ACTINEL DIABETIC- dextromethorphan hydrobromide, guaifenesin liquid
ACTINEL Diabetic by
Drug Labeling and Warnings
ACTINEL Diabetic by is a Otc medication manufactured, distributed, or labeled by ACTIPHARMA, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
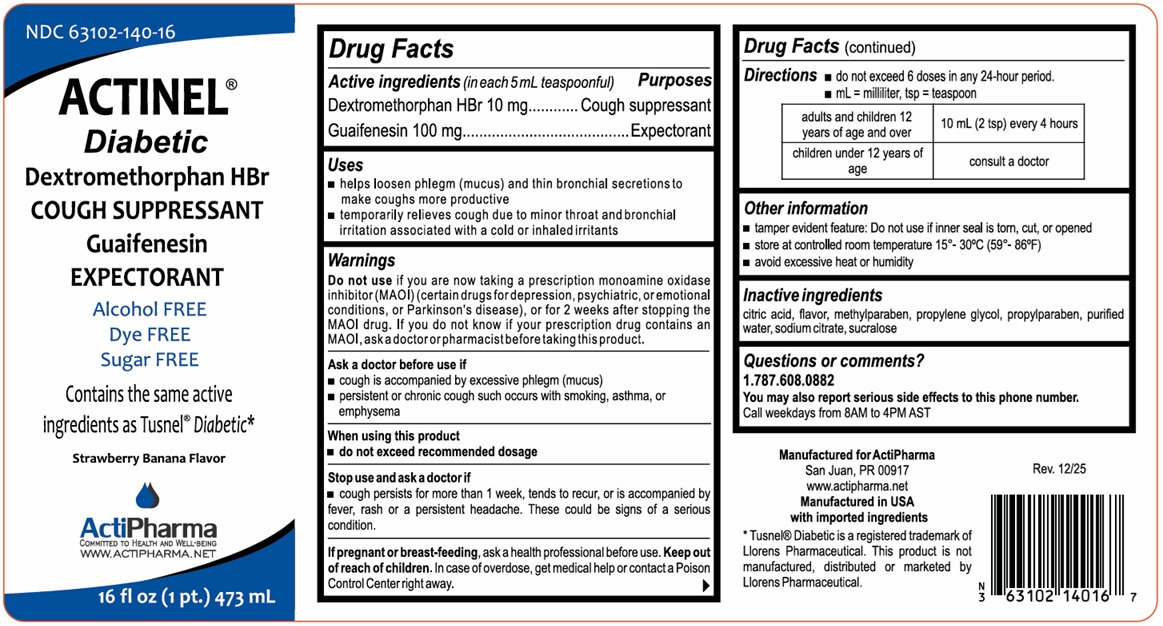
- Drug Facts
- Active ingredients (in each 5 mL teaspoonful)
- Purposes
- Uses
-
Warnings
Do not use if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
Ask a doctor before use if
cough is accompanied by excessive phlegm (mucus)
persistent or chronic cough such occurs with smoking, asthma, or emphysemaWhen using this product
do not exceed recommended dosageStop use and ask a doctor if
cough persists for more than 1 week, tends to recur, or is accompanied by fever, rash or a persistent headache, These could be signs of a serious condition.If pregnant or breast-feeding, ask a health professional before use.
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
SPL UNCLASSIFIED SECTION
Alcohol FREE
Dye FREE
Sugar FREE
Contains the same active ingredients as Tusnel® Diabetic*
ActiPharma
COMMITTED TO HEALTH AND WELL-BEING
WWW.ACTIPHARMA.NETManufactured for ActiPharma
San Juan, PR 00917Manufactured in USA with imported ingredients
*Tusnel® Diabetic is a registered trademark of Llorens Pharmaceutical. This product is not manufactured, distributed or marketed by Llorens Pharmaceutical.
- Packaging
-
INGREDIENTS AND APPEARANCE
ACTINEL DIABETIC
dextromethorphan hydrobromide, guaifenesin liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 63102-140 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 10 mg in 5 mL GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 100 mg in 5 mL Inactive Ingredients Ingredient Name Strength CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) SUCRALOSE (UNII: 96K6UQ3ZD4) Product Characteristics Color Score Shape Size Flavor STRAWBERRY (Strawberry Banana Flavor) Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 63102-140-16 473 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 02/12/2026 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 02/12/2026 Labeler - ACTIPHARMA, LLC (079340948)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.