a healthcare professional.
- STORAGE AND HANDLING
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
NASOCALM
sodium chloride sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 85436-001 Route of Administration NASAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) (SODIUM CATION - UNII:LYR4M0NH37) SODIUM CHLORIDE 0.9 mg in 100 mg Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Product Characteristics Color Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 85436-001-01 1 in 1 BOX 03/28/2025 1 100 mg in 1 BOTTLE; Type 0: Not a Combination Product 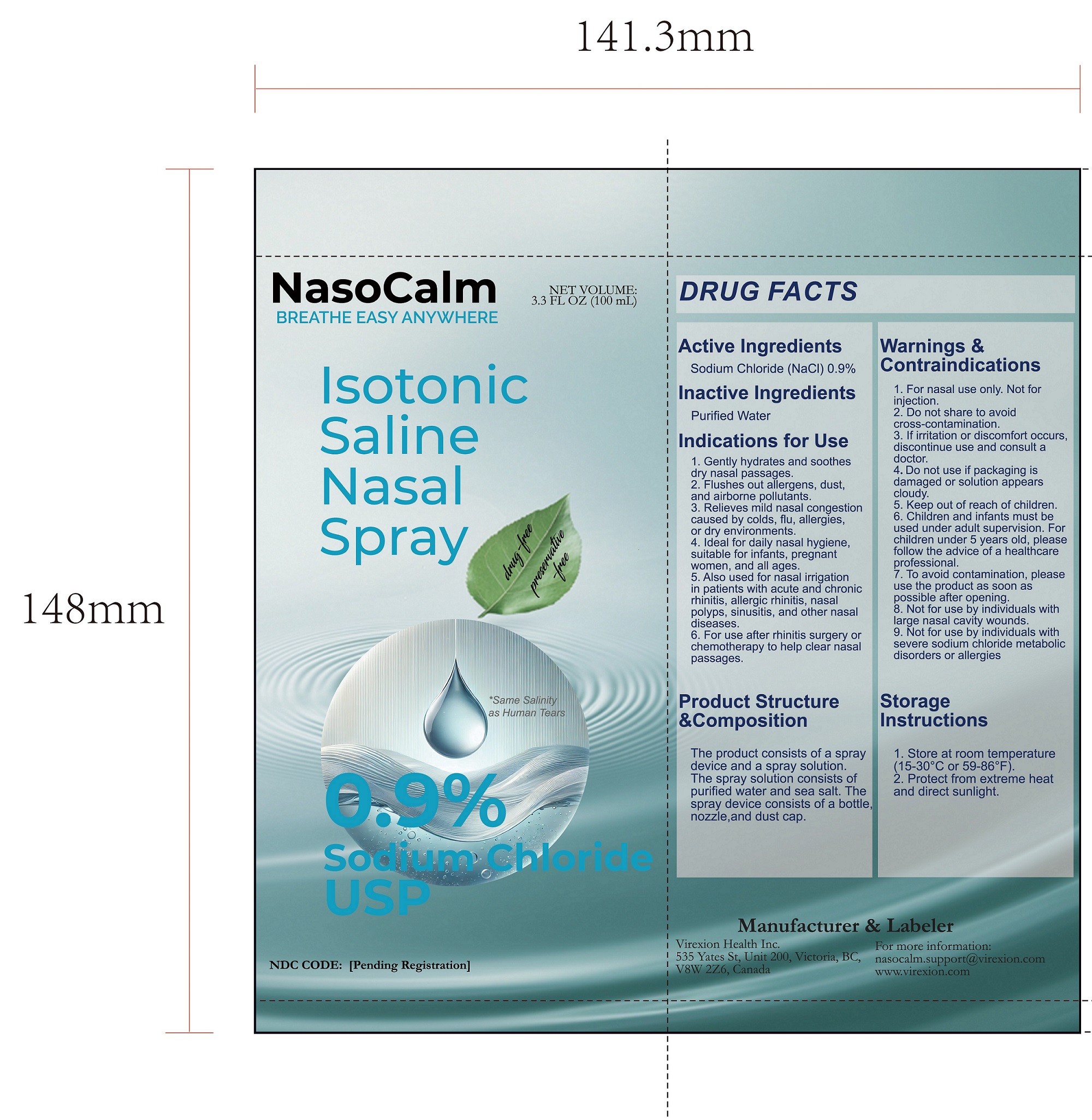
Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M016 03/28/2025 Labeler - Virexion Health Inc. (243333153) Establishment Name Address ID/FEI Business Operations Virexion Health Inc. 243333153 manufacture(85436-001) , label(85436-001)
Trademark Results [Nasocalm]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 NASOCALM 98316088 not registered Live/Pending |
Virexion Technology Limited 2023-12-15 |
 NASOCALM 86259156 not registered Dead/Abandoned |
Pulmatrix Inc. 2014-04-22 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
