BeC Racepinephrine by Blue Echo Care LLC
BeC Racepinephrine by
Drug Labeling and Warnings
BeC Racepinephrine by is a Otc medication manufactured, distributed, or labeled by Blue Echo Care LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
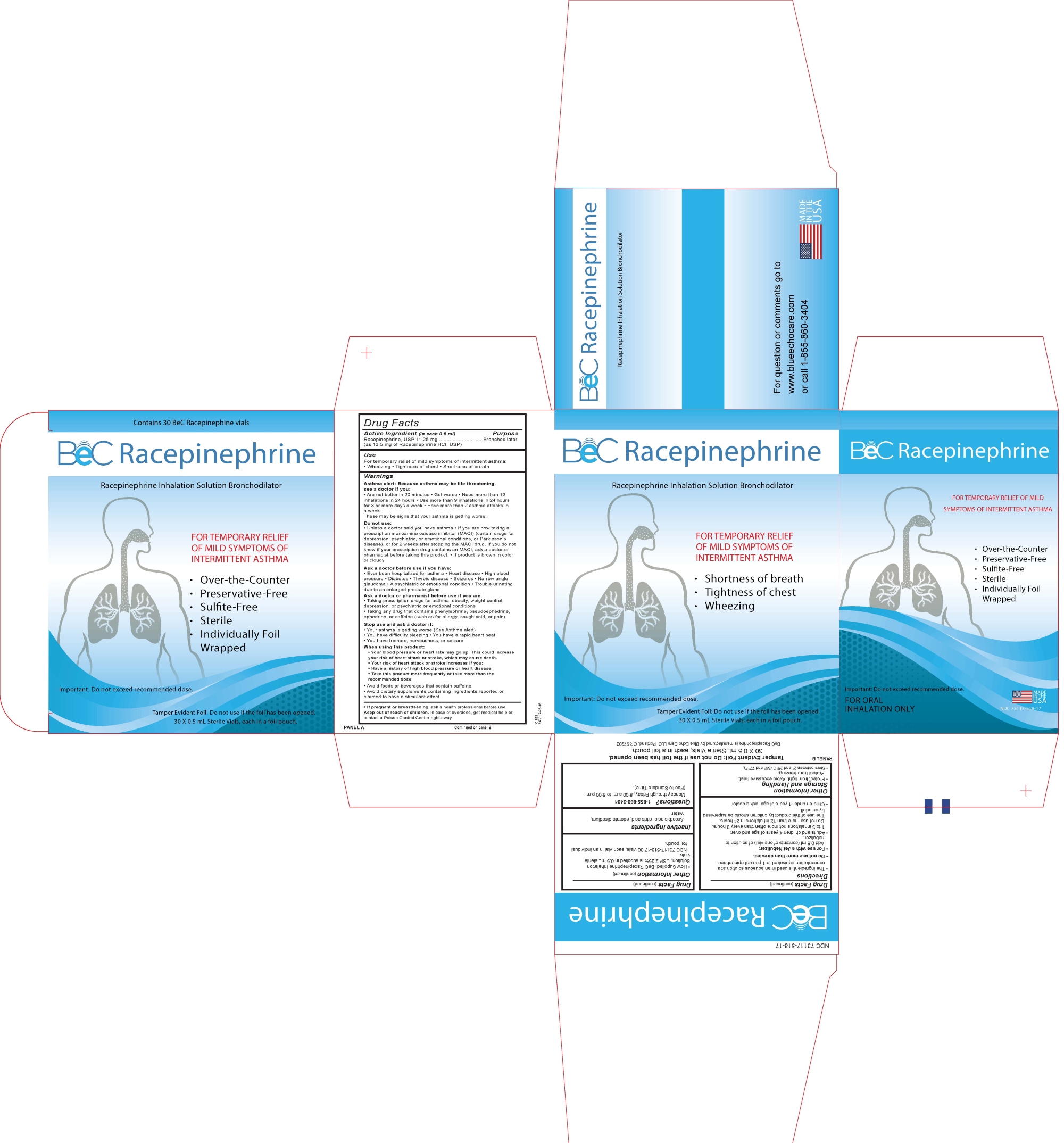
BEC RACEPINEPHRINE- racepinephrine liquid
Blue Echo Care LLC
----------
Uses
For temporary relief of mild symptoms of intermittent asthma:
Wheezing
Tightness of chest
Shortness of breath
Warning
Asthma alert: Because asthma may be life threatening, see a doctor if you:
- Are not better in 20 minutes
- Get worse
- Need more than 12 inhalations in 24 hours
- Use more than 9 inhalations in 24 hours for 3 or more days a week
- Have more than 2 asthma attacks in a week
These may be signs that your asthma is getting worse.
Do not use:
- Unless a doctor said you have asthma
- If you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains a MAOI, ask a doctor or pharmacist before taking this product.
- If product is brown in color or cloudy
Ask a doctor before use if you have:
- Ever been hospitalized for asthma
- Heart disease
- High blood pressure
- Diabetes
- Thyroid disease
- Seizures
- Narrow angle glaucoma
- A psychiatric or emotional condition
- Trouble urinating due to an enlarged prostate gland
Ask a doctor or pharmacist before use if you are:
- Taking prescription drugs for asthma, obesity, weight control, depression, or psychiatric or emotional conditions.
- Taking any drug that contains phenylephrine, pseudoephedrine, ephedrine, or caffeine (such as for allergy, cough-cold, or pain).
Stop use and ask a doctor if:
- Your asthma is getting worse (See Asthma alert)
- You have difficulty sleeping
- You have a rapid heartbeat
- You have tremors, nervousness, or seizure
When using this product:
- Your blood pressure or heart rate may go up. This could increase your risk of heart attack or stroke, which may cause death.
- Your risk of heart attack or stroke increases if you:
- Have a history of high blood pressure or heart disease
- Take this product more frequently or take more than the recommended dose.
- Avoid foods or beverages that contain caffeine
- Avoid dietary supplements containing ingredients reported or claimed to have a stimulant effect.
- If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away.
Directions
- The ingredient is used in an aqueous solution at a concentration equivalent to 1 percent epinephrine.
- Do not use more than directed
- For use with a Jet Nebulizer.
- Add 0.5 mL (contents of one vial) of solution to Nebulizer.
- Adults and children 4 years of age and over:
1 to 3 inhalations not more often than every 3 hours.
Do not use more than 12 inhalations in 24 hours.
The use of this product by children should be supervised by an adult.
- Children under 4 years of age: ask a doctor.
Other Information
Storage and Handling
Protect from light. Avoid excessive heat. Protect from freezing.
Store between 2°C and 25°C (36°F and 77°F).
How Supplied: BeC ® Racepinephrine Inhalation Solution, USP 2.25% is supplied in 0.5 mL sterile vials.
30 vials, each vial in an individual pouch.
| BEC RACEPINEPHRINE
racepinephrine liquid |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - Blue Echo Care LLC (117048847) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Blue Echo Care LLC | 117048847 | manufacture(73117-518) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.