DESFERAL- deferoxamine mesylate injection, powder, for solution
Desferal by
Drug Labeling and Warnings
Desferal by is a Prescription medication manufactured, distributed, or labeled by MITEM PHARMA, Excella GmbH & Co. KG Nürnberger Str. 12 90537 Feucht Germany, Novartis Pharma AG Lichtstrasse 35 CH-4056 Basel Switzerland, SGS International Services Laboratory (ISL) Limited Ringaskiddy Cork, P43 FR 63 Ireland (IRL), SGS Institut Fresenius GmbH Tegeler Weg 33 10589 Berlin Germany, SGS Institut Fresenius GmbH Im Maisel 14 65232 Taunusstein Germany, Novartis Pharma Stein AG Schaffhauserstrasse 4332 Stein Switzerland, Pharmanalytica SA Via Balestra 6600 Locarno Switzerland, Packaging Coordinators, LLC 3001 Red Lion Road Philadelphia, PA 19114, Wasserburger Arzneimittelwerk GmbH Herderstraβe 1,2 and Molkerei-Bauer-Straβe 18 83512 Wasserburg Germany, Labor LS SE & Co. KG Mangelsfeld 4, 5, 6, Bad Bocklet, Bavaria 97708 Germany, Solvias AG Römerpark 2 4303 Kaiseraugst Switzerland. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- INDICATIONS & USAGE
- DOSAGE & ADMINISTRATION
- DOSAGE FORMS & STRENGTHS
- CONTRAINDICATIONS
- WARNINGS AND PRECAUTIONS
- ADVERSE REACTIONS
- DRUG INTERACTIONS
- USE IN SPECIFIC POPULATIONS
- OVERDOSAGE
- DESCRIPTION
- CLINICAL PHARMACOLOGY
- NONCLINICAL TOXICOLOGY
- HOW SUPPLIED
- 88436-1 - Section Title Not Found In Database
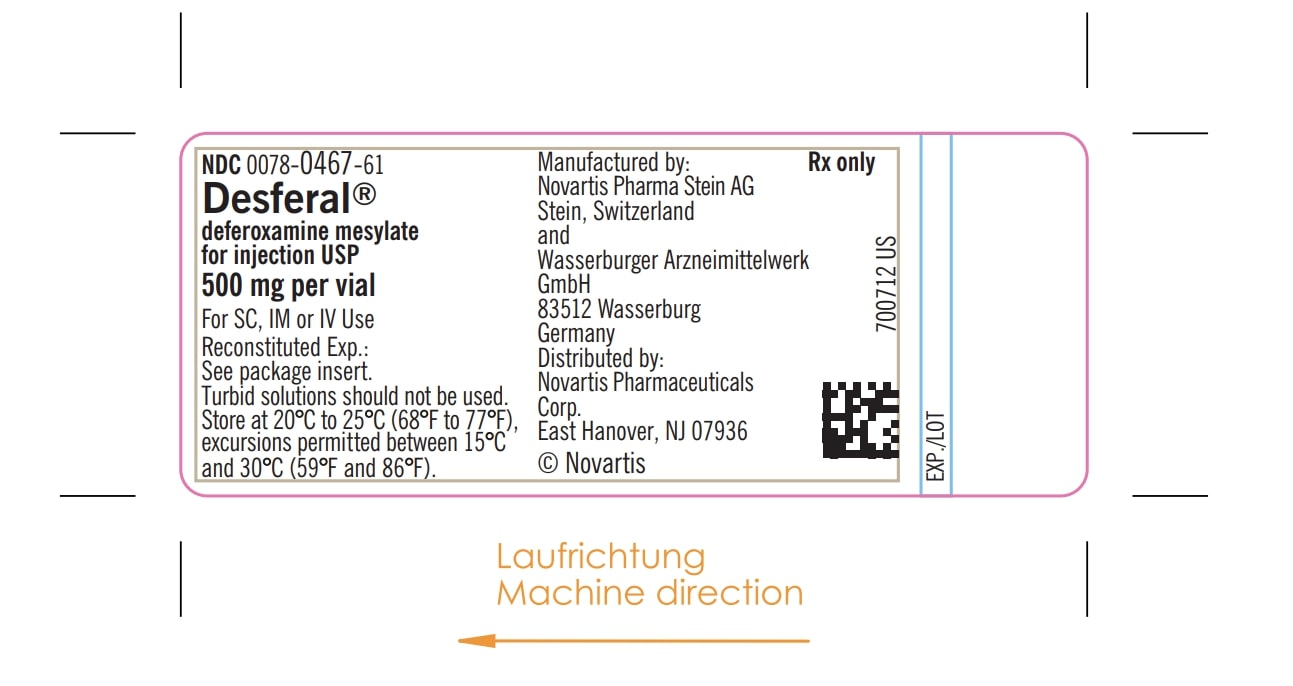
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
DESFERAL
deferoxamine mesylate injection, powder, for solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 87081-467 Route of Administration SUBCUTANEOUS, INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEFEROXAMINE MESYLATE (UNII: V9TKO7EO6K) (DEFEROXAMINE - UNII:J06Y7MXW4D) DEFEROXAMINE MESYLATE 500 mg in 5 mL Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 87081-467-91 4 mL in 1 VIAL; Type 0: Not a Combination Product 04/02/1968 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA016267 04/02/1968 Labeler - MITEM PHARMA (500300624) Establishment Name Address ID/FEI Business Operations Excella GmbH & Co. KG Nürnberger Str. 12 90537 Feucht Germany 329809800 api manufacture(87081-467) , analysis(87081-467) Establishment Name Address ID/FEI Business Operations Novartis Pharma AG Lichtstrasse 35 CH-4056 Basel Switzerland 482347168 analysis(87081-467) Establishment Name Address ID/FEI Business Operations SGS International Services Laboratory (ISL) Limited Ringaskiddy Cork, P43 FR 63 Ireland (IRL) 985785798 analysis(87081-467) Establishment Name Address ID/FEI Business Operations SGS Institut Fresenius GmbH Tegeler Weg 33 10589 Berlin Germany 341259550 analysis(87081-467) Establishment Name Address ID/FEI Business Operations SGS Institut Fresenius GmbH Im Maisel 14 65232 Taunusstein Germany 317219699 analysis(87081-467) Establishment Name Address ID/FEI Business Operations Novartis Pharma Stein AG Schaffhauserstrasse 4332 Stein Switzerland 488152505 manufacture(87081-467) , analysis(87081-467) , pack(87081-467) Establishment Name Address ID/FEI Business Operations Pharmanalytica SA Via Balestra 6600 Locarno Switzerland 487967499 analysis(87081-467) Establishment Name Address ID/FEI Business Operations Packaging Coordinators, LLC 3001 Red Lion Road Philadelphia, PA 19114 078525133 pack(87081-467) Establishment Name Address ID/FEI Business Operations Wasserburger Arzneimittelwerk GmbH Herderstraβe 1,2 and Molkerei-Bauer-Straβe 18 83512 Wasserburg Germany 326482247 manufacture(87081-467) , analysis(87081-467) , pack(87081-467) Establishment Name Address ID/FEI Business Operations Labor LS SE & Co. KG Mangelsfeld 4, 5, 6, Bad Bocklet, Bavaria 97708 Germany 314929072 analysis(87081-467) Establishment Name Address ID/FEI Business Operations Solvias AG Römerpark 2 4303 Kaiseraugst Switzerland 480739627 analysis(87081-467)
Trademark Results [Desferal]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 DESFERAL 72130271 0737030 Live/Registered |
Ciba Limited 1961-10-19 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.