MUCUS RELIEF ER DM EXTENDED RELEASE- guaifenesin and dextromethorphan hydrobromide tablet, extended release
Mucus Relief ER DM by
Drug Labeling and Warnings
Mucus Relief ER DM by is a Otc medication manufactured, distributed, or labeled by KROGER COMPANY, RECKITT BENCKISER HEALTHCARE INTERNATIONAL LTD. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
-
Uses
- helps loosen phlegm (mucus) and thin bronchial secretions to rid the bronchial passageways of bothersome mucus and make coughs more productive
- temporarily relieves:
- cough due to minor throat and bronchial irritation as may occur with the common cold or inhaled irritants
- the intensity of coughing
- the impulse to cough to help you get to sleep
-
Warnings
Do not use
- for children under 12 years of age
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if you have
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis, or emphysema
- cough accompanied by too much phlegm (mucus)
- Directions
- Other information
- Inactive ingredients
- Questions?
- SPL UNCLASSIFIED SECTION
-
PRINCIPAL DISPLAY PANEL - 20 Tablet Blister Pack Carton
COMPARE TO the active ingredients of
MUCINEX® DM*
See back panelNDC: 30142-991-20
Kroger®
OUR PHARMACIST RECOMMENDED
Mucus Relief
ER DMGuaifenesin 600 mg &
Dextromethorphan HBr 30 mg
Extended-Release
TabletsEXPECTORANT &
COUGH SUPPRESSANT12
HOUR- Controls Cough
- Thins and Loosens Mucus
- Immediate & Extended Release
actual
size20 EXTENDED-RELEASE
TABLETS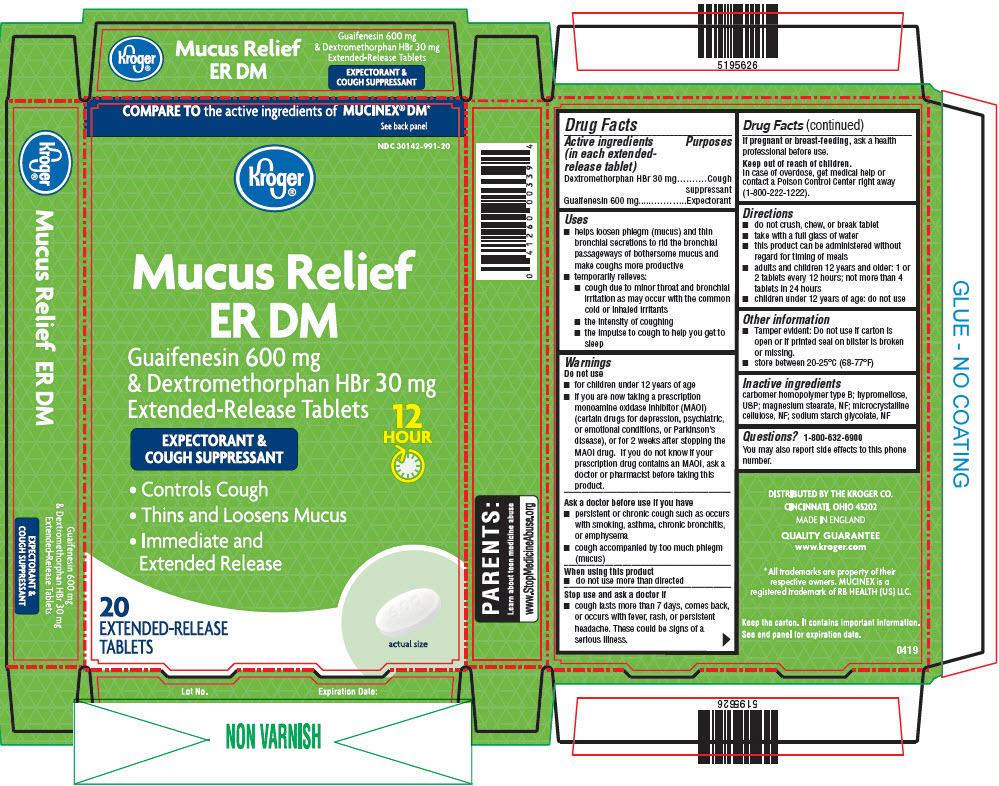
-
INGREDIENTS AND APPEARANCE
MUCUS RELIEF ER DM EXTENDED RELEASE
guaifenesin and dextromethorphan hydrobromide tablet, extended releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 30142-991 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Guaifenesin (UNII: 495W7451VQ) (Guaifenesin - UNII:495W7451VQ) Guaifenesin 600 mg Dextromethorphan Hydrobromide (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) Dextromethorphan Hydrobromide 30 mg Inactive Ingredients Ingredient Name Strength CARBOMER HOMOPOLYMER TYPE B (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: HHT01ZNK31) hypromellose, unspecified (UNII: 3NXW29V3WO) magnesium stearate (UNII: 70097M6I30) microcrystalline cellulose (UNII: OP1R32D61U) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) Product Characteristics Color WHITE Score no score Shape OVAL Size 16mm Flavor Imprint Code xeunciM;600 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 30142-991-20 1 in 1 CARTON 05/30/2019 1 20 in 1 BLISTER PACK; Type 0: Not a Combination Product 2 NDC: 30142-991-40 2 in 1 CARTON 05/30/2019 2 40 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA021620 05/30/2019 Labeler - KROGER COMPANY (006999528) Establishment Name Address ID/FEI Business Operations RECKITT BENCKISER HEALTHCARE INTERNATIONAL LTD 230780363 MANUFACTURE(30142-991) , LABEL(30142-991)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.