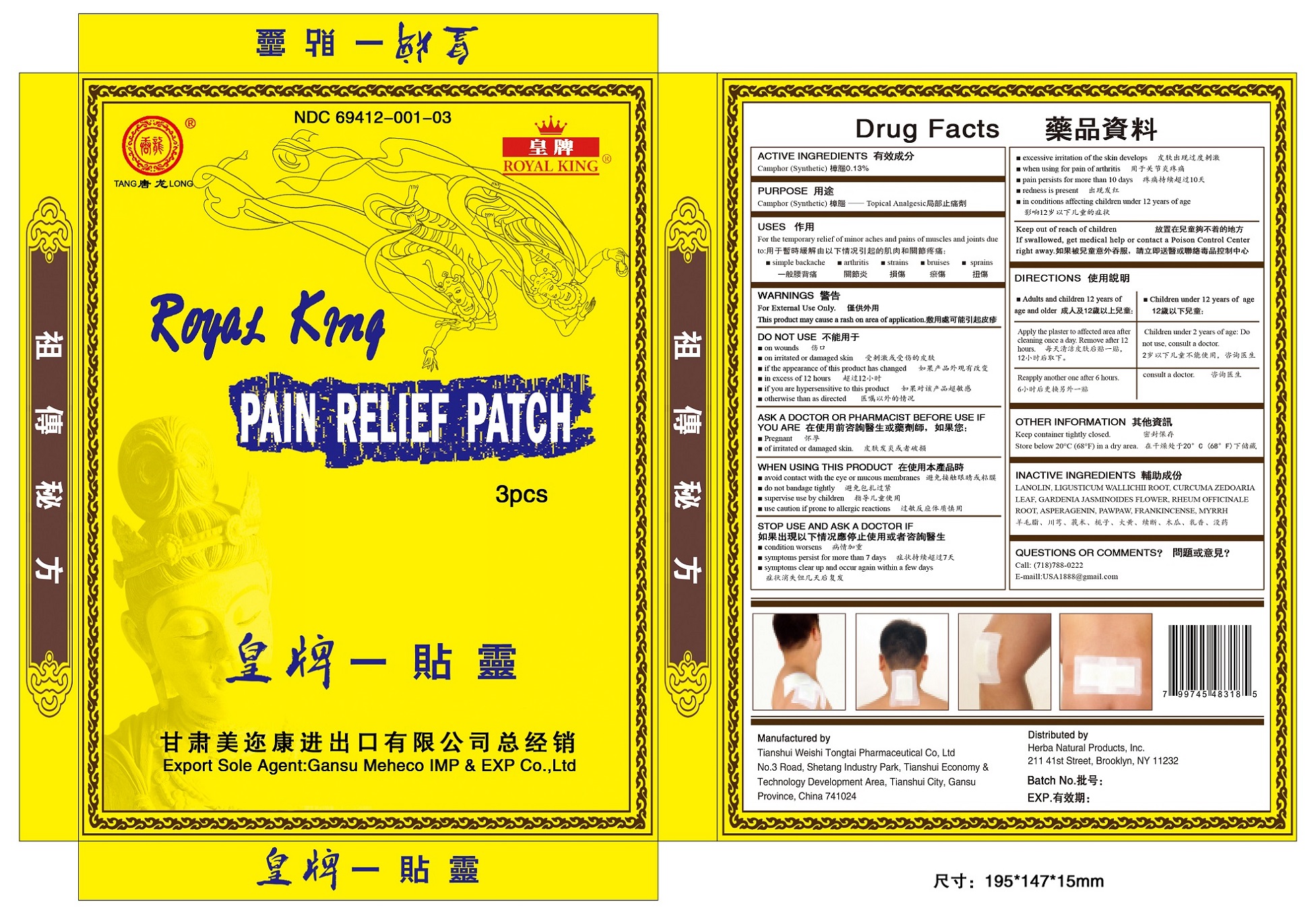
ROYAL KING PAIN RELIEF PATCH- camphor patch
Royal King Pain Relief Patch by
Drug Labeling and Warnings
Royal King Pain Relief Patch by is a Otc medication manufactured, distributed, or labeled by Tianshui Weishi Tongtai Pharmaceutical Co Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- ACTIVE INGREDIENTS
- PURPOSE
- USES
- WARNINGS
- DO NOT USE
- ASK A DOCTOR OR PHARMACIST BEFORE USE IF YOU ARE
- WHEN USING THIS PRODUCT
- STOP USE AND ASK A DOCTOR IF
- KEEP OUT OF REACH OF CHILDREN
- DIRECTIONS
- OTHER INFORMATION
- INACTIVE INGREDIENTS
- QUESTIONS OR COMMENTS
- DRUG FACTS
-
INGREDIENTS AND APPEARANCE
ROYAL KING PAIN RELIEF PATCH
camphor patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 69412-001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) (CAMPHOR (SYNTHETIC) - UNII:5TJD82A1ET) CAMPHOR (SYNTHETIC) 0.13 g in 100 g Inactive Ingredients Ingredient Name Strength LANOLIN (UNII: 7EV65EAW6H) LIGUSTICUM WALLICHII ROOT (UNII: R81AD159QS) CURCUMA ZEDOARIA LEAF (UNII: M9E9LQH7N4) GARDENIA JASMINOIDES FLOWER (UNII: 2TU49609L2) RHEUM OFFICINALE ROOT (UNII: A47JP5ZG9M) ASPERAGENIN (UNII: 56JLP9B53W) PAWPAW (UNII: 8P95FP65YO) FRANKINCENSE (UNII: R9XLF1R1WM) MYRRH (UNII: JC71GJ1F3L) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 69412-001-03 3 in 1 BOX 12/20/2016 1 7.9 g in 1 PATCH; Type 1: Convenience Kit of Co-Package Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 12/20/2016 Labeler - Tianshui Weishi Tongtai Pharmaceutical Co Ltd (544305099) Registrant - Tianshui Weishi Tongtai Pharmaceutical Co Ltd (544305099) Establishment Name Address ID/FEI Business Operations Tianshui Weishi Tongtai Pharmaceutical Co Ltd 544305099 manufacture(69412-001)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.