MESALAMINE tablet, delayed release
mesalamine by
Drug Labeling and Warnings
mesalamine by is a Prescription medication manufactured, distributed, or labeled by Northstar Rx LLC., Zydus Pharmaceuticals USA Inc., Zydus Lifesciences Limited. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use MESALAMINE DELAYED-RELEASE TABLETS safely and effectively. See full prescribing information for MESALAMINE DELAYED-RELEASE TABLETS .
MESALAMINE Delayed-release Tablets, for oral use
Initial U.S. Approval: 1987INDICATIONS AND USAGE
Mesalamine delayed-release tablets are locally acting 5-aminosalicylic acid (5-ASA) indicated for the induction of remission in adults with active, mild to moderate ulcerative colitis and for the maintenance of remission of ulcerative colitis. (1)
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
Delayed-Release Tablets: 1.2 g (3)
CONTRAINDICATIONS
Patients with known hypersensitivity to salicylates or aminosalicylates or to any of the ingredients of mesalamine delayed-release tablets. (4)
WARNINGS AND PRECAUTIONS
- Renal impairment may occur. Assess renal function at the beginning of treatment and periodically during treatment. (5.1)
- Mesalamine-induced acute intolerance syndrome has been reported. Observe patients closely for worsening of these symptoms while on treatment. (5.2)
- Use caution when treating patients who are hypersensitive to sulfasalazine. (5.3)
- Mesalamine-induced cardiac hypersensitivity reactions (myocarditis and pericarditis) have been reported. (5.3)
- Hepatic failure has been reported in patients with pre-existing liver disease. Use caution when treating patients with liver disease. (5.4)
- Upper GI tract obstruction may delay onset of action. (5.5)
- Patients with pre-existing skin conditions have reported more severe photosensitivity reactions. (5.6)
- Use of mesalamine may lead to spuriously elevated test results when measuring urinary normetanephrine by liquid chromatography with electrochemical detection. (5.7)
ADVERSE REACTIONS
- The most common adverse reactions (incidence ≥ 2 %) are ulcerative colitis, headache, flatulence, liver function test abnormality, and abdominal pain. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Zydus Pharmaceuticals (USA) Inc. at 1-877-993-8779 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 7/2019
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Renal Impairment
5.2 Mesalamine-Induced Acute Intolerance Syndrome
5.3 Hypersensitivity Reactions
5.4 Hepatic Impairment
5.5 Upper GI Tract Obstruction
5.6 Photosensitivity
5.7 Interference with Laboratory Tests
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Nephrotoxic Agents, Including Non-Steroidal Anti-Inflammatory Drugs
7.2 Azathioprine or 6-mercaptopurine
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.3 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 ANIMAL PHARMACOLOGY AND OR TOXICOLOGY
14 CLINICAL STUDIES
14.1 Active, Mild to Moderate Ulcerative Colitis
14.2 Maintenance of Remission in Patients with Ulcerative Colitis
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- * Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
-
2 DOSAGE AND ADMINISTRATION
The recommended dosage for the induction of remission in adult patients with active, mild to moderate ulcerative colitis is two to four 1.2 g tablets taken once daily with a meal for a total daily dose of 2.4 g or 4.8 g. The recommended dosage for the maintenance of remission is two 1.2 g tablets taken once daily with a meal for a total daily dose of 2.4 g.
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Renal Impairment
Renal impairment, including minimal change nephropathy, acute and chronic interstitial nephritis, and, rarely, renal failure, has been reported in patients given products such as mesalamine delayed-release tablets that contain mesalamine or are converted to mesalamine.
It is recommended that patients have an evaluation of renal function prior to initiation of mesalamine delayed-release tablets therapy and periodically while on therapy. Exercise caution when using mesalamine delayed-release tablets in patients with known renal dysfunction or a history of renal disease.
In animal studies, the kidney was the principal organ for toxicity [see Drug Interactions (7.1) and Nonclinical Toxicology (13.2 )].
5.2 Mesalamine-Induced Acute Intolerance Syndrome
Mesalamine has been associated with an acute intolerance syndrome that may be difficult to distinguish from an exacerbation of ulcerative colitis. Although the exact frequency of occurrence has not been determined, it has occurred in 3% of patients in controlled clinical trials of mesalamine or sulfasalazine. Symptoms include cramping, acute abdominal pain and bloody diarrhea, and sometimes fever, headache, and rash. Observe patients closely for worsening of these symptoms while on treatment. If acute intolerance syndrome is suspected, promptly discontinue treatment with mesalamine delayed-release tablets.
5.3 Hypersensitivity Reactions
Some patients who have experienced a hypersensitivity reaction to sulfasalazine may have a similar reaction to mesalamine delayed-release tablets or to other compounds that contain or are converted to mesalamine.
Mesalamine-induced cardiac hypersensitivity reactions (myocarditis and pericarditis) have been reported with mesalamine delayed-release tablets and other mesalamine medications. Take caution in prescribing this medicine to patients with conditions predisposing them to the development of myocarditis or pericarditis.
5.4 Hepatic Impairment
There have been reports of hepatic failure in patients with pre-existing liver disease who have been administered mesalamine. Exercise caution when administering mesalamine delayed-release tablets to patients with liver disease.
5.5 Upper GI Tract Obstruction
Pyloric stenosis or other organic or functional obstruction in the upper gastrointestinal tract may cause prolonged gastric retention of mesalamine delayed-release tablets, which would delay mesalamine release in the colon.
5.6 Photosensitivity
Patients with pre-existing skin conditions such as atopic dermatitis and atopic eczema have reported more severe photosensitivity reactions.
5.7 Interference with Laboratory Tests
Use of mesalamine may lead to spuriously elevated test results when measuring urinary normetanephrine by liquid chromatography with electrochemical detection because of the similarity in the chromatograms of normetanephrine and mesalamine's main metabolite, N-acetyl-5-aminosalicylic acid (N-Ac-5-ASA). Consider an alternative, selective assay for normetanephrine.
-
6 ADVERSE REACTIONS
The most serious adverse reactions seen in mesalamine delayed-release tablets clinical trials or with other products that contain or are metabolized to mesalamine are:
- Renal impairment, including renal failure [See Warnings and Precautions (5.1)]
- Mesalamine-induced acute intolerance syndrome [See Warnings and Precautions (5.2)]
- Hypersensitivity reactions [See Warnings and Precautions (5.3)]
- Hepatic impairment, including hepatic failure [See Warnings and Precautions (5.4)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Mesalamine delayed-release tablets have been evaluated in 1368 patients with ulcerative colitis in controlled and open-label trials.
Induction of Remission
In two 8-week, placebo-controlled clinical trials involving 535 patients with ulcerative colitis, 356 received 2.4 g/day or 4.8 g/day mesalamine delayed-release tablets and 179 received placebo. The most frequent adverse reaction leading to discontinuation from mesalamine delayed-release tablets therapy was exacerbation of ulcerative colitis (0.8%).
Pancreatitis occurred in less than 1% of patients during clinical trials and resulted in discontinuation of therapy with mesalamine delayed-release tablets in patients experiencing this event.
Adverse reactions occurring in mesalamine delayed-release tablets or placebo groups at a frequency of at least 1% in two 8-week, double-blind, placebo-controlled trials are listed in Table 1. The most common adverse reactions with mesalamine delayed-release tablets 2.4 g/day and 4.8 g/day were headache (5.6% and 3.4%, respectively) and flatulence (4% and 2.8%, respectively).
Table1 Adverse Reactions in Two Eight-Week, Placebo-Controlled Trials Experienced by at Least 1 % of the Mesalamine delayed-release Tablets Group and at a Rate Greater than Placebo* * Adverse reactions for which the placebo rate equalled or exceeded the rate for at least one of the mesalamine delayed-release tablets treatment groups were abdominal pain, dizziness, dyspepsia, and nausea.
Header$Adverse Reaction
Mesalamine Delayed-release Tablets
2.4 g/day
(n = 177)
Mesalamine Delayed-release Tablets
4.8 g/day
(n = 179)
Placebo
(n = 179)
Headache
10 (5.6%)
6 (3.4%)
1 (0.6%)
Flatulence
7 (4%)
5 (2.8%)
5 (2.8%)
Liver Function Test Abnormal
1 (0.6%)
4 (2.2%)
2 (1.1%)
Alopecia
0
2 (1.1%)
0
Pruritus
1 (0.6%)
2 (1.1%)
2 (1.1%)
The following adverse reactions, presented by body system, were reported infrequently (less than 1%) by mesalamine delayed-release tablets-treated ulcerative colitis patients in the two controlled trials.
Cardiac Disorder
tachycardia
Vascular Disorders
hypertension, hypotension
Skin and Subcutaneous Tissue Disorders
acne, prurigo, rash, urticaria
Gastrointestinal Disorders
abdominal distention, colitis, diarrhea, pancreatitis, rectal polyp, vomiting
Investigations
decreased platelet count
Musculoskeletal and Connective Tissue Disorders
arthralgia, back pain
Nervous System Disorders
somnolence, tremor
Respiratory, Thoracic and Mediastinal Disorders
pharyngolaryngeal pain
General Disorders and Administrative Site Disorders
asthenia, face edema, fatigue, pyrexia
Ear and Labyrinth Disorders
ear pain
Maintenance of Remission of Ulcerative Colitis
The dose evaluated in three studies of mesalamine delayed-release tablets given for the maintenance of remission in patients with ulcerative colitis was 1.2 g twice daily or 2.4 g/once daily. One of these studies was a 6-month double-blind comparator study while two were 12- to 14-month open-label studies.
The most common adverse reactions with mesalamine delayed-release tablets in the maintenance arms of long-term trials were colitis ulcerative (5.8%), headache (2.9%), liver function test abnormal (2.3%), and abdominal pain (2.2%). Of the 1082 subjects in the all maintenance studies pooled, 1.9% had severe adverse reactions. The most common severe adverse reactions were gastrointestinal disorders; these were mainly symptoms associated with ulcerative colitis.
Table 2 Adverse Reactions in Three Maintenance Trials Experienced by at Least 1% of the Mesalamine delayed-release tablets Group (maintenance phases of trials) All Mesalamine Delayed-release Tablets
(n=1082)
Adverse Reaction
n
%
Colitis ulcerative
63
(5.8%)
Headache
31
(2.9%)
Liver function test abnormal
25
(2.3%)
Abdominal pain
24
(2.2%)
Diarrhea
18
(1.7%)
Abdominal distension
14
(1.3%)
Abdominal pain upper
13
(1.2%)
Dyspepsia
13
(1.2%)
Back pain
13
(1.2%)
Rash
13
(1.2%)
Arthralgia
12
(1.1%)
Fatigue
11
(1%)
Hypertension
10
(1%)
The following adverse reactions, presented by body system, were reported infrequently (less than 1%) by mesalamine delayed-release tablets-treated patients with ulcerative colitis in the three long-term maintenance trials (maintenance phases of these trials):
Cardiac Disorder
tachycardia
Skin and Subcutaneous Tissue Disorders
acne, alopecia, pruritus, urticaria
Gastrointestinal Disorders
colitis, flatulence, nausea, pancreatitis, rectal polyp, vomiting
Nervous System Disorders
dizziness
Respiratory, Thoracic and Mediastinal Disorders
pharyngolaryngeal pain
General Disorders and Administrative Site Disorders
asthenia, pyrexia
Ear and Labyrinth Disorders
ear pain
6.2 Postmarketing Experience
In addition to the adverse reactions reported above in clinical trials involving mesalamine delayed-release tablets, the adverse reactions listed below have been identified during post-approval use of mesalamine delayed-release tablets and other mesalamine-containing products. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Body as a Whole
lupus-like syndrome, drug fever
Cardiac Disorders
pericarditis, pericardial effusion, myocarditis
Gastrointestinal
pancreatitis, cholecystitis, gastritis, gastroenteritis, gastrointestinal bleeding, perforated peptic ulcer
Hepatic
jaundice, cholestatic jaundice, hepatitis, liver necrosis, liver failure, Kawasaki-like syndrome including changes in liver enzymes
Hematologic
agranulocytosis, aplastic anemia
Immune System Disorders
anaphylactic reaction, angioedema, Stevens-Johnson syndrome (SJS), drug reaction with eosinophilia and systemic symptoms (DRESS)
Musculoskeletal and Connective Tissue Disorders
myalgia, lupus-like syndrome
Neurological/Psychiatric
peripheral neuropathy, Guillain-Barre syndrome, transverse myelitis, intracranial hypertension
Renal Disorders
interstitial nephritis, nephrogenic diabetes insipidus
Respiratory, Thoracic and Mediastinal Disorders
interstitial lung disease, hypersensitivity pneumonitis (including interstitial pneumonitis, allergic alveolitis, eosinophilic pneumonitis)
Skin
psoriasis, pyoderma gangrenosum, erythema nodosum, photosensitivity
Urogenital
reversible oligospermia
-
7 DRUG INTERACTIONS
No investigations of interaction between mesalamine delayed-release tablets and other drugs, except for certain antibiotics, have been performed [see Clinical Pharmacology (12.3)]; however, the following drug-drug interactions have been reported for products containing mesalamine:
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Published data from meta-analyses, cohort studies, and case series on the use of mesalamine during pregnancy have not reliably informed an association with mesalamine and major birth defects, miscarriage, or adverse maternal or fetal outcomes (see Data). There are adverse effects on maternal and fetal outcomes associated with ulcerative colitis in pregnancy (see Clinical Considerations).
In animal reproduction studies, there were no adverse developmental outcomes with administration of oral mesalamine during organogenesis to pregnant rats and rabbits at doses 1.8 and 2.9 times, respectively, the maximum recommended human dose (see Data).
The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the
U.S. general population, the estimated background risk of major birth defects and miscarriages in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Clinical Considerations
Disease-associated maternal and embryo/fetal risk
Published data suggest that increased disease activity is associated with the risk of developing adverse pregnancy outcomes in women with ulcerative colitis. Adverse pregnancy outcomes include preterm delivery (before 37 weeks of gestation), low birth weight (less than 2500 g) infants, and small for gestational age at birth.
Data
Human Data
Published data from meta-analyses, cohort studies, and case series on the use of mesalamine during early pregnancy (first trimester) and throughout pregnancy have not reliably informed an association of mesalamine and major birth defects, miscarriage, or adverse maternal or fetal outcomes. There is no clear evidence that mesalamine exposure in early pregnancy is associated with an increased risk of major congenital malformations, including cardiac malformations. Published epidemiologic studies have important methodological limitations which hinder interpretation of the data, including inability to control for confounders, such as underlying maternal disease, maternal use of concomitant medications, and missing information on the dose and duration of use for mesalamine products.
Animal Data
Reproduction studies with mesalamine during organogenesis have been performed in rats at doses up to 1000 mg/kg/day (1.8 times the maximum recommended human dose based on a body surface area comparison) and rabbits at doses up to 800 mg/kg/day (2.9 times the maximum recommended human dose based on a body surface area comparison) and have revealed no evidence of harm to the fetus due to mesalamine.
8.3 Lactation
Data from published literature report the presence of mesalamine and its metabolite, N-acetyl- 5-aminosalicylic acid in human milk in small amounts with relative infant doses (RID) of 0.1% or less for mesalamine (see Data). There are case reports of diarrhea in breastfed infants exposed to mesalamine (see Clinical Considerations). There is no information on the effects of the drug on milk production. The lack of clinical data during lactation precludes a clear determination of the risk of mesalamine to an infant during lactation; therefore, the developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for mesalamine and any potential adverse effects on the breastfed child from mesalamine or from the underlying maternal condition.
Clinical Considerations
Advise the caregiver to monitor the breastfed infant for diarrhea.
Data
In published lactation studies, maternal mesalamine doses from various oral and rectal formulations and products ranged from 500 mg to 4.8 g daily. The average concentration of mesalamine in milk ranged from non-detectable to 0.5 mg/L. The average concentration of N-acetyl-5-aminosalicylic acid in milk ranged from 0.2 to 9.3 mg/L. Based on these concentrations, estimated infant daily dosages for an exclusively breastfed infant are 0 to 0.075 mg/kg/day (RID 0% to 0.1%) of mesalamine and 0.03 to 1.4 mg/kg/day of N-acetyl-5-aminosalicylic acid.
8.4 Pediatric Use
Safety and effectiveness of mesalamine delayed-release tablets in pediatric patients have not been established.
8.5 Geriatric Use
Reports from uncontrolled clinical studies and postmarketing reporting systems suggested a higher incidence of blood dyscrasias (i.e., neutropenia and pancytopenia) in patients who were 65 years or older who were taking mesalamine-containing products such as mesalamine delayed-release tablets. Take caution to monitor blood cell counts closely during mesalamine therapy.
Clinical trials of mesalamine delayed-release tablets did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently from younger patients. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. Systemic exposures are increased in elderly subjects [see Clinical Pharmacology (12.3)]. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concurrent disease or other drug therapy in elderly patients.
-
10 OVERDOSAGE
Mesalamine is an aminosalicylate, and symptoms of salicylate toxicity may include tinnitus, vertigo, headache, confusion, drowsiness, sweating, seizures, hyperventilation, dyspnea, vomiting, and diarrhea. Severe intoxication may lead to disruption of electrolyte balance and blood pH, hyperthermia, dehydration, and end organ damage.
There is no specific known antidote for mesalamine overdose; however, conventional therapy for salicylate toxicity may be beneficial in the event of acute overdosage. Correct fluid and electrolyte imbalance by the administration of appropriate intravenous therapy. Maintain adequate renal function.
-
11 DESCRIPTION
Each mesalamine delayed-release tablet for oral administration contains 1.2 g 5-aminosalicylic acid (5-ASA; mesalamine), an anti-inflammatory agent. Mesalamine also has the chemical name 5-amino-2-hydroxybenzoic acid and its structural formula is:
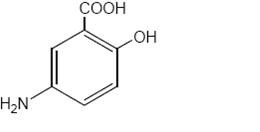
Molecular weight: 153.14
Mesalamine, USP is a light tan to pink colored, needle-shaped crystals. Color may darken on exposure to air. It is odorless or may have a slight characteristic odor.
The tablet is coated with a pH-dependent polymer film, which breaks down at or above pH 6.8, normally in the terminal ileum where mesalamine then begins to be released from the tablet core. The tablet core contains mesalamine with hydrophilic excipients and provides for extended release of mesalamine.
Each mesalamine delayed-release tablet intended for oral administration contains 1.2 g of mesalamine. In addition, each tablet contains the following inactive ingredients: carboxymethylcellulose sodium, colloidal silicon dioxide, hypromellose, iron oxide red, iron oxide yellow, magnesium stearate, methacrylic acid copolymer, microcrystalline cellulose, polyethylene glycol, sodium starch glycolate, triethyl citrate, talc and titanium dioxide.
USP Drug Release Test pending
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
The mechanism of action of mesalamine is not fully understood, but it appears to have a topical anti-inflammatory effect on the colonic epithelial cells. Mucosal production of arachidonic acid metabolites, both through the cyclooxygenase and lipoxygenase pathways, is increased in patients with chronic inflammatory bowel disease, and it is possible that mesalamine diminishes inflammation by blocking cyclooxygenase and inhibiting prostaglandin production in the colon.
Mesalamine has the potential to inhibit the activation of nuclear factor kappa B (NFкB) and consequently the production of key pro-inflammatory cytokines. It has been proposed that reduced expression of PPARγ nuclear receptors (γ-form of peroxisome proliferator-activated receptors) may be implicated in ulcerative colitis. There is evidence that mesalamine produces pharmacodynamic effects through direct activation of PPARγ receptors in the colonic/rectal epithelium.
12.2 Pharmacodynamics
The pharmacodynamic actions of mesalamine occur in the colonic/rectal mucosae local to the delivery of drug from mesalamine delayed-release tablets into the lumen. There is information suggesting that severity of colonic inflammation in ulcerative colitis patients treated with mesalamine is inversely correlated with mucosal concentrations of mesalamine. Plasma concentrations representing systemically absorbed mesalamine are not believed to contribute extensively to efficacy.
12.3 Pharmacokinetics
The total absorption of mesalamine from mesalamine delayed-release tablets 2.4 g or 4.8 g given once daily for 14 days to healthy volunteers was found to be approximately 21 to 22 % of the administered dose.
Gamma-scintigraphy studies have shown that a single dose of mesalamine delayed-release tablets 1.2 g (one tablet) passed intact through the upper gastrointestinal tract of fasted healthy volunteers. Scintigraphic images showed a trail of radio-labeled tracer in the colon, suggesting that mesalamine had distributed through this region of the gastrointestinal tract.
In a single-dose study, mesalamine delayed-release tablets 1.2 g, 2.4 g and 4.8 g were administered in the fasted state to healthy subjects. Plasma concentrations of mesalamine were detectable after 2 hours and reached a maximum by 9 to 12 hours on average for the doses studied. The pharmacokinetic parameters are highly variable among subjects (Table 3). Mesalamine systemic exposure in terms of area under the plasma concentration-time curve (AUC) was slightly more than dose proportional between 1.2 g and 4.8 g mesalamine delayed-release tablets. Maximum plasma concentrations (Cmax) of mesalamine increased approximately dose proportionately between 1.2 g and 2.4 g and sub-proportionately between 2.4 g and 4.8 g mesalamine delayed-release tablets, with the dose normalized value at 4.8 g representing, on average, 74 % of that at 2.4 g based on geometric means.
Table 3 Mean (SD) PK Parameters for Mesalamine Following Single Dose Administration of Mesalamine Delayed-release Tablets Under Fasting Conditions *Arithmetic mean of parameter values are presented except for Tmax and Tlag.
†N=43, ‡N=27, §N=36, ¶Median (min, max), #N=46,ÞN =33
Parameter* of Mesalamine
Mesalamine
Delayed-release Tablets
1.2 g
(N=47)
Mesalamine
Delayed-release Tablets
2.4 g
(N=48)
Mesalamine
Delayed-release Tablets
4.8 g
(N=48)
AUC0-t (ng.h/mL)
9039† (5054)
20538 (12980)
41434 (26640)
AUC0-∞ (ng.h/mL)
9578‡ (5214)
21084 (13185)
44775§ (30302)
Cmax (ng/mL)
857 (638)
1595 (1484)
2154 (1140)
Tmax¶ (h)
9#(4 to 32.1)
12 (4 to 34.1)
12 (4 to 34)
Tlag¶ (h)
2# (0 to 8)
2 (1 to 4)
2 (1 to 4)
T1/2 (h) (Terminal Phase)
8.56‡ (6.38)
7.05Þ (5.54)
7.25§ (8.32)
Administration of a single dose of mesalamine delayed-release tablets 4.8 g with a high fat meal resulted in further delay in absorption, and plasma concentrations of mesalamine were detectable 4 hours following dosing. However, a high fat meal increased systemic exposure of mesalamine (mean Cmax: ↑ 91 %; mean AUC: ↑ 16 %) compared to results in the fasted state. Mesalamine delayed-release tablets were administered with food in the controlled clinical trials that supported its approval.
In a single and multiple dose pharmacokinetic study of mesalamine delayed-release tablets, 2.4 g or 4.8 g was administered once daily with standard meals to 28 healthy volunteers per dose group. Plasma concentrations of mesalamine were detectable after 4 hours and were maximal by 8 hours after the single dose. Steady state was achieved generally by 2 days after dosing. Mean AUC at steady state was only modestly greater (1.1- to 1.4-fold) than predictable from single dose pharmacokinetics.
In a single dose pharmacokinetic study of mesalamine delayed-release tablets, 4.8 g was administered in the fasted state to 71 healthy male and female volunteers (28 young (18 to 35 years); 28 elderly (65 to 75 years); 15 elderly (> 75 years)). Increased age resulted in increased systemic exposure (approximately 2-fold in Cmax), to mesalamine and its metabolite N-acetyl-5-aminosalicylic acid. Increased age resulted in a slower apparent elimination of mesalamine, though there was high between-subject variability. Systemic exposures in individual subjects were inversely correlated with renal function as assessed by estimated creatinine clearance.
Table 4 Mean (SD) PK Parameters for Mesalamine Following Single-Dose Administration of Mesalamine Delayed-release Tablets 4.8 g under Fasting Conditions to Young and Elderly Subjects Arithmetic mean (SD) data are presented, N = Number of subjects
*N=15, †N=16, ‡N=13, §Median (min-max)
Parameter of 5-ASA
Young Subjects (18 to 35 years ) (N=28)
Elderly Subjects (65 to 75 years ) (N=28)
Elderly Subjects (>75 years ) (N=15)
AUC0-t (ng.h/mL)
51570 (23870)
73001 (42608)
65820 (25283)
AUC0-∞ (ng.h/mL)
58057* (22429)
89612† (40596)
63067‡ (22531)
Cmax (ng/mL)
2243 (1410)
4999 (4381)
4832 (4383)
tmax§ (h)
22 (5.98 to 48)
12.5 (4 to 36)
16 (4 to 26)
tlag§ (h)
2 (1 to 6)
2 (1 to 4)
2 (2 to 4)
t½ (h), terminal phase
5.68* (2.83)
9.68† (7.47)
8.67‡ (5.84)
Renal clearance (L/h)
2.05 (1.33)
2.04 (1.16)
2.13 (1.20)
Mesalamine is approximately 43% bound to plasma proteins at the concentration of 2.5 mcg/mL.
Metabolism
The only major metabolite of mesalamine (5-aminosalicylic acid) is N-acetyl-5-aminosalicylic acid. Its formation is brought about by N-acetyltransferase (NAT) activity in the liver and intestinal mucosa cells, principally by NAT-1.
Elimination
Elimination of mesalamine is mainly via the renal route following metabolism to N-acetyl-5-aminosalicylic acid (acetylation). However, there is also limited excretion of the parent drug in urine. Of the approximately 21 to 22% of the dose absorbed, less than 8% of the dose was excreted unchanged in the urine after 24 hours, compared with greater than 13% for N-acetyl-5-aminosalicylic acid. The apparent terminal half-lives for mesalamine and its major metabolite after administration of mesalamine delayed-release tablets 2.4 g and 4.8 g were, on average, 7 to 9 hours and 8 to 12 hours, respectively.
Drug Interactions
The potential effect of mesalamine (4.8 g given once daily) on the pharmacokinetics of four commonly used antibiotics were evaluated in healthy subjects. The four antibiotics studied and their dosing regimens were as follows: amoxicillin (single 500 mg dose), ciprofloxacin XR (single 500 mg dose), metronidazole (750 mg twice daily for 3.5 days), and sulfamethoxazole/trimethoprim (800 mg/160 mg twice daily for 3.5 days). Coadministration of mesalamine did not result in clinically significant changes in the pharmacokinetics of any of the four antibiotics. The change in Cmax and AUC of amoxicillin, ciprofloxacin and metronidazole when they were coadministered with mesalamine were all ≤ 3%. There was an increase of 12% in Cmax and an increase of 15% in AUC of sulfamethoxazole when sulfamethoxazole/trimethoprim was coadministered with mesalamine [see Drug Interactions (7)].
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In a 104-week dietary carcinogenicity study in CD-1 mice, mesalamine at doses up to 2500 mg/kg/day was not tumorigenic. This dose is 2.2 times the maximum recommended human dose (based on a body surface area comparison) of mesalamine delayed-release tablets. Furthermore, in a 104 week dietary carcinogenicity study in Wistar rats, mesalamine up to a dose of 800 mg/kg/day was not tumorigenic. This dose is 1.4 times the recommended human dose (based on a body surface area comparison) of mesalamine delayed-release tablets.
Mutagenesis
No evidence of mutagenicity was observed in an in vitro Ames test or an in vivo mouse micronucleus test.
Impairment of Fertility
No effects on fertility or reproductive performance were observed in male or female rats at oral doses of mesalamine up to 400 mg/kg/day (0.7 times the maximum recommended human dose based on a body surface area comparison).
13.2 ANIMAL PHARMACOLOGY AND OR TOXICOLOGY
In animal studies with mesalamine, a 13-week oral toxicity study in mice and 13-week and 52-week oral toxicity studies in rats and cynomolgus monkeys have shown the kidney to be the major target organ of mesalamine toxicity. Oral daily doses of 2400 mg/kg in mice and 1150 mg/kg in rats produced renal lesions including granular and hyaline casts, tubular degeneration, tubular dilation, renal infarct, papillary necrosis, tubular necrosis, and interstitial nephritis. In cynomolgus monkeys, oral daily doses of 250 mg/kg or higher produced nephrosis, papillary edema, and interstitial fibrosis.
-
14 CLINICAL STUDIES
14.1 Active, Mild to Moderate Ulcerative Colitis
Two similarly designed, randomized, double-blind, placebo-controlled trials were conducted in 517 adult patients with active, mild to moderate ulcerative colitis. The study population was primarily Caucasian (80%), had a mean age of 42 years (6% age 65 years or older), and was approximately 50% male. Both studies used mesalamine delayed-release tablets doses of 2.4 g/day and 4.8 g/day administered once daily for 8 weeks except for the 2.4 g/day group in Study 1, which was given in two divided doses (1.2 g twice daily). The primary efficacy endpoint in both trials was to compare the percentage of patients in remission after 8 weeks of treatment for the mesalamine delayed-release tablets treatment groups versus placebo. Remission was defined as an Ulcerative Colitis Disease Activity Index (UC-DAI) of ≤ 1, with scores of zero for rectal bleeding and for stool frequency, and a sigmoidoscopy score reduction of 1 point or more from baseline.
In both studies, the mesalamine delayed-release tablets doses of 2.4 g/day and 4.8 g/day demonstrated superiority over placebo in the primary efficacy endpoint (Table 4). Both mesalamine delayed-release tablets doses also provided consistent benefit in secondary efficacy parameters, including clinical improvement, treatment failure, clinical remission, and sigmoidoscopic improvement. Mesalamine delayed-release tablets 2.4 g/day and 4.8 g/day had similar efficacy profiles.
Table 5Patients in Remission at Week 8 Dose
Study 1
(n=262)
n/N (%)
Study 2
(n=255)
n/N (%)
Mesalamine Delayed-release Tablets
2.4 g/day
30/88 (34.1)
34/84 (40.5)
Mesalamine Delayed-release Tablets
4.8 g/day
26/89 (29.2)
35/85 (41.2)
Placebo
11/85 (12.9)
19/86 (22.1)
14.2 Maintenance of Remission in Patients with Ulcerative Colitis
A multicenter, randomized, double-blind, active comparator study was conducted in a total of 826 adult patients in remission from ulcerative colitis. The study population had a mean age of 45 years (8% age 65 years or older), were 52% male, and were primarily Caucasian (64%).
Maintenance of remission was assessed using a modified Ulcerative Colitis Disease Activity Index (UC-DAI). For this trial, maintenance of remission was based on maintaining endoscopic remission defined as a modified UC-DAI endoscopy subscore of ≤ 1. An endoscopy subscore of 0 represented normal mucosal appearance with intact vascular pattern and no friability or granulation. For this trial the endoscopy score definition of 1 (mild disease) was modified such that it could include erythema, decreased vascular pattern, and minimal granularity; however, it could not include friability.
Subjects were randomized in a 1:1 ratio to receive either mesalamine delayed-release tablets 2.4 g/day administered once daily or mesalamine delayed release 1.6 g/day administered as 0.8 g twice daily. The proportion of patients who maintained remission at Month 6 in this study using mesalamine delayed-release tablets 2.4 g once daily (83.7%) was similar to that seen using the comparator (mesalamine delayed release) 1.6 g/day (81.5%).
-
16 HOW SUPPLIED/STORAGE AND HANDLING
Mesalamine Delayed-release Tablets, 1.2 g are pale red-brown, oval-shaped, biconvex, bevel film-coated tablets debossed with the '711' on one side and plain on other side and are supplied as follows:
NDC: 16714-830-01 in bottles of 120 tablets
Storage
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature].
Dispense in a tight, light-resistant container.
-
17 PATIENT COUNSELING INFORMATION
- Instruct patients not to take mesalamine delayed-release tablets if they have hypersensitivity to salicylates (e.g., aspirin) or other mesalamines.
- Inform patients to let their physicians know all medications they are taking and if they:
- are allergic to sulfasalazine, salicylates or mesalamine;
- are taking non-steroidal anti-inflammatory drugs (NSAIDs) or other nephrotoxic agents;
- are taking azathioprine, or 6-mercaptopurine;
- experience cramping, abdominal pain, bloody diarrhea, fever, headache or rash;
- have a history of myocarditis or pericarditis;
- have kidney or liver disease;
- have a history of stomach blockage;
- have increased sensitivity of the skin to sun and ultraviolet light
- Patients should be instructed to swallow mesalamine delayed-release tablets whole, taking care not to break the outer coating.
- SPL UNCLASSIFIED SECTION
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
MESALAMINE
mesalamine tablet, delayed releaseProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 16714-830 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MESALAMINE (UNII: 4Q81I59GXC) (MESALAMINE - UNII:4Q81I59GXC) MESALAMINE 1.2 g Inactive Ingredients Ingredient Name Strength CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) FERRIC OXIDE RED (UNII: 1K09F3G675) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) HYPROMELLOSES (UNII: 3NXW29V3WO) MAGNESIUM STEARATE (UNII: 70097M6I30) METHACRYLIC ACID (UNII: 1CS02G8656) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIETHYL CITRATE (UNII: 8Z96QXD6UM) Product Characteristics Color RED (BROWN) Score no score Shape OVAL (OVAL) Size 10mm Flavor Imprint Code 711 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 16714-830-01 120 in 1 BOTTLE; Type 0: Not a Combination Product 06/25/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA091640 06/25/2018 Labeler - Northstar Rx LLC. (830546433) Registrant - Zydus Pharmaceuticals (USA) Inc. (156861945) Establishment Name Address ID/FEI Business Operations Cadila Healthcare Limited 918596198 ANALYSIS(16714-830) , MANUFACTURE(16714-830)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
