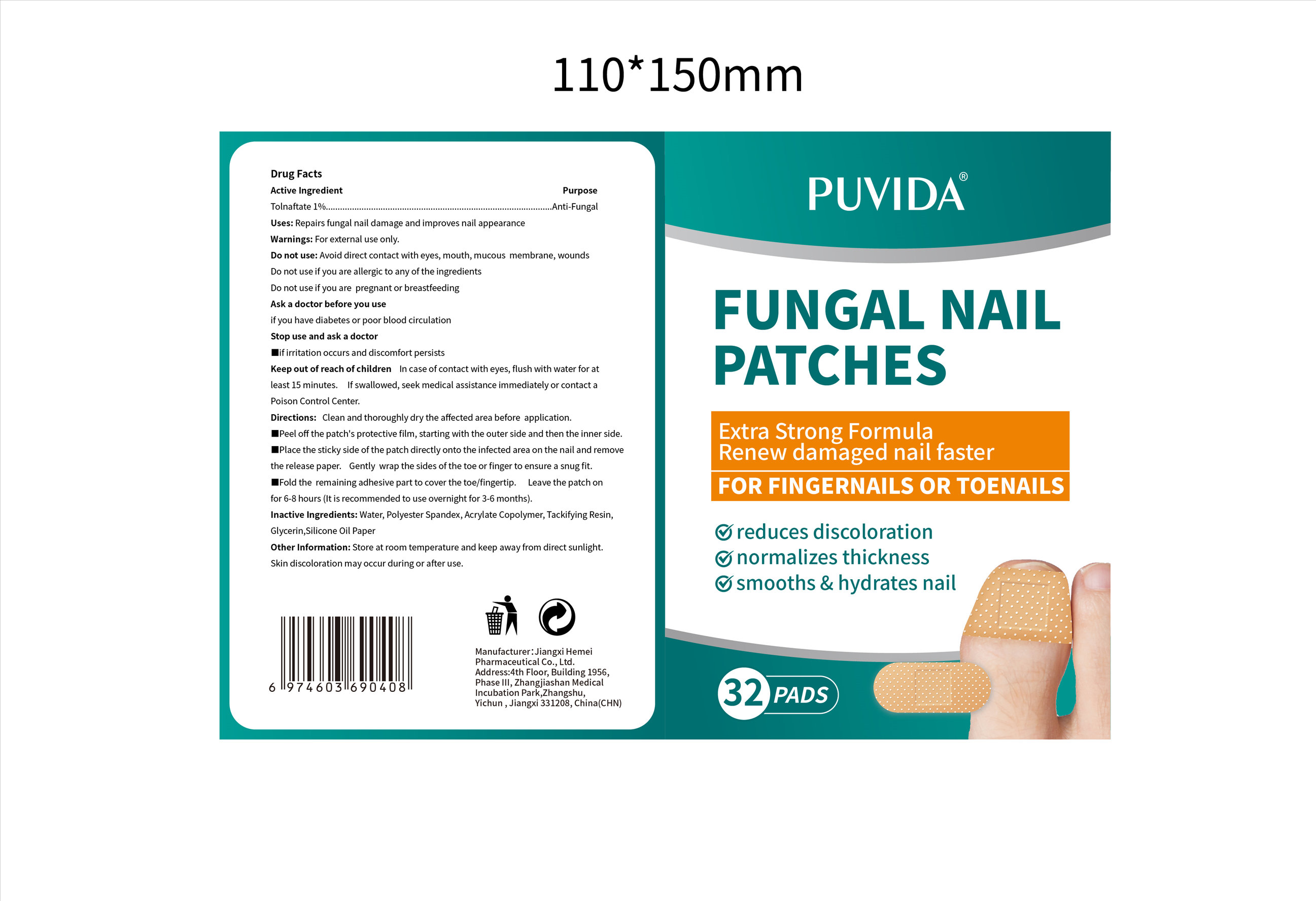
PUVIDA FUNGAL NAIL by Jiangxi Hemei Pharmaceutical Co., Ltd 84010-158 complete
PUVIDA FUNGAL NAIL by
Drug Labeling and Warnings
PUVIDA FUNGAL NAIL by is a Otc medication manufactured, distributed, or labeled by Jiangxi Hemei Pharmaceutical Co., Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
PUVIDA FUNGAL NAIL- tolnaftate 1% fungal nail patch
Jiangxi Hemei Pharmaceutical Co., Ltd
----------
84010-158 complete
Do not use
Do not use if you are allergic to any of the ingredients
Do not use if you are pregnant or breastfeeding
Keep Oot Of Reach Of Children
In case of contact with eyes, flush with water for at least 15 minutes. lf swallowed, seek medical assistance immediately or contact a Poison Control Center.
Directions
Clean and thoroughly dry the affected area before application. ■Peel off the patch's protective film, starting with the outer side and then the inner side. ■Place the sticky side of the patch directly onto the infected area on the nail and remove the release paper. Gently wrap the sides of the toe or finger to ensure a snug fit. ■Fold the remaining adhesive part to cover the toe/fingertip. Leave the patch onfor 6-8 hours (It is recommended to use overnight for 3-6 months).
Other information
Store at room temperature and keep away from direct sunlight.
Skin discoloration may occur during or after use.
| PUVIDA FUNGAL NAIL
tolnaftate 1% fungal nail patch |
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
| Labeler - Jiangxi Hemei Pharmaceutical Co., Ltd (724892056) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Jiangxi Hemei Pharmaceutical Co., Ltd | 724892056 | manufacture(84010-158) | |