BLACK COBRA TATTOO NUMBING CREAM 4% LIDOCAINE- lidocaine cream
Black Cobra Tattoo Numbing Cream 4% Lidocaine by
Drug Labeling and Warnings
Black Cobra Tattoo Numbing Cream 4% Lidocaine by is a Otc medication manufactured, distributed, or labeled by Foshan Sugar Max Cosmetics CO.,Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- WHEN USING
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- INACTIVE INGREDIENT
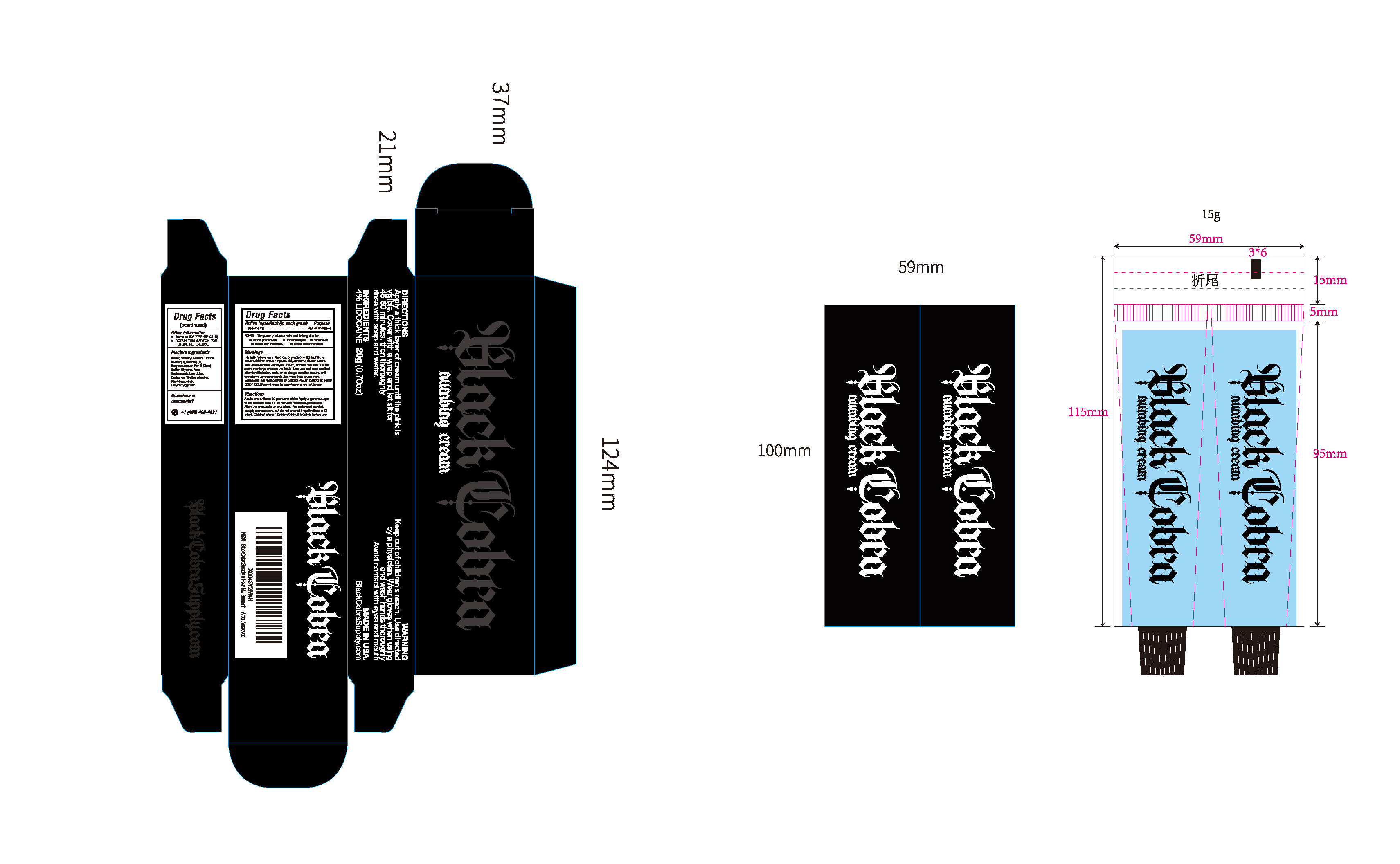
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
BLACK COBRA TATTOO NUMBING CREAM 4% LIDOCAINE
lidocaine creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 84938-002 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 0.04 g in 1 g Inactive Ingredients Ingredient Name Strength ALOE BARBADENSIS LEAF JUICE (UNII: RUE8E6T4NB) TRIETHANOLAMINE (UNII: 9O3K93S3TK) CETEARYL ALCOHOL (UNII: 2DMT128M1S) PHENOXYETHANOL (UNII: HIE492ZZ3T) AQUA (UNII: 059QF0KO0R) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) COCOS NUCIFERA (COCONUT) OIL (UNII: Q9L0O73W7L) BUTYROSPERMUM PARKII (SHEA) BUTTER (UNII: K49155WL9Y) GLYCERIN (UNII: PDC6A3C0OX) CARBOMER (UNII: 0A5MM307FC) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 84938-002-01 20 g in 1 BOTTLE; Type 0: Not a Combination Product 05/26/2025 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 05/26/2025 Labeler - Foshan Sugar Max Cosmetics CO.,Ltd (700689935) Establishment Name Address ID/FEI Business Operations Foshan Sugar Max Cosmetics CO.,Ltd 700689935 manufacture(84938-002)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.