IBUPROFEN PM- diphenhydramine hydrochloride, ibuprofen capsule, liquid filled
Ibuprofen PM by
Drug Labeling and Warnings
Ibuprofen PM by is a Otc medication manufactured, distributed, or labeled by L.N.K. International, Inc., LNK International, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active ingredients (in each capsule)
- Purpose
- Uses
-
Warnings
Allergy alert: Ibuprofen may cause a severe allergic reaction, especially in people allergic to aspirin. Symptoms may include:
- rash
- facial swelling
- blisters
- hives
- skin reddening
- shock
- asthma (wheezing)
If an allergic reaction occurs, stop use and seek medical help right away.
Stomach bleeding warning: This product contains a nonsteroidal anti-inflammatory drug (NSAID), which may cause severe stomach bleeding. The chance is higher if you
- are age 60 or older
- have had stomach ulcers or bleeding problems
- take a blood thinning (anticoagulant) or steroid drug
- take other drugs containing prescription or nonprescription NSAIDs [aspirin, ibuprofen, naproxen, or others]
- have 3 or more alcoholic drinks every day while using this product
- take more or for a longer time than directed
Do not use
- if you have ever had an allergic reaction to any other pain reliever/fever reducer
- unless you have time for a full night's sleep
- in children under 12 years of age
- with any other product containing diphenhydramine, even one used on skin
- right before or after heart surgery
- if you have sleeplessness without pain
Ask a doctor before use if
- you have problems or serious side effects from taking pain relievers or fever reducers
- you have a history of stomach problems, such as heartburn
- you have high blood pressure, heart disease, liver cirrhosis, kidney disease, or asthma
- you are taking a diuretic
- you have glaucoma
- you have a breathing problem such as emphysema or chronic bronchitis
- stomach bleeding warning applies to you
- you have trouble urinating due to an enlarged prostate gland
Ask a doctor or pharmacist before use if you are
- taking sedatives or tranquilizers, or any other sleep-aid
- under a doctor's care for any continuing medical illness
- taking any other antihistamines
- taking any other drug
- taking aspirin for heart attack or stroke, because ibuprofen may decrease this benefit of aspirin
When using this product
- drowsiness will occur
- avoid alcoholic drinks
- do not drive a motor vehicle or operate machinery
- take with food or milk if stomach upset occurs
- the risk of heart attack or stroke may increase if you use more than directed or for longer than directed
Stop use and ask a doctor if
- you experience any of the following signs of stomach bleeding:
- feel faint
- vomit blood
- have bloody or black stools
- have stomach pain that does not get better
- pain gets worse or lasts more than 10 days
- sleeplessness persists continuously for more than 2 weeks. Insomnia may be a symptom of a serious underlying medical illness.
- redness or swelling is present in the painful area
- any new symptoms appear
- Directions
- Other information
-
Inactive ingredients
anidrisorb, D&C red no. 33, FD&C blue no. 1, gelatin, hydrolysed gelatin, medium chain triglyceride, opacode white ink, polyethylene glycol 600, potassium hydroxide, purified water.
Ingredients of opacode white ink: lecithin (soya), n-butyl alcohol, purified water, shellac glaze in ethanol, simethicone and titanium dioxide. - Questions or comments?
-
Principal Display Panel
QUALITY
PLUSNDC: 50844-611-19
†Compare to the
active ingredients in
Advil® PM LIQUI-GELS®IBUPROFEN PM
SOFTGELS
Ibuprofen 200 mg / Diphenhydramine HCl 25 mg
Pain Reliever (NSAID) / Nighttime Sleep-AidSEE NEW WARNINGS INFORMATION
8 Softgels**
**Liquid Filled Capsules†This product is not manufactured or distributed by
Pfizer Consumer Healthcare, owner of the registered
trademark Advil® PM LIQUI-GELS®.
50844 ORG111261119Product of India
Distributed by
LNK INTERNATIONAL, INC.
60 Arkay Drive, Hauppauge, NY 11788
USATAMPER EVIDENT: DO NOT USE IF CARTON IS
OPENED OR IF BLISTER UNIT IS TORN, BROKEN,
OR SHOWS ANY SIGNS OF TAMPERING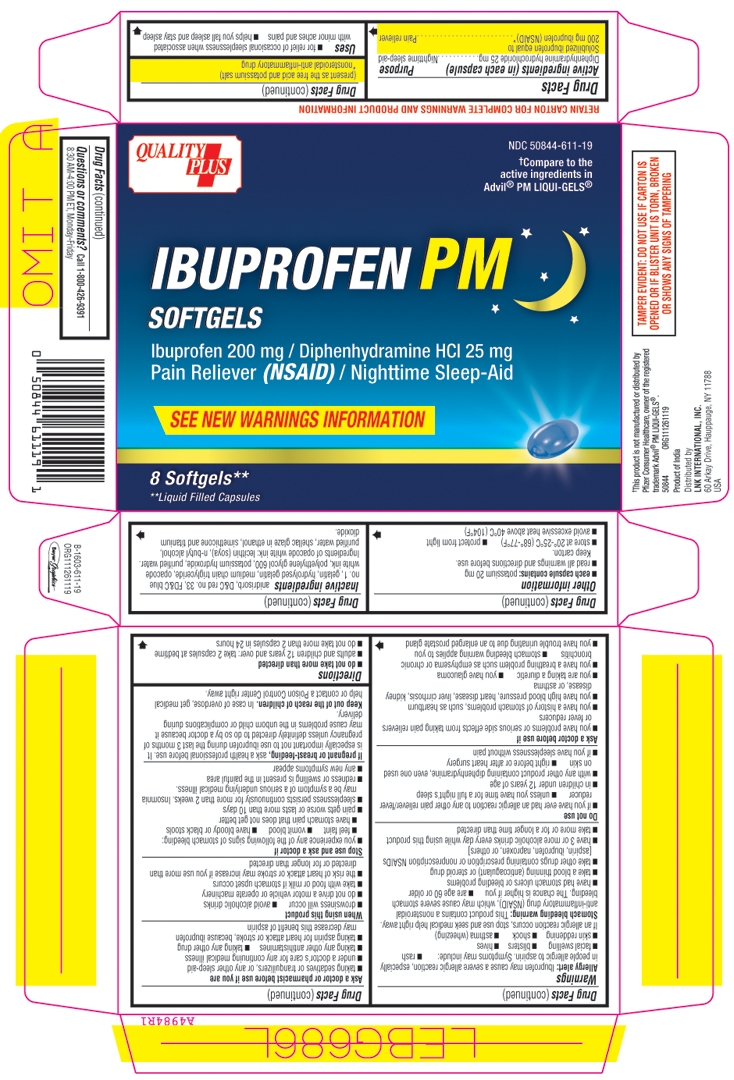
Quality Plus 44-611
-
INGREDIENTS AND APPEARANCE
IBUPROFEN PM
diphenhydramine hydrochloride, ibuprofen capsule, liquid filledProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 50844-611 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIPHENHYDRAMINE HYDROCHLORIDE (UNII: TC2D6JAD40) (DIPHENHYDRAMINE - UNII:8GTS82S83M) DIPHENHYDRAMINE HYDROCHLORIDE 25 mg IBUPROFEN (UNII: WK2XYI10QM) (IBUPROFEN - UNII:WK2XYI10QM) IBUPROFEN 200 mg Inactive Ingredients Ingredient Name Strength FD&C BLUE NO. 1 (UNII: H3R47K3TBD) GELATIN, UNSPECIFIED (UNII: 2G86QN327L) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) POLYETHYLENE GLYCOL 600 (UNII: NL4J9F21N9) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) WATER (UNII: 059QF0KO0R) D&C RED NO. 33 (UNII: 9DBA0SBB0L) Product Characteristics Color PURPLE (bluish tint) Score no score Shape OVAL Size 15mm Flavor Imprint Code 1007 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 50844-611-19 1 in 1 CARTON 07/08/2013 1 8 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA200888 07/08/2013 Labeler - L.N.K. International, Inc. (038154464) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 038154464 PACK(50844-611)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.