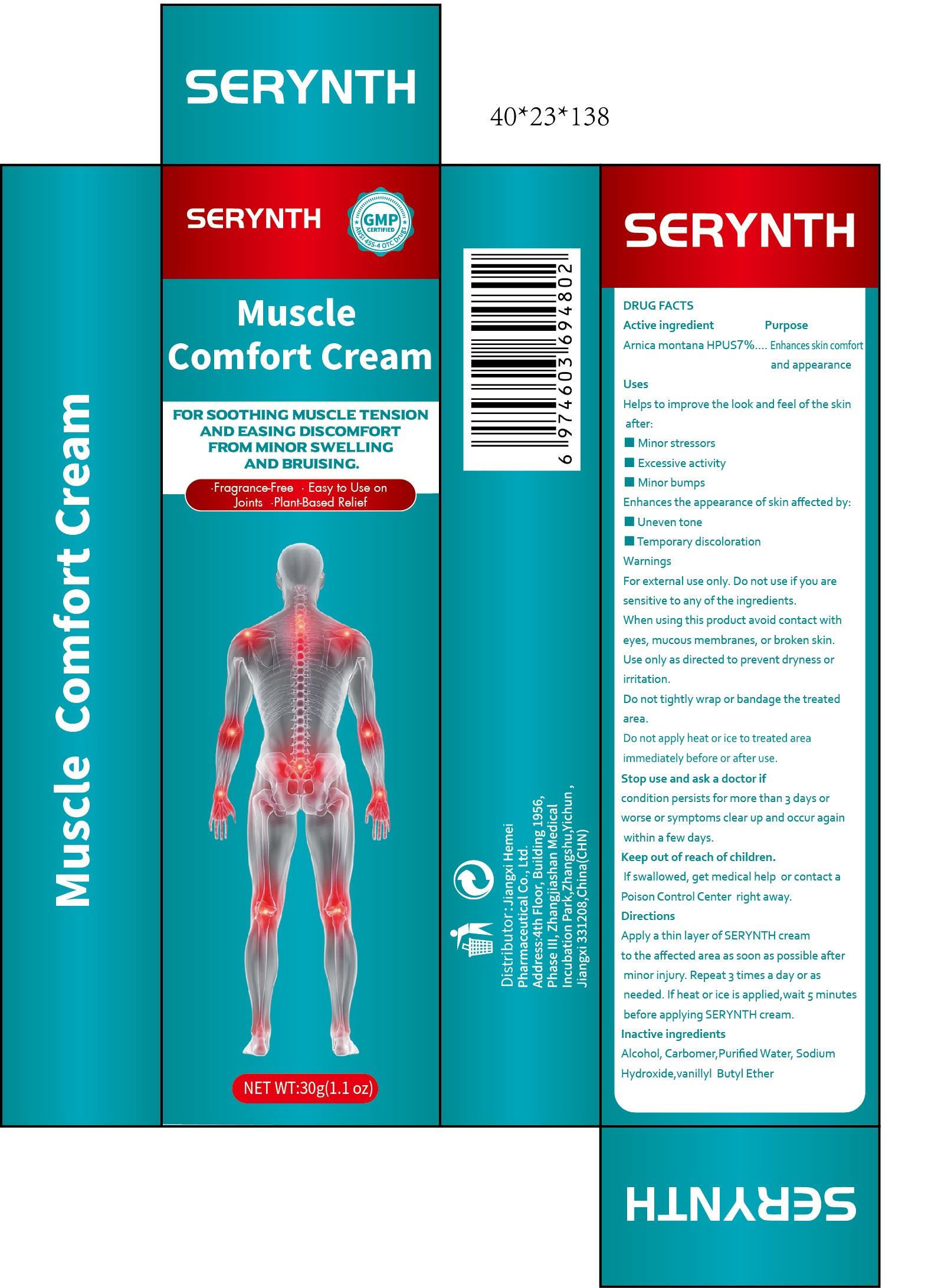
SERYNTH Muscle Comfort by Jiangxi Hemei Pharmaceutical Co., Ltd 84010-149complete
SERYNTH Muscle Comfort by
Drug Labeling and Warnings
SERYNTH Muscle Comfort by is a Otc medication manufactured, distributed, or labeled by Jiangxi Hemei Pharmaceutical Co., Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
SERYNTH MUSCLE COMFORT- arnica montana hpus 7% muscle comfort cream
Jiangxi Hemei Pharmaceutical Co., Ltd
----------
84010-149complete
Use
Helps to improve the look and feel ofthe skin after:
■Minor stressors
■Excessive activity
■ Minor bumps
Enhances the appearance of skin affected by:
■ Uneven tone■Temporary discoloration
Do not use
Do not use if you are allergic to healsure or to any of this product's inactive ingredients
When Using
When using this product avoid contact with eyes, mucous membranes, or broken skin.Use only as directed to prevent dryness or
irritation.
Do not tightly wrap or bandage the treated area. Do not apply heat or ice to treated area immediately before or after use.
Stop Use
condition persists for more than 3 days or worse or symptoms clear up and occur again within a few days.
Ask Doctor
condition persists for more than 3 days or worse or symptoms clear up and occur again within a few days.
Keep Out Of Reach Of Children
If swallowed, get medical help or contact a Poison Control Center right away
| SERYNTH MUSCLE COMFORT
arnica montana hpus 7% muscle comfort cream |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - Jiangxi Hemei Pharmaceutical Co., Ltd (724892056) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Jiangxi Hemei Pharmaceutical Co., Ltd | 724892056 | manufacture(84010-149) | |