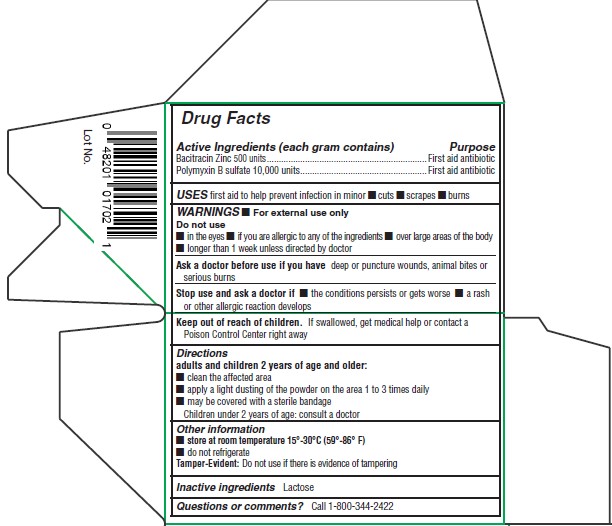
POLVO DE SULFA POWDER - CCP as CMO (42523-115)
POLVO DE SULFA FIRST AID ANTIBIOTIC by
Drug Labeling and Warnings
POLVO DE SULFA FIRST AID ANTIBIOTIC by is a Otc medication manufactured, distributed, or labeled by Coastal Contract Packaging, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
POLVO DE SULFA FIRST AID ANTIBIOTIC- bacitracin zinc, polymyxin b sulfate powder
Coastal Contract Packaging, Inc.
----------
POLVO DE SULFA POWDER - CCP as CMO (42523-115)
Warnings
- For external use only
Do not use
- in the eyes
- if you are allergic to any of the ingredients
- over large areas of the body
- longer than 1 week unless directed by doctor
Ask a doctor before use if you have deep or puncture wounds, animal bites or serious burns
Stop use and ask a doctor if
- the conditions persists or gets worse
- a rash or other allergic reaction develops
Keep out of reach of childern.If swallowed, get medical help or contact a Poison Control Center immediately.
| POLVO DE SULFA FIRST AID ANTIBIOTIC
bacitracin zinc, polymyxin b sulfate powder |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Coastal Contract Packaging, Inc. (137341611) |
Revised: 6/2025
Document Id: 37b88f6e-726a-eb56-e063-6294a90a072d
Set id: 37b88f6e-7269-eb56-e063-6294a90a072d
Version: 1
Effective Time: 20250616