FEXOFENADINE HYDROCHLORIDE tablet, film coated
Fexofenadine Hydrochloride by
Drug Labeling and Warnings
Fexofenadine Hydrochloride by is a Otc medication manufactured, distributed, or labeled by Mylan Institutional Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active ingredient (in each tablet)
- Purpose
- Uses
- Warnings
- Ask a doctor before use
- When using this product
- Stop use and ask a doctor if
- If pregnant or breast-feeding,
- Keep out of reach of children.
-
Directions - 60 mg (12 Hour Relief)
adults and children 12 years of age and over
take one 60 mg tablet with water every 12 hours; do not take more than 2 tablets in 24 hours
children under 12 years of age
do not use
adults 65 years of age and older
ask a doctor
consumers with kidney disease
ask a doctor
Directions - 180 mg (24 Hour Relief)
adults and children 12 years of age and over
take one 180 mg tablet with water once a day; do not take more than 1 tablet in 24 hours
children under 12 years of age
do not use
adults 65 years of age and older
ask a doctor
consumers with kidney disease
ask a doctor
- Other information
- Inactive ingredients
- Questions?
-
HOW SUPPLIED
Fexofenadine Hydrochloride Tablets, USP are available containing 60 mg or 180 mg of fexofenadine hydrochloride, USP.
The 60 mg tablets are blue film-coated, round, biconvex, beveled edge, unscored tablets debossed with M over 753 on one side of the tablet and blank on the other side. They are available as follows:
NDC: 51079-547-20 - Unit dose blister packages of 100
(10 cards of 10 tablets each).The 180 mg tablets are blue film-coated, caplet-shaped, biconvex, beveled edge, unscored tablets debossed with M 755 on one side of the tablet and blank on the other side. They are available as follows:
NDC: 51079-548-20 - Unit dose blister packages of 100
(10 cards of 10 tablets each).Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Protect from excessive moisture.
Manufactured by:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.Distributed by:
Mylan Institutional Inc.
Rockford, IL 61103 U.S.A.S-11117 R4
12/16 -
Principal Display Panel – 60 mg
NDC: 51079-547-20
Fexofenadine HCl
Tablets, USP
60 mgAntihistamine
Non-Drowsy
Indoor/Outdoor Allergy Relief
12 HR
(See Uses section of enclosed leaflet)
TAMPER EVIDENT: DO NOT USE IF BLISTER UNITS ARE
TORN, BROKEN OR SHOW ANY SIGNS OF TAMPERING.Manufactured by:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.S-11114 R4
- This unit dose package is not child resistant.
- For institutional use only.
- Keep this and all drugs out of the reach of children.
- This container provides light-resistance.
- See window for lot number and expiration date.
Distributed by:
Mylan Institutional Inc.
Rockford, IL 61103 U.S.A. -
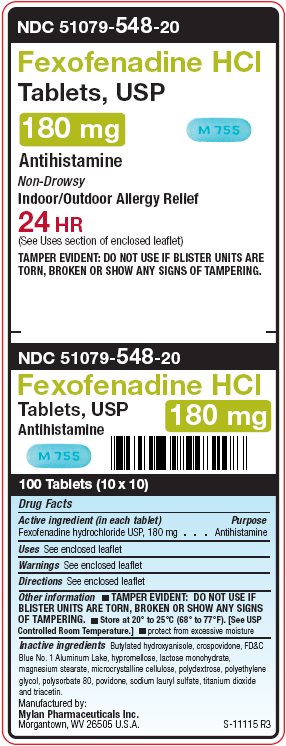
Principal Display Panel – 180 mg
NDC: 51079-548-20
Fexofenadine HCl
Tablets, USP
180 mgAntihistamine
Non-Drowsy
Indoor/Outdoor Allergy Relief
24 HR
(See Uses section of enclosed leaflet)
TAMPER EVIDENT: DO NOT USE IF BLISTER UNITS ARE
TORN, BROKEN OR SHOW ANY SIGNS OF TAMPERING.Manufactured by:
Mylan Pharmaceuticals Inc.
Morgantown, WV 26505 U.S.A.S-11115 R3
- This unit dose package is not child resistant.
- For institutional use only.
- Keep this and all drugs out of the reach of children.
- This container provides light-resistance.
- See window for lot number and expiration date.
Distributed by:
Mylan Institutional Inc.
Rockford, IL 61103 U.S.A. -
INGREDIENTS AND APPEARANCE
FEXOFENADINE HYDROCHLORIDE
fexofenadine hydrochloride tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 51079-547(NDC:0378-0781) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FEXOFENADINE HYDROCHLORIDE (UNII: 2S068B75ZU) (FEXOFENADINE - UNII:E6582LOH6V) FEXOFENADINE HYDROCHLORIDE 60 mg Inactive Ingredients Ingredient Name Strength BUTYLATED HYDROXYANISOLE (UNII: REK4960K2U) CROSPOVIDONE (15 MPA.S AT 5%) (UNII: 68401960MK) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYDEXTROSE (UNII: VH2XOU12IE) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) SODIUM LAURYL SULFATE (UNII: 368GB5141J) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIACETIN (UNII: XHX3C3X673) Product Characteristics Color BLUE Score no score Shape ROUND Size 8mm Flavor Imprint Code M;753 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 51079-547-20 100 in 1 BOX, UNIT-DOSE 02/13/2012 1 NDC: 51079-547-01 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA077081 02/13/2012 FEXOFENADINE HYDROCHLORIDE
fexofenadine hydrochloride tablet, film coatedProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 51079-548(NDC:0378-0782) Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength FEXOFENADINE HYDROCHLORIDE (UNII: 2S068B75ZU) (FEXOFENADINE - UNII:E6582LOH6V) FEXOFENADINE HYDROCHLORIDE 180 mg Inactive Ingredients Ingredient Name Strength BUTYLATED HYDROXYANISOLE (UNII: REK4960K2U) CROSPOVIDONE (15 MPA.S AT 5%) (UNII: 68401960MK) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POLYDEXTROSE (UNII: VH2XOU12IE) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) SODIUM LAURYL SULFATE (UNII: 368GB5141J) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) TRIACETIN (UNII: XHX3C3X673) Product Characteristics Color BLUE Score no score Shape OVAL (capsule-shaped) Size 18mm Flavor Imprint Code M;755 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 51079-548-20 100 in 1 BOX, UNIT-DOSE 09/27/2011 1 NDC: 51079-548-01 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA077081 09/27/2011 Labeler - Mylan Institutional Inc. (039615992)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.