Dye-Free Children's Loratadine Chewable Tablets USP, 5 mg
DYE-FREE CHILDRENS LORATADINE by
Drug Labeling and Warnings
DYE-FREE CHILDRENS LORATADINE by is a Otc medication manufactured, distributed, or labeled by Ohm Laboratories Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
DYE-FREE CHILDRENS LORATADINE- loratadine tablet, chewable
Ohm Laboratories Inc.
----------
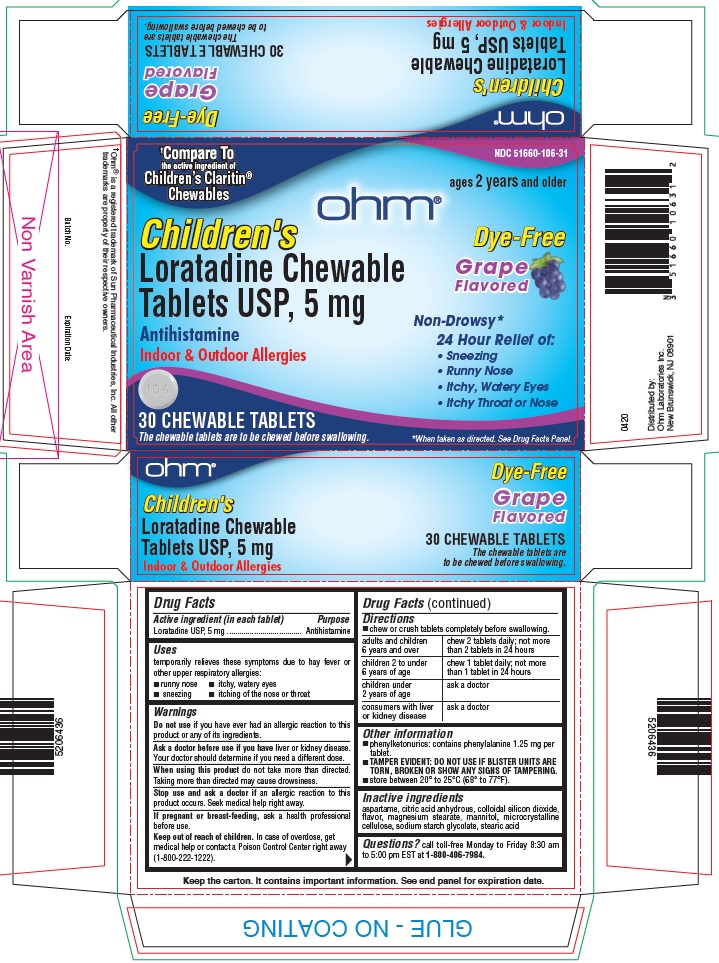
Dye-Free Children's Loratadine Chewable Tablets USP, 5 mg
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- sneezing
- itchy, watery eyes
- itching of the nose or throat
Warnings
Ask a doctor before use if you haveliver or kidney disease. Your doctor should determine if you need a different dose.
When using this productdo not take more than directed. Taking more than directed may cause drowsiness.
Directions
- chew or crush tablets completely before swallowing.
|
adults and children 6 years and over |
chew 2 tablets daily; not more than 2 tablets in 24 hours |
|
children 2 to under 6 years of age |
chew 1 tablet daily; not more than 1 tablet in 24 hours |
|
children under 2 years of age |
ask a doctor |
|
consumers with liver or kidney disease |
ask a doctor |
Other information
- phenylketonurics: contains phenylalanine 1.25 mg per tablet.
- TAMPER EVIDENT: DO NOT USE IF BLISTER UNITS ARE TORN, BROKEN OR SHOW ANY SIGNS OF TAMPERING.
- store between 20° to 25°C (68° to 77°F).
Inactive ingredients
aspartame, citric acid anhydrous, colloidal silicon dioxide, flavor, magnesium stearate, mannitol, microcrystalline cellulose, sodium starch glycolate, stearic acid
Questions?
call toll-free Monday to Friday 8:30 am to 5:00 pm EST at 1-800-406-7984.
Keep the carton. It contains important information. See end panel for expiration date.
PRINCIPAL DISPLAY PANEL - 5 mg Tablet Blister Pack Carton
NDC: 51660-106-31
†Compare to the active ingredient of Children's Claritin ®Chewable
ages 2 years and older
Children's
Loratadine Chewable Tablets USP, 5 mg
Antihistamine
Indoor & Outdoor Allergies
30 CHEWABLE TABLETS
The chewable tablets are to be chewed before swallowing.
Dye-Free
Grape Flavored
Non-Drowsy*
24 Hour Relief of:
- Sneezing
- Runny Nose
- Itchy, Watery Eyes
- Itchy Throat or Nose
*When taken as directed. See Drug Facts Panel.
†Ohm® is a registered trademark of Sun Pharmaceutical Industries, Inc. All other trademarks are property of their respective owners.
| DYE-FREE CHILDRENS LORATADINE
loratadine tablet, chewable |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Ohm Laboratories Inc. (184769029) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Ohm Laboratories Inc. | 184769029 | manufacture(51660-106) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.