BUDESONIDE NASAL- budesonide spray, metered
Budesonide Nasal by
Drug Labeling and Warnings
Budesonide Nasal by is a Prescription medication manufactured, distributed, or labeled by Apotex Corp.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use Budesonide Nasal Spray safely and effectively. See full prescribing information for Budesonide Nasal Spray.
Budesonide Nasal Spray, 32 mcg
INITIAL U.S. APPROVAL: 1999INDICATIONS AND USAGE
Budesonide nasal spray is a corticosteroid indicated for:
- Treatment of seasonal or perennial allergic rhinitis in adults and children ≥ 6 years. (1.1)
DOSAGE AND ADMINISTRATION
For intranasal use only.
Starting dosage:
- Adult and children 6 years of age and older: 64 mcg (one spray per nostril) once daily. (2)
Maximum dosages:
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
Hypersensitivity to ingredients. (4)
WARNINGS AND PRECAUTIONS
- Local Nasal Effects: Epistaxis, Candida albicans infections, nasal septal perforation, impaired wound healing. Monitor patients periodically for signs of adverse effects on the nasal mucosa. Avoid use in patients with recent nasal ulcers, nasal surgery, or nasal trauma. (5.1)
- Hypersensitivity reactions: Anaphylaxis, urticaria, rash, dermatitis, angioedema and pruritus may occur after administration of budesonide nasal spray. (5.2)
- Immunosuppression: Potential worsening of infections (e.g., existing tuberculosis, fungal bacterial, viral, or parasitic infections; or ocular herpes simplex). Use with caution in patients with these infections. More serious or even a fatal course of chickenpox or measles can occur in susceptible patients. (5.3)
- Hypercorticism and adrenal suppression: May occur with very high dosages or at the regular dose in susceptible individuals. If such changes occur, reduce treatment with budesonide nasal spray slowly. (5.4)
- Effect on growth: Monitor growth of pediatric patients. (5.6)
- Glaucoma and cataracts: Close monitoring is warranted. (5.7)
ADVERSE REACTIONS
- Most common adverse reactions (incidence ≥ 2%): epistaxis, pharyngitis, bronchospasm, coughing, nasal irritation. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Apotex Corp. at 1-800-667-4708 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 7/2012
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
1.1 Treatment of Seasonal or Perennial Allergic Rhinitis
2 DOSAGE AND ADMINISTRATION
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Local Nasal Effects
5.2 Hypersensitivity Reactions Including Anaphylaxis
5.3 Immunosuppression
5.4 Hypothalamic-Pituitary-Adrenal Axis Effects
5.5 Interactions with Strong Cytochrome P450 3A4 Inhibitors
5.6 Effect on Growth
5.7 Glaucoma and Cataracts
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Post-marketing Experience
7 DRUG INTERACTIONS
7.1 Inhibitors of Cytochrome P450 3A4
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.3 Nursing Mothers
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
15 REFERENCES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
17.1 Local Nasal Effects
17.2 Hypersensitivity including Anaphylaxis
17.3 Immunosuppression
17.4 Reduced Growth Velocity
17.5 Glaucoma and Cataracts
17.6 Use Daily
17.7 How to Use Budesonide Nasal Spray
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
The recommended starting dosage for adults and children 6 years of age and older is 64 mcg per day administered as one spray per nostril of budesonide nasal spray 32 mcg once daily. Some patients who do not achieve symptom control at the recommended starting dosage may benefit from an increased dosage. The maximum recommended dosage for adults (12 years of age and older) is 256 mcg per day administered as four sprays per nostril once daily of budesonide nasal spray 32 mcg and the maximum recommended dose for pediatric patients (6 to <12 years of age) is 128 mcg per day administered as two sprays per nostril once daily of budesonide nasal spray 32 mcg.
It is always desirable to titrate an individual patient to the minimum effective dosage to reduce the possibility of side effects. An improvement in nasal symptoms may be noted in patients within 10 hours of first using budesonide nasal spray, however, clinical improvement usually takes 1-2 days with maximum benefit in approximately 2 weeks. When the maximum benefit has been achieved and symptoms have been controlled, reducing the dosage may be effective in maintaining control of the allergic rhinitis symptoms in patients who were initially controlled on higher dosages.
Prior to initial use, the container must be shaken gently and the pump must be primed by actuating eight times. If used daily, the pump does not need to be reprimed. If not used for two consecutive days, reprime with one spray or until a fine spray appears. If not used for more than 14 days, rinse the applicator and reprime with two sprays or until a fine spray appears. Shake the container gently before each use.
Illustrated Patient’s Instructions for Use accompany each package of budesonide nasal spray 32 mcg.
- 3 DOSAGE FORMS AND STRENGTHS
-
4 CONTRAINDICATIONS
Budesonide nasal spray is contraindicated in patients with hypersensitivity to any of its ingredients [see Warnings and Precautions (5.2)].
-
5 WARNINGS AND PRECAUTIONS
5.1 Local Nasal Effects
Epistaxis
In clinical studies of 3 to 52 weeks’ duration epistaxis was observed more frequently in patients treated with budesonide nasal spray than those who received placebo [see Adverse Reactions (6.1)].
Candida Infection
In clinical studies with budesonide administered intranasally, the development of localized infections of the nose and pharynx with Candida albicans has occurred. When such an infection develops, it may require treatment with appropriate local or systemic therapy and discontinuation of treatment with budesonide nasal spray. Patients using budesonide nasal spray over several months or longer should be examined periodically for evidence of Candida infection or other signs of adverse effects on the nasal mucosa.
Nasal Septal Perforation
Instances of nasal septum perforation have been reported following the intranasal application of corticosteroids, including budesonide [see Adverse Reactions (6.2) ].
Impaired Wound Healing
Because of the inhibitory effect of corticosteroids on wound healing, patients who have experienced recent nasal septal ulcers, nasal surgery, or nasal trauma should not use a nasal corticosteroid until healing has occurred.
5.2 Hypersensitivity Reactions Including Anaphylaxis
Hypersensitivity reactions including anaphylactic reaction, urticaria, rash, dermatitis, angioedema and pruritus may occur [see Contraindications (4) and Adverse Reactions, Post-marketing Experience (6.2)].
5.3 Immunosuppression
Patients who are on drugs that suppress the immune system are more susceptible to infections than healthy individuals. Chicken pox and measles, for example, can have a more serious or even fatal course in susceptible children or adults using corticosteroids. In such children or adults who have not had these diseases or been properly immunized, particular care should be taken to avoid exposure. How the dose, route, and duration of corticosteroid administration affect the risk of developing a disseminated infection is not known. The contribution of the underlying disease and/or prior corticosteroid treatment to the risk is also not known. If exposed to chicken pox, therapy with varicella zoster immune globulin (VZIG) or pooled intravenous immunoglobulin (IVIG), as appropriate, may be indicated. If exposed to measles, prophylaxis with pooled intramuscular immunoglobulin (IG) may be indicated. (See the respective package inserts for complete VZIG and IG prescribing information). If chicken pox develops, treatment with antiviral agents may be considered.
The clinical course of chicken pox or measles infection in patients on intranasal or inhaled corticosteroids has not been studied. While there is no data with intranasal corticosteroids, a clinical study has examined the immune responsiveness to the varicella vaccine in asthma patients 12 months to 8 years of age who were treated with budesonide inhalation suspension.
An open-label, nonrandomized clinical study examined the immune responsiveness to varicella vaccine in 243 asthma patients 12 months to 8 years of age who were treated with budesonide inhalation suspension 0.25 mg to 1 mg daily (n=151) or non-corticosteroid asthma therapy (n=92) (i.e., beta2-agonists, leukotriene receptor antagonists, or cromones). The percentage of patients developing a seroprotective antibody titer ≥ 5.0 (gpELISA value) in response to the vaccination was similar in patients treated with budesonide inhalation suspension (85%) compared to patients treated with non-corticosteroids asthma therapy (90%). No patient treated with budesonide inhalation suspension developed chicken pox as a result of vaccination.
Corticosteroids should be used with caution, if at all, in patients with active or quiescent tuberculosis infection, untreated fungal, bacterial, systemic viral or parasitic infections; or ocular herpes simplex.
5.4 Hypothalamic-Pituitary-Adrenal Axis Effects
Hypercorticism and Adrenal Suppression: When intranasal steroids are used at higher than recommended dosages or in susceptible individuals at recommended dosages, systemic corticosteroid effects such as hypercorticism and adrenal suppression may appear. If such changes occur, the dosage of budesonide nasal spray should be discontinued slowly, consistent with accepted procedures for discontinuing oral corticosteroid therapy.
The replacement of a systemic corticosteroid with a topical corticosteroid can be accompanied by signs of adrenal insufficiency, and in addition some patients may experience symptoms of corticosteroid withdrawal, e.g., joint and/or muscular pain, fatigue, weakness, nausea, vomiting, hypotension, lassitude, and depression. Patients previously treated for prolonged periods with systemic corticosteroids should be weaned off slowly when transferred to topical corticosteroids and carefully monitored for acute adrenal insufficiency in response to stress. In those patients who have asthma or other clinical conditions requiring long-term systemic corticosteroid treatment, too rapid a decrease in systemic corticosteroids may cause a severe exacerbation of their symptoms.
5.5 Interactions with Strong Cytochrome P450 3A4 Inhibitors
Caution should be exercised when considering the co-administration of budesonide nasal spray with ketoconazole and other known strong CYP3A4 inhibitors (e.g., ritonavir, atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, saquinavir, telithromycin) because adverse effects related to increased systemic exposure to budesonide may occur [see Drug Interactions (7.1), Clinical Pharmacology (12.3)].
5.6 Effect on Growth
Intranasal corticosteroids, including budesonide may cause a reduction in growth velocity when administered to pediatric patients. Monitor the growth routinely of pediatric patients receiving long-term treatment with budesonide nasal spray. To minimize the systemic effects of intranasal corticosteroids, including budesonide nasal spray, titrate each patient’s dosage to the lowest one that effectively controls his/her symptoms [see Use in Specific Populations, Pediatric Use (8.4) ].
5.7 Glaucoma and Cataracts
Glaucoma, increased intraocular pressure and cataracts have been reported following the intranasal application of corticosteroids, including budesonide. Therefore, close monitoring is warranted in patients with a change in vision or with a history of increased intraocular pressure, glaucoma, and/or cataracts [see Adverse Reactions (6.2)].
-
6 ADVERSE REACTIONS
Systemic and intranasal corticosteroids use may result in the following:
- Epistaxis, Candida albicans infection, nasal septum perforation, and impaired wound healing [see Warnings and Precautions (5.1)1].
- Hypersensitivity Including Anaphylaxis [see Warnings and Precautions (5.2)].
- Immunosuppression [see Warnings and Precautions (5.3) ].
- Hypercorticism and Adrenal Suppression [see Warnings and Precautions (5.4) ].
- Growth Effect [see Warnings and Precautions (5.6) and Use in Specific Populations (8.4)].
- Glaucoma and Cataracts [see Warnings and Precautions (5.7)]
- 1
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The incidence of common adverse reactions in Table 1 is based upon two U.S. and five non-U.S. controlled clinical trials in 1,526 patients with seasonal or perennial rhinitis in adults and children ≥ 6 years treated with budesonide nasal spray at doses up to 400 mcg once daily for 3-6 weeks. This population included 745 females and 781 males with a mean age of 31 years (range of 6 - 85 years, 349 were 6 < 18 years). The racial distribution of patients receiving budesonide nasal spray was 93% white, 3% black and 4% other. Table 1 describes adverse reactions occurring at an incidence of 2% or greater and more commonly among budesonide nasal spray-treated patients than in placebo-treated patients in controlled clinical trials.
Table 1. Adverse Reactions occurring at an incidence ≥ 2% and more commonly than placebo in the budesonide nasal spray group in patients 6 years and older Adverse Event Budesonide Nasal Spray Placebo Vehicle Epistaxis
8%
5%
Pharyngitis
4%
3%
Bronchospasm
2%
1%
Coughing
2%
<1%
Nasal Irritation
2%
<1%
A similar adverse reaction profile was observed in the subgroup of pediatric patients 6 to 12 years of age. These patients are included in Table 1.
Two to three percent (2-3%) of patients in clinical trials discontinued because of adverse reactions. Systemic corticosteroid side effects were not reported during controlled clinical studies with budesonide nasal spray.
If recommended doses are exceeded, or if individuals are particularly sensitive, symptoms of hypercorticism, i.e., Cushing’s Syndrome, and adrenal suppression could occur.
6.2 Post-marketing Experience
The following adverse reactions have been reported during post-approval use of budesonide nasal spray. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Immune system disorders: immediate and delayed hypersensitivity reactions (including anaphylactic reaction, urticaria, rash, dermatitis, angioedema and pruritus), [see Warnings and Precautions (5.2) and Contraindications (4)]
Eye disorders: glaucoma, increased intraocular pressure, cataracts [see Warnings and Precautions (5.7)]
Respiratory, thoracic, and mediastinal disorders: nasal septum perforation, anosmia, pharynx disorders (throat irritation, throat pain, swollen throat, burning throat, and itchy throat), and wheezing
Cardiac disorders: palpitations
Musculoskeletal and connective tissue disorders: growth suppression [see Warnings and Precautions (5.6) and Use in Specific Populations (8.4) ]
-
7 DRUG INTERACTIONS
7.1 Inhibitors of Cytochrome P450 3A4
The main route of metabolism of corticosteroids, including budesonide, is via cytochrome P450 (CYP) isoenzyme 3A4 (CYP3A4). After oral administration of ketoconazole, a strong inhibitor of CYP3A4, the mean plasma concentration of orally administered budesonide increased. Concomitant administration of CYP3A4 may inhibit the metabolism of, and increase the systemic exposure to, budesonide. Caution should be exercised when considering the co-administration of budesonide nasal spray with long-term ketoconazole and other known strong CYP3A4 inhibitors (e.g., ritonavir, atazanavir, clarithromycin, indinavir, itraconazole, nefazodone, nelfinavir, saquinavir, telithromycin) [see Warnings and Precautions (5.6) and Clinical Pharmacology (12.3)].
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Teratogenic Effects: Pregnancy Category B. The impact of budesonide on human pregnancy outcomes has been evaluated through assessments of birth registries linked with maternal usage of inhaled budesonide (i.e., PULMICORT TURBUHALER) and intranasally administered budesonide (i.e., Budesonide Nasal Spray). The results from population-based prospective cohort epidemiological studies reviewing data from three Swedish registries covering approximately 99% of the pregnancies from 1995- 2001 (i.e., Swedish Medical Birth Registry; Registry of Congenital Malformations; Child Cardiology Registry) indicate no increased risk for overall congenital malformations from the use of inhaled or intranasal budesonide during early pregnancy.
Congenital malformations were studied in 2,014 infants born to mothers reporting the use of inhaled budesonide for asthma in early pregnancy (usually 10-12 weeks after the last menstrual period), the period when most major organ malformations occur.1 The rate of overall congenital malformations was similar compared to the general population rate (3.8 % vs. 3.5%, respectively). The number of infants born with orofacial clefts and cardiac defects was similar to the expected number in the general population (4 children vs. 3.3 and 18 children vs. 17-18, respectively). In a follow-on study bringing the total number of infants to 2,534, the rate of overall congenital malformations among infants whose mothers were exposed to inhaled budesonide during early pregnancy was not different from the rate for all newborn babies during the same period (3.6%).2 A third study from the Swedish Medical Birth Registry of 2,968 pregnancies exposed to inhaled budesonide, the majority of which were first trimester exposures, reported gestational age, birth weight, birth length, stillbirths, and multiple births similar for exposed infants compared to nonexposed infants.3
Congenital malformations were studied in 2,113 infants born to mothers reporting the use of intranasal budesonide in early pregnancy. The rate of overall congenital malformations was similar compared to the general population rate (4.5% vs. 3.5%, respectively). The adjusted odds ratio (OR) was 1.06 (95% CI 0.86-1.31). The number of infants born with orofacial clefts was similar to the expected number in the general population (3 children vs. 3, respectively). The number of infants born with cardiac defects exceeded that expected in the general population (28 children vs. 17.8 respectively). The systemic exposure from intranasal budesonide is 6-fold less than from inhaled budesonide and an association of cardiac defects was not seen with higher exposures of budesonide.
Despite the animal findings, it would appear that the possibility of fetal harm is remote if the drug is used during pregnancy. Nevertheless, because the studies in humans cannot rule out the possibility of harm, budesonide nasal spray should be used during pregnancy only if clearly needed.
Budesonide produced fetal loss, decreased pup weights, and skeletal abnormalities at a subcutaneous dose in rabbits that was approximately 2 times the maximum recommended daily intranasal dose in adults on a mcg/m2 basis and at a subcutaneous dose in rats that was approximately 16 times the maximum recommended daily intranasal dose in adults on a mcg/m2 basis. No teratogenic or embryocidal effects were observed in rats when budesonide was administered by inhalation at doses up to approximately 8 times the maximum recommended daily intranasal dose in adults on a mcg/m2 basis.
Experience with oral corticosteroids since their introduction in pharmacologic, as opposed to physiologic doses suggests that rodents are more prone to teratogenic effects from corticosteroids than humans.
Nonteratogenic Effects: Hypoadrenalism may occur in infants born of mothers receiving corticosteroids during pregnancy. Such infants should be carefully observed.
8.3 Nursing Mothers
Budesonide is secreted in human milk. Data with budesonide delivered via dry powder inhaler indicates that the total daily oral dose of budesonide available in breast milk to the infant is approximately 0.3% to 1% of the dose inhaled by the mother [see Clinical Pharmacology, Pharmacokinetics, Special Populations, Nursing (12.3)]. No studies have been conducted in breastfeeding women specifically with budesonide nasal spray; however, the dose of budesonide available to the infant in breast milk, as a percentage of the maternal dose, would be expected to be similar. Budesonide nasal spray should be used in nursing women only if clinically appropriate. Prescribers should weigh the known benefits of breastfeeding for the mother and infant against the potential risks of minimal budesonide exposure in the infant. Dosing considerations include prescription or titration to the lowest clinically effective dose and use of budesonide nasal spray immediately after breastfeeding to maximize the time interval between dosing and breastfeeding to minimize infant exposure.
8.4 Pediatric Use
Safety and effectiveness in pediatric patients below 6 years of age have not been established.
Controlled clinical studies have shown that intranasal corticosteroids may cause a reduction in growth velocity in pediatric patients. This effect has been observed in the absence of laboratory evidence of hypothalamic-pituitary-adrenal (HPA)-axis suppression, suggesting that growth velocity is a more sensitive indicator of systemic corticosteroid exposure in pediatric patients than some commonly used tests of HPA-axis function. The long-term effects of this reduction in growth velocity associated with intranasal corticosteroids, including the impact on final adult height, are unknown. The potential for "catch-up" growth following discontinuation of treatment with intranasal corticosteroids has not been adequately studied.
The growth of pediatric patients receiving intranasal corticosteroids, including budesonide nasal spray, should be monitored routinely (e.g., via stadiometry). The potential growth effects of prolonged treatment should be weighed against clinical benefits obtained and the risks and benefits associated with alternative therapies. To minimize the systemic effects of intranasal corticosteroids, including budesonide nasal spray, each patient should be titrated to the lowest dose that effectively controls his/her symptoms.
A one-year placebo-controlled clinical growth study was conducted in 229 pediatric patients (ages 4 through 8 years of age) to assess the effect of budesonide nasal spray (single-daily dose of 64 mcg, the recommended starting dose for children ages 6 years and above) on growth velocity. From a population of 141 patients receiving budesonide nasal spray and 67 receiving placebo, the point estimate for growth velocity with budesonide nasal spray was 0.25 cm/year lower than that noted with placebo (95% confidence interval ranging from 0.59 cm/year lower than placebo to 0.08 cm/year higher than placebo).
In a study of asthmatic children 5-12 years of age, those treated with budesonide administered via a dry powder inhaler 200 mcg twice daily (n=311) had a 1.1-centimeter (0.433 inch) reduction in growth compared with those receiving placebo (n=418) at the end of one year; the difference between these two treatment groups did not increase further over three years of additional treatment. By the end of four years, children treated with budesonide dry powder inhaler and children treated with placebo had similar growth velocities. Conclusions drawn from this study may be confounded by the unequal use of corticosteroids in the treatment groups and inclusion of data from patients attaining puberty during the course of the study.
The systemic effects of inhaled corticosteroids are related to the systemic exposure to such drugs. Pharmacokinetic studies have demonstrated that in both adults and children, systemic exposure to budesonide at the highest recommended doses of budesonide nasal spray would be expected to be no greater than exposure at the lowest recommended doses via a dry powder inhaler. Therefore, the systemic effects (HPA axis and growth) of budesonide delivered from budesonide nasal spray would be expected to be no greater than what is reported for inhaled budesonide when administered via the dry powder inhaler.
The potential for budesonide nasal spray to cause growth suppression in susceptible patients or when given at doses above 64 mcg daily cannot be ruled out. The recommended dosage range in patients 6 to 11 years of age is 64 to 128 mcg per day [see Dosage and Administration (2)].
8.5 Geriatric Use
Of the 2,461 patients in clinical studies of budesonide nasal spray, 5% were 60 years of age and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, except for an adverse reaction reporting frequency of epistaxis that increased with age. Further, other reported clinical experience has not identified any other differences in responses between elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
8.6 Hepatic Impairment
Formal pharmacokinetic studies using budesonide nasal spray have not been conducted in patients with hepatic impairment. However, since budesonide is predominantly cleared by hepatic metabolism, impairment of liver function may lead to accumulation of budesonide in plasma. Therefore, patients with hepatic disease should be closely monitored.
-
10 OVERDOSAGE
Acute overdosage with this dosage form is unlikely since one 120 spray bottle of budesonide nasal spray 32 mcg only contains approximately 5.4 mg of budesonide. Chronic overdosage may result in signs/symptoms of hypercorticism [see Warnings and Precautions (5.4)].
-
11 DESCRIPTION
Budesonide, the active ingredient of Budesonide Nasal Spray, is an anti-inflammatory synthetic corticosteroid.
It is designated chemically as (RS)-11-beta, 16-alpha, 17, 21-tetrahydroxypregna-1,4-diene-3,20-dione cyclic 16, 17-acetal with butyraldehyde.
Budesonide is provided as the mixture of two epimers (22R and 22S).
The empirical formula of budesonide is C25H34O6 and its molecular weight is 430.5.
Its structural formula is:
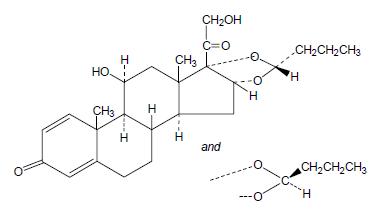
Budesonide is a white or almost white, crystalline powder that is practically insoluble in water and in heptane, sparingly soluble in ethanol, and freely soluble in chloroform.
Its partition coefficient between octanol and water at pH 5 is 1.6 x 103.
Budesonide nasal spray is an unscented, metered-dose, manual-pump spray formulation containing a micronized suspension of budesonide in an aqueous medium. Microcrystalline cellulose and carboxymethyl cellulose sodium, dextrose anhydrous, polysorbate 80, disodium edetate, potassium sorbate, and purified water are contained in this medium; hydrochloric acid is added to adjust the pH to a target of 4.0 to 5.0.
Budesonide nasal spray delivers 32 mcg of budesonide per spray.
Each bottle of budesonide nasal spray 32 mcg contains 120 metered sprays after initial priming.
Prior to initial use, the container must be shaken gently and the pump must be primed by actuating eight times. If used daily, the pump does not need to be reprimed. If not used for two consecutive days, reprime with one spray or until a fine spray appears. If not used for more than 14 days, rinse the applicator and reprime with two sprays or until a fine spray appears.
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Budesonide is an anti-inflammatory corticosteroid that exhibits potent glucocorticoid activity and weak mineralocorticoid activity. In standard in vitro and animal models, budesonide has approximately a 200-fold higher affinity for the glucocorticoid receptor and a 1000-fold higher topical anti-inflammatory potency than cortisol (rat croton oil ear edema assay). As a measure of systemic activity, budesonide is 40 times more potent than cortisol when administered subcutaneously and 25 times more potent when administered orally in the rat thymus involution assay. The clinical significance of this is unknown.
The activity of budesonide nasal spray is due to the parent drug, budesonide. In glucocorticoid receptor affinity studies, the 22R form was two times as active as the 22S epimer. In vitro studies indicated that the two forms of budesonide do not interconvert.
The precise mechanism of corticosteroid actions on inflammation in seasonal and perennial allergic rhinitis is not well known. Inflammation is an important component in the pathogenesis of seasonal and perennial allergic rhinitis. Corticosteroids have a wide range of inhibitory activities against multiple cell types (e.g., mast cells, eosinophils, neutrophils, macrophages, and lymphocytes) and mediators (e.g., histamine, eicosanoids, leukotrienes, and cytokines) involved in allergic and non-allergic-mediated inflammation. These anti-inflammatory actions of corticosteroids may contribute to their efficacy in seasonal and perennial allergic rhinitis.
12.2 Pharmacodynamics
A 3-week clinical study in seasonal rhinitis, comparing budesonide nasal inhaler, orally ingested budesonide, and placebo in 98 patients with allergic rhinitis due to birch pollen, demonstrated that the therapeutic effect of budesonide nasal inhaler can be attributed to the topical effects of budesonide.
HPA Axis Effects:
The effects of budesonide nasal spray on adrenal function have been evaluated in several clinical trials. In a four-week clinical trial, 61 adult patients who received 256 mcg daily of budesonide nasal spray demonstrated no significant differences from patients receiving placebo in plasma cortisol levels measured before and 60 minutes after 0.25 mg intramuscular cosyntropin. There were no consistent differences in 24-hour urinary cortisol measurements in patients receiving up to 400 mcg daily. Similar results were seen in a study of 150 children and adolescents aged 6 to 17 with perennial rhinitis who were treated with 256 mcg daily for up to 12 months. After treatment with the recommended maximal daily dose of budesonide nasal spray (256 mcg) for seven days, there was a small, but statistically significant decrease in the area under the plasma cortisol-time curve over 24 hours (AUC0-24h) in healthy adult volunteers.
A dose-related suppression of 24-hour urinary cortisol excretion was observed after administration of budesonide nasal spray doses ranging from 100-800 mcg daily for up to four days in 78 healthy adult volunteers. The clinical relevance of these results is unknown.
12.3 Pharmacokinetics
Absorption
After intranasal administration of a single dose of budesonide nasal spray (128 mcg), the mean peak plasma concentration of approximately 0.3 nmol/L occurs about 0.5 hours post-dose. Compared to an intravenous dose, approximately 34% of the delivered intranasal dose reaches the systemic circulation, most of which is absorbed through the nasal mucosa. While budesonide is well absorbed from the GI tract, the oral bioavailability of budesonide is low (~10%) primarily due to extensive first pass metabolism in the liver.
Distribution
The volume of distribution of budesonide was approximately 2-3 L/kg. It was 85-90% bound to plasma proteins. The volume of distribution for the 22R epimer is almost twice that of the 22S epimer. Protein binding was constant over a concentration range (1-100 nmol/L) achieved with, and exceeding, recommended doses of budesonide nasal spray. Budesonide showed little or no binding to corticosteroid binding globulin. Budesonide rapidly equilibrated with red blood cells in a concentration independent manner with a blood/plasma ratio of about 0.8.
Metabolism
In vitro studies with human liver homogenates have shown that budesonide is rapidly and extensively metabolized. Two major metabolites formed via cytochrome P450 (CYP) isoenzyme 3A4 (CYP3A4)-catalyzed biotransformation have been isolated and identified as 16α-hydroxyprednisolone and 6β-hydroxybudesonide. The corticosteroid activity of each of these two metabolites is less than 1% of that of the parent compound. No qualitative difference between the in vitro and in vivo metabolic patterns have been detected. Negligible metabolic inactivation was observed in human lung and serum preparations.
Excretion/Elimination
The 22R form of budesonide was preferentially cleared by the liver with systemic clearance of 1.4 L/min vs. 1.0 L/min for the 22S form. The terminal half-life, 2 to 3 hours, was the same for both epimers and was independent of dose. Budesonide was excreted in urine and feces in the form of metabolites. Approximately 2/3 of an intranasal radiolabeled dose was recovered in the urine and the remainder in the feces. No unchanged budesonide was detected in the urine.
Specific Populations
Geriatric
The pharmacokinetics of budesonide nasal spray in geriatric patients have not been specifically studied.
Pediatric
Following administration of budesonide nasal spray, the time to reach peak drug concentrations and plasma half-life were similar in children and in adults. Children had plasma concentrations approximately twice those observed in adults due primarily to differences in weight between children and adults.
Gender
No specific pharmacokinetic study has been conducted to evaluate the effect of gender on budesonide pharmacokinetics. However, following administration of 400 mcg of budesonide nasal spray to 7 male and 8 female volunteers in a pharmacokinetic study, no major gender differences in the pharmacokinetic parameters were found.
Race
No specific study has been undertaken to evaluate the effect of race on budesonide pharmacokinetics.
Nursing Mothers
The disposition of budesonide when delivered by oral inhalation from a dry powder inhaler at doses of 200 or 400 mcg twice daily for at least 3 months was studied in eight lactating women with asthma from 1 to 6 months postpartum. Systemic exposure to budesonide in these women appears to be comparable to that in non-lactating women with asthma from other studies. Breast milk obtained over eight hours post-dose revealed that the maximum concentration of budesonide for the 400 and 800 mcg doses was 0.39 and 0.78 nmol/L, respectively, and occurred within 45 minutes after dosing. The estimated oral daily dose of budesonide from breast milk to the infant was approximately 0.007 and 0.014 mcg/kg/day for the two dose regimens used in this study, which represents approximately 0.3% to 1% of the dose inhaled by the mother. Budesonide levels in plasma samples obtained from five infants at about 90 minutes after breastfeeding (and about 140 minutes after drug administration to the mother) were below quantifiable levels (<0.02 nmol/L in four infants and <0.04 nmol/L in one infant) [see Use in Specific Populations, Nursing Mothers (8.3)].
Renal or Hepatic Impairment
The pharmacokinetics of budesonide have not been investigated in patients with renal impairment. Reduced liver function may affect the elimination of corticosteroids. The pharmacokinetics of budesonide were affected by compromised liver function as evidenced by a doubled systemic availability after oral ingestion. The relevance of this finding to intranasally administered budesonide has not been established.
Drug-Drug Interactions
Inhibitors of cytochrome P450 enzymes
Ketoconazole: Ketoconazole, a strong inhibitor of cytochrome P450 (CYP) isoenzyme 3A4 (CYP3A4), the main metabolic enzyme for corticosteroids, increased plasma levels of orally ingested budesonide [see Warnings and Precautions (5.5) and Drug Interactions (7.1)].
Cimetidine: At recommended doses, cimetidine, a non-specific inhibitor of CYP enzymes, had a slight but clinically insignificant effect on the pharmacokinetics of oral budesonide.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 104-week oral study in Sprague-Dawley rats, a statistically significant increase in the incidence of gliomas was observed in the male rats receiving an oral dose of budesonide 50 mcg/kg/day (approximately twice the maximum recommended daily intranasal dose in adults and children on a mcg/m2 basis). No tumorigenicity was seen in male rats at oral doses up to 25 mcg/kg (approximately equal to the maximum recommended daily intranasal dose in adults and children on a mcg/m2 basis, and in female rats at oral doses up to 50 mcg/kg approximately two times the maximum recommended daily intranasal dose in adults and children on a mcg/m2 basis). In two additional two-year studies in male Fischer and Sprague-Dawley rats, budesonide caused no gliomas at an oral dose of 50 mcg/kg (approximately twice the maximum recommended daily intranasal dose in adults and children on a mcg/m2 basis). However, in the male Sprague-Dawley rats, budesonide caused a statistically significant increase in the incidence of hepatocellular tumors at an oral dose of 50 mcg/kg (approximately twice the maximum recommended daily intranasal dose in adults and children on a mcg/m2 basis). The concurrent reference corticosteroids (prednisolone and triamcinolone acetonide) in these two studies showed similar findings.
There was no evidence of a carcinogenic effect when budesonide was administered orally for 91-weeks to mice at doses up to 200 mcg/kg/day (approximately 3 times the maximum recommended daily intranasal dose in adults and children on a mcg/m2 basis).
Budesonide was not mutagenic or clastogenic in six different test systems: Ames Salmonella/microsome plate test, mouse micronucleus test, mouse lymphoma test, chromosome aberration test in human lymphocytes, sex-linked recessive lethal test in Drosophila melanogaster, and DNA repair analysis in rat hepatocyte culture.
In rats, budesonide had no effect on fertility at subcutaneous doses up to 80 mcg/kg (approximately 3 times the maximum recommended daily intranasal dose in adults on mcg/m2 basis).
At a subcutaneous dose of 20 mcg/kg/day (less than the maximum recommended daily intranasal dose in adults on a mcg/m2 basis), decreases in maternal body weight gain, prenatal viability, and viability of the young at birth and during lactation were observed. No such effects were noted at 5 mcg/kg (less than the maximum recommended daily intranasal dose in adults on a mcg/m2 basis).
13.2 Animal Toxicology and/or Pharmacology
Budesonide was teratogenic and embryocidal in rabbits and rats. Budesonide produced fetal loss, decreased pup weights, and skeletal abnormalities at a subcutaneous dose of 25 mcg/kg in rabbits (approximately 2 times the maximum recommended daily intranasal dose in adults on a mcg/m2 basis) and at a subcutaneous dose of 500 mcg/kg in rats (approximately 16 times the maximum recommended daily intranasal dose in adults on a mcg/m2 basis). No teratogenic or embryocidal effects were observed in rats when budesonide was administered by inhalation doses up to 250 mcg/kg (approximately 8 times the maximum recommended daily intranasal dose in adults on a mcg/m2 basis).
-
14 CLINICAL STUDIES
The therapeutic efficacy of budesonide nasal spray has been evaluated in placebo-controlled clinical trials of seasonal and perennial allergic rhinitis of 3-6 weeks duration.
The number of patients treated with budesonide in these studies was 90 males and 51 females aged 6-12 years and 691 males and 694 females 12 years and above. The patients were predominantly Caucasian.
Overall, the results of these clinical trials showed that budesonide nasal spray administered once daily provides statistically significant reduction in the severity of nasal symptoms of seasonal and perennial allergic rhinitis including runny nose, sneezing, and nasal congestion.
An improvement in nasal symptoms may be noted in patients within 10 hours of first using budesonide nasal spray. This time to onset is supported by an environmental exposure unit study in seasonal allergic rhinitis patients that demonstrated that budesonide nasal spray led to a statistically significant improvement in nasal symptoms compared to placebo by 10 hours. Further support comes from a clinical study of patients with perennial allergic rhinitis which demonstrated a statistically significant improvement in nasal symptoms for both budesonide nasal spray and for the active comparator (mometasone furoate) compared to placebo by 8 hours. Onset was also assessed in this study with peak nasal inspiratory flow rate and this endpoint failed to show efficacy for either active treatment. Although statistically significant improvements in nasal symptoms compared to placebo were noted within 8-10 hours in these studies, about one half to two thirds of the ultimate clinical improvement with budesonide nasal spray occurs over the first 1-2 days, and maximum benefit may not be achieved until approximately 2 weeks after initiation of treatment.
-
15 REFERENCES
1 Kallen B, Rydhstroem H, Aberg A. Congenital malformations after the use of inhaled budesonide in early pregnancy. Obstet Gynecol 1999; 93:392-395.
2 Ericson A, Kallen B. Use of drugs during pregnancy: unique Swedish registration method that can be improved. Swedish Medical Products Agency 1999;1:8-11.
3 Norjavaara E, Gerhardsson de Verdier M. Normal pregnancy outcomes in a population-based study including 2968 pregnant women exposed to budesonide. J Allergy Clin Immunol 2003;111:736-742.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Budesonide Nasal Spray, 32 mcg is available in an amber glass bottle with a metered-dose pump spray and a protection cap. Budesonide Nasal Spray, 32 mcg (NDC: 60505-0839-2) provides 120 metered sprays after initial priming; net fill weight 8.6 g. The Budesonide Nasal Spray, 32 mcg bottle has been filled with an excess to accommodate the priming activity. The bottle should be discarded after 120 sprays following initial priming, since the amount of budesonide delivered per spray thereafter may be substantially less than the labeled dose. Each spray delivers 32 mcg of budesonide to the patient.
Budesonide Nasal Spray should be stored upright at 20º to 25°C (68º to 77°F) [See USP controlled room temperature]. Do not freeze. Protect from light.
Shake gently before use. Do not spray in eyes.
-
17 PATIENT COUNSELING INFORMATION
[See FDA-Approved Patient Labeling]
Patients being treated with budesonide nasal spray should receive the following information and instructions. This information is intended to aid the patient in the safe and effective use of the medication. It is not a disclosure of all possible adverse or intended effects. For proper use of budesonide nasal spray and to attain maximum improvement, the patient should read and follow the accompanying FDA Approved Patient Labeling.
17.1 Local Nasal Effects
Patients should be advised that epistaxis and localized infections with Candida albicans occurred in the nose and pharynx in some patients. If candidiasis develops, it should be treated with appropriate local or systemic therapy and discontinue treatment with budesonide nasal spray. In addition, nasal corticosteroids are associated with nasal septal perforation and impaired wound healing. Patients who have experienced recent nasal ulcers, nasal surgery, or nasal trauma should not use budesonide nasal spray until healing has occurred [ see Warnings and Precautions (5.1)].
17.2 Hypersensitivity including Anaphylaxis
Patients should be advised that hypersensitivity reactions including anaphylactic reaction, urticaria, rash, dermatitis, angioedema and pruritus have been reported with use of budesonide nasal spray. Discontinue budesonide nasal spray if such reactions occur [see Contraindications (4),Warnings and Precautions (5.2) and Adverse Reactions (6)].
17.3 Immunosuppression
Patients who are on immunosuppressant doses of corticosteroids should be warned to avoid exposure to chickenpox or measles and, if exposed, to consult their physician without delay. Patients should be informed of potential worsening of existing tuberculosis, fungal, bacterial, viral, or parasitic infection, or ocular herpes simplex [see Warnings and Precautions (5.3)].
17.4 Reduced Growth Velocity
Patients should be advised that intranasal corticosteroids, including budesonide, may cause a reduction in growth velocity when administered to pediatric patients. Physicians should closely follow the growth of children and adolescents taking corticosteroids by any route [see Warnings and Precautions (5.7)].
17.5 Glaucoma and Cataracts
Patients should be advised that long-term use of intranasal corticosteroids, including budesonide, may increase the risk of some eye problems (cataracts and glaucoma). Patients should inform his/her healthcare provider if a change in vision is noted while using budesonide nasal spray [see Warnings and Precautions (5.7)].
17.6 Use Daily
Patients should use budesonide nasal spray at regular intervals since its effectiveness depends on its regular use. Patients may note an improvement in nasal symptoms within 10 hours of first using budesonide nasal spray. Maximum benefit may not be achieved until approximately 2 week after initiation of treatment [see Dosage and Administration (2)].
Patients should take the medication as directed and should not exceed the prescribed dosage. The patient should contact the physician if symptoms do not improve after two weeks, or if the condition worsens. Patients who experience recurrent episodes of epistaxis (nosebleeds) or nasal septum discomfort while taking this medication should contact their physician. For proper use of budesonide nasal spray and to attain maximum improvement, the patient should read and follow the accompanying patient information carefully. Do not use budesonide nasal spray after the labeled number of sprays have been used (does not include priming) or after the expiration date shown on the carton or bottle label.
17.7 How to Use Budesonide Nasal Spray
Patients should be carefully instructed on the use of this drug product to assure optimal dose delivery [see Patient Information].
Pulmicort Turbuhaler® is a registered trademark of AstraZeneca.
Manufactured by: Manufactured for: Apotex Inc. Apotex Corp. Toronto, Ontario Weston, Florida Canada 33326 M9L 1T9 285138 January 2011 -
Patient Information
Budesonide (byoo des’ oh nide) Nasal Spray, 32 mcg
For use in your nose only
Read the Patient Information that comes with budesonide nasal spray before you start using it and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or treatment. If you have questions about budesonide nasal spray, ask your healthcare provider or pharmacist.
What is budesonide nasal spray?
Budesonide nasal spray is a prescription medicine used to treat seasonal and year-round allergy symptoms in adults and children 6 years of age and older.
Budesonide nasal spray contains budesonide, which is a man-made (synthetic) corticosteroid. Intranasal corticosteroids are natural hormones found in the body that reduce swelling of the lining of your nose. When you spray budesonide nasal spray into your nose, it helps reduce the nasal symptoms of allergic rhinitis (inflammation of the lining of the nose), such as stuffy nose, runny nose, itching and sneezing.
The safety and effectiveness of budesonide nasal spray has not been shown in children under 6 years of age.
Who should not use budesonide nasal spray?
Do not use budesonide nasal spray:
- if you are allergic to budesonide or any of the ingredients in budesonide nasal spray. See the end of this leaflet for a complete list of the ingredients in budesonide nasal spray.
What should I tell my healthcare provider before using budesonide nasal spray?
Before you use budesonide nasal spray, tell your healthcare provider or pharmacist if you:
- have recently been around anyone with chicken pox or measles
- have liver problems
- have any untreated infections
- have ever had an infection called tuberculosis
- have an eye infection
- have recently had surgery or an injury to your nose
- have any other medical conditions
- are pregnant or plan to become pregnant. It is not known if budesonide nasal spray will harm your unborn baby. Talk to your doctor if you are pregnant or plan to become pregnant.
- are breastfeeding or plan to breastfeed. Budesonide nasal spray can pass into your breast milk. Talk to your doctor about the best way to feed your baby if you take budesonide nasal spray.
Tell your healthcare provider about all the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements.
Budesonide nasal spray may affect the way other medicines work, and other medicines may affect how budesonide nasal spray works.
Know the medicines you take. Keep a list of your medicines with you to show your healthcare provider and pharmacist when you get a new medicine.
How should I use budesonide nasal spray?
- Budesonide nasal spray is for use in your nose only. Do not spray it in your eyes or mouth.
- Use budesonide nasal spray exactly as your healthcare provider tells you to use it.
- It is very important that you use budesonide nasal spray regularly. Do not stop using budesonide nasal spray or change your dose without talking to your healthcare provider, even if you are feeling better.
- Talk to your healthcare provider if your symptoms do not improve after taking budesonide nasal spray for 2 weeks or if your symptoms get worse.
- An adult should help a young child use this medicine.
- See the Patient Instructions for Use at the end of this leaflet for complete information on how to use budesonide nasal spray.
What are the possible side effects of budesonide nasal spray?
Budesonide nasal spray may cause serious side effects including:
- hole in the cartilage inside the nose (nasal septal perforation). Tell your healthcare provider if you have a whistling sound from your nose when you breathe.
- slow wound healing. You should not use budesonide nasal spray until your nose has healed if you have a sore in your nose, if you have had surgery on your nose, or if your nose has been injured.
- fungal infection in your nose.
-
allergic reactions. Tell your healthcare provider or get medical help right away if you have:
- skin rash, redness or swelling
- severe itching
- swelling of the face, mouth and tongue
- immune system problems that may increase your risk of infections. You are more likely to get infections if you take medicines that weaken your body’s ability to fight infections. Avoid contact with people who have contagious diseases such as chicken pox or measles while using budesonide nasal spray. Symptoms of infection may include fever, pain, aches, chills, feeling tired, nausea and vomiting.
- adrenal insufficiency. Adrenal insufficiency is a condition in which the adrenal glands do not make enough steroid hormones. Symptoms of adrenal insufficiency may include tiredness, weakness, nausea, vomiting and low blood pressure.
- slowed or delayed growth in children. A child’s growth should be checked regularly while using budesonide nasal spray.
- eye problems, such as glaucoma and cataracts. Tell your healthcare provider if you have a change in vision or have a history of increased intraocular pressure, glaucoma, and/or cataracts.
Call your healthcare provider or get medical help right away if you have symptoms of any of the serious side effects listed above.
The most common side effects of budesonide nasal spray include:
- nose bleeds
- sore throat
- breathing difficulties such as wheezing, or chest tightening
- cough
- irritation of your nose
Tell your healthcare provider if you have any side effect that bothers you or does not go away.
These are not all of the possible side effects of budesonide nasal spray. For more information, ask your healthcare provider or pharmacist.
Call your healthcare provider for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
You may report side effects to Apotex Corp. at 1-800-667-4708.
What should I know about allergic rhinitis?
“Rhinitis” means inflammation of the lining of the nose. It is sometimes called “hay fever.” Allergic rhinitis can be caused by allergies to pollen, animal dander, house dust mite, and mold spores. If you have allergic rhinitis, your nose becomes stuffy, runny, and itchy. You may also sneeze a lot. You may have red, itchy, watery eyes; itchy throat; or blocked, itchy ears.
Budesonide nasal spray helps to relieve your nasal symptoms.
If you also have itchy, watery eyes, you should tell your healthcare provider. He or she can prescribe additional medication to treat these symptoms.
How should I store budesonide nasal spray?
- Store budesonide nasal spray at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature].
- Do not freeze budesonide nasal spray.
- Protect budesonide nasal spray from light.
- Do not use budesonide nasal spray after the labeled number of sprays have been used (does not include priming) or after the expiration date shown on the carton or bottle label.
- Keep the protective cap on budesonide nasal spray when not in use. (Please see Prior to Use on reverse side).
- Keep budesonide nasal spray and all medications out of the reach of children.
General Information about the safe and effective use of budesonide nasal spray:
Do not use budesonide nasal spray for a condition for which it was not prescribed. Do not give budesonide nasal spray to other people, even if they have the same symptoms that you have. It may harm them.
This Patient Information leaflet summarizes the most important information about budesonide nasal spray. If you would like more information, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for information about budesonide nasal spray that is written for health professionals.
For more information, please call Apotex Corp., Consumer Affairs at 1-800-706-5575 (Monday to Friday, 8:30 am – 5:00 pm Eastern Standard Time).
What are the Ingredients of budesonide nasal spray?
Active ingredient: budesonide
Inactive ingredients:Microcrystalline cellulose and carboxymethyl cellulose sodium, dextrose anhydrous, polysorbate 80, disodium edetate, potassium sorbate, purified water and hydrochloric acid.
Patient Instructions for Use
For use in your nose only. Do not spray in your eyes or mouth.
Read the Patient Instructions for Use carefully before you start to use budesonide nasal spray. If you have any questions, ask your healthcare provider.
How to prime your budesonide nasal spray
Before you use budesonide nasal spray, the bottle must be primed. To prime budesonide nasal spray:
- Pull to remove the protective cap off the nasal spray unit.
- Shake the bottle gently for a few seconds before each use.
- Hold the bottle firmly, as shown in Figure A, with your index and middle finger on either side of the spray tip and your thumb underneath the bottle.
- Activate the pump by quickly and firmly pressing down on the white collar while holding the base of the bottle with your thumb.
- Before your first use of budesonide nasal spray, shake the bottle gently. The pump must be primed by pressing down on the white collar 8 times. The pump is now ready to use. If used daily the pump does not need to be reprimed. If not used for 2 days in a row, reprime with 1 spray or until a fine spray appears. If not used for more than 14 days, rinse the spray tip of the pump using the cleaning steps listed at the end of this leaflet. After cleaning reprime with 2 sprays or until a fine spray appears.
Each bottle of budesonide nasal spray contains enough medicine for you to spray medicine from the bottle 120 times after the bottle is primed.
You should not use the bottle of budesonide nasal spray after 120 sprays. Additional sprays after 120 may not contain the right amount of medicine. You should keep track of the number of sprays you use from each bottle of budesonide nasal spray and throw away any remaining medicine that may be left in the bottle. Refill your prescription monthly.
How to use your budesonide nasal spray
Follow these instructions for daily use of budesonide nasal spray:
- Gently blow your nose to clear your nostrils, if necessary.
- Shake the bottle gently for a few seconds and remove the protective cap.
- Hold the bottle firmly with your index and middle finger on either side of the spray tip and your thumb underneath the bottle (See Figure A).
- Insert the spray tip into your nostril (the tip should not reach far into your nose). Close the other nostril with a finger and lean your head slightly forward so the spray will aim toward the back of your nose (See Figure B).
- For each spray, activate the pump by quickly and firmly pressing down on the white collar while holding the base of the bottle with your thumb. Breathe gently inward through the nostril.
- After spraying into your nostril, lean your head backward for a few seconds (Figure C).
- If a second spray is needed in the same nostril, repeat steps 3 through 6.
- Repeat steps 3 through 7 for your other nostril.
- Avoid blowing your nose for 15 minutes after you use budesonide nasal spray.
- Wipe the spray tip with a clean tissue (Figure D), and replace the protective cap. Store the bottle in an upright position.
How to clean your budesonide nasal spray
Rinse the protective cap and the spray tip regularly. To do this:
- Remove the protective cap and lift off the spray tip.
- Wash only the protective cap and the spray tip in warm water and rinse them in cold tap water
- Allow the protective cap and the spray tip to air-dry completely before reassembling the nasal spray.
- If the spray tip becomes blocked, it can be cleared by repeating Steps 1 through 3. Do not unblock the nasal applicator with a pin or other sharp object.
For additional information about Budesonide Nasal Spray, please call Apotex Corp., Consumer Affairs at 1-800-706-5575 (Monday to Friday, 8:30 am – 5:00 pm Eastern Standard Time).
Manufactured by: Manufactured for: Apotex Inc. Apotex Corp. Toronto, Ontario Weston, Florida Canada 33326 M9L 1T9 285138 January 2011 -
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 32 mcg Bottle Label
APOTEX CORP. NDC: 60505-0839-2
Budesonide Nasal Spray
32 mcg
Rx
120 Metered Sprays

-
PRINCIPAL DISPLAY PANEL
PRINCIPAL DISPLAY PANEL - 32 mcg Carton Label
APOTEX CORP. NDC: 60505-0839-2
Budesonide Nasal Spray
32 mcg
Rx
120 Metered Sprays

-
INGREDIENTS AND APPEARANCE
BUDESONIDE NASAL
budesonide spray, meteredProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 60505-0839 Route of Administration NASAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BUDESONIDE (UNII: Q3OKS62Q6X) (BUDESONIDE - UNII:Q3OKS62Q6X) BUDESONIDE 32 ug Inactive Ingredients Ingredient Name Strength MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED FORM (UNII: K679OBS311) ANHYDROUS DEXTROSE (UNII: 5SL0G7R0OK) POLYSORBATE 80 (UNII: 6OZP39ZG8H) EDETATE DISODIUM (UNII: 7FLD91C86K) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) WATER (UNII: 059QF0KO0R) HYDROCHLORIC ACID (UNII: QTT17582CB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 60505-0839-2 1 in 1 CARTON 05/12/2014 1 120 in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA078949 05/12/2014 Labeler - Apotex Corp. (845263701)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.