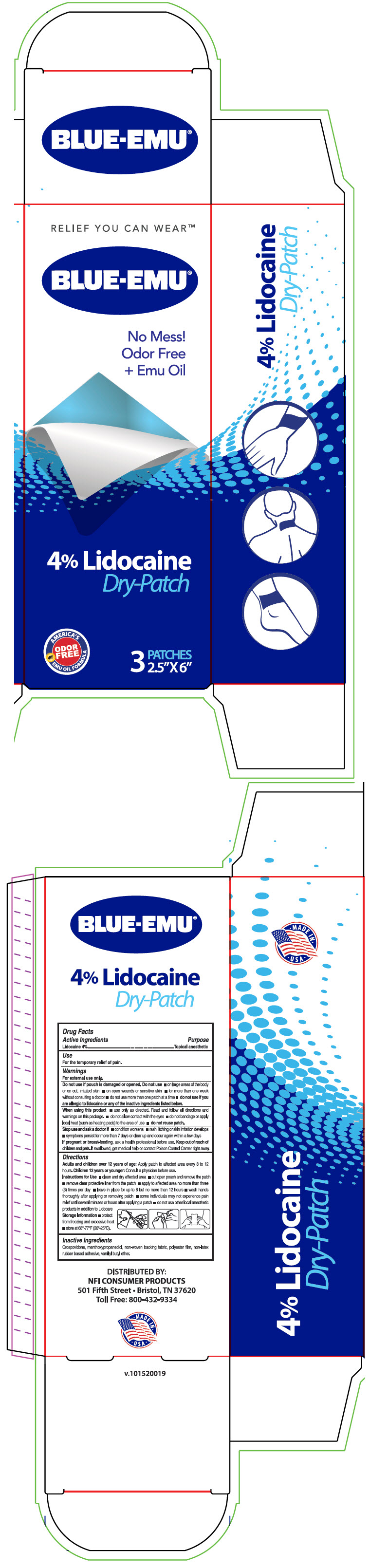
Blue-Emu® Lidocaine Dry-Patch
Blue-Emu Lidocaine Dry by
Drug Labeling and Warnings
Blue-Emu Lidocaine Dry by is a Otc medication manufactured, distributed, or labeled by NFI, LLC, ProSolus, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
BLUE-EMU LIDOCAINE DRY- lidocaine patch
Kingsway Pharmaceuticals dba NFI, LLC
----------
Blue-Emu® Lidocaine Dry-Patch
Warnings
For external use only.
Do not use if pouch is damaged or opened. Do not use
- on large areas of the body or on cut, irritated skin
- on open wounds or sensitive skin
- for more than one week without consulting a doctor
- do not use more than one patch at a time
- do not use if you are allergic to lidocaine or any of the inactive ingredients listed below.
When using this product
- use only as directed. Read and follow all directions and warnings on this package.
- do not allow contact with the eyes
- do not bandage or apply local heat (such as heating pads) to the area of use
- do not reuse patch.
Directions
Instructions for Use
- clean and dry affected area
- cut open pouch and remove the patch
- remove clear protective liner from the patch
- apply to affected area no more than three (3) times per day
- leave in place for up to 8 but no more than 12 hours
- wash hands thoroughly after applying or removing patch
- some individuals may not experience pain relief until several minutes or hours after applying a patch
- do not use other local anesthetic products in addition to Lidocare
| BLUE-EMU LIDOCAINE DRY
lidocaine patch |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - Kingsway Pharmaceuticals dba NFI, LLC (121681919) |
| Registrant - Kingsway Pharmaceuticals dba NFI, LLC (121681919) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| ProSolus, Inc. | 969793079 | MANUFACTURE(69993-425) | |
Revised: 12/2024
Document Id: 71cd72be-ad71-43f2-8bd7-df2182c271d0
Set id: 3aa15458-30c2-4a63-9c9b-b65e8fe3e5de
Version: 3
Effective Time: 20241209