Alclene Hand Sanitizer by Global Cosmetics (China) Company Limited alclene Hand Sanitizer
Alclene Hand Sanitizer by
Drug Labeling and Warnings
Alclene Hand Sanitizer by is a Otc medication manufactured, distributed, or labeled by Global Cosmetics (China) Company Limited. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
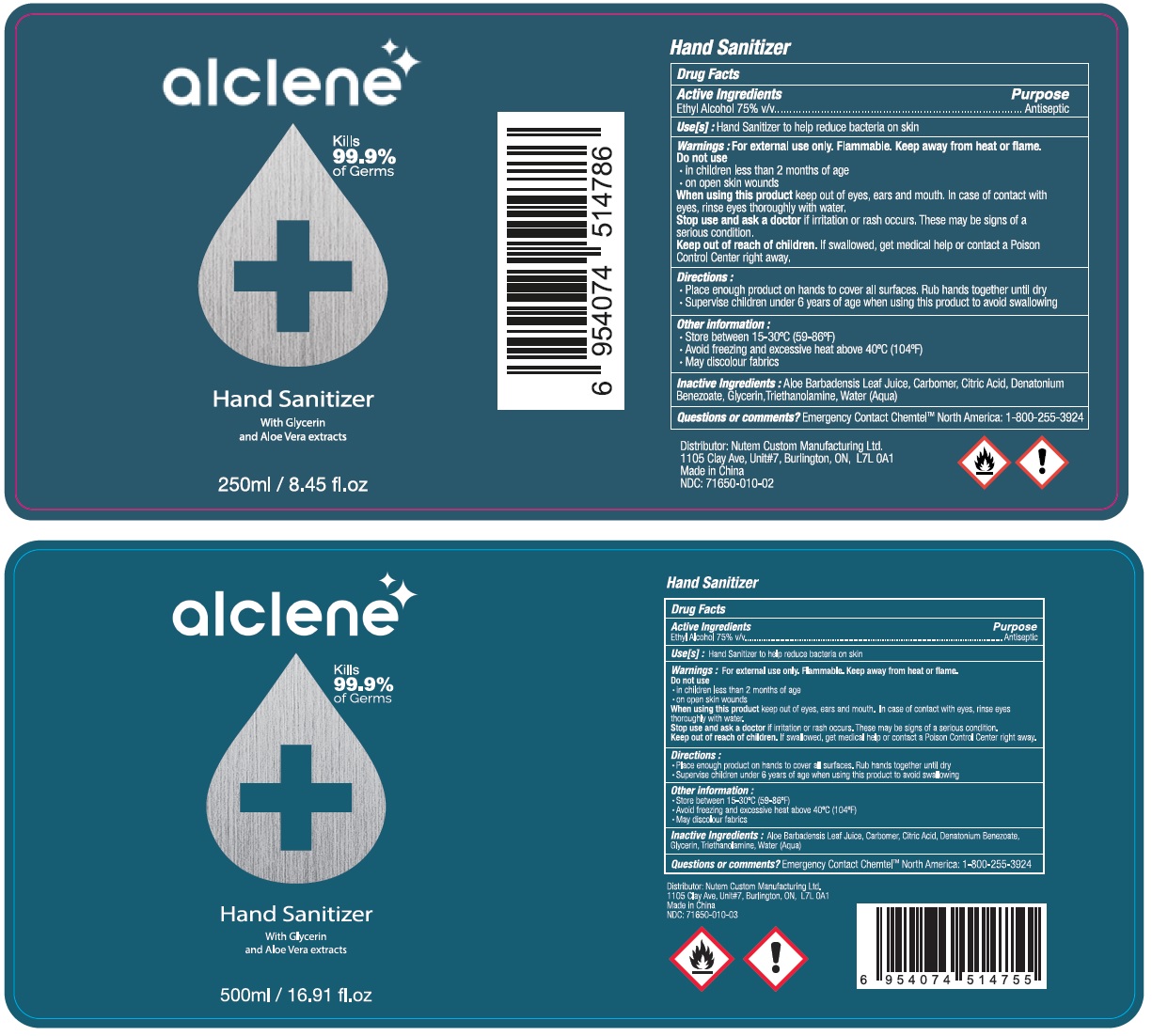
ALCLENE HAND SANITIZER- alcohol gel
Global Cosmetics (China) Company Limited
Disclaimer: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
----------
alclene Hand Sanitizer
Warnings : For external use only. Flammable. Keep away from heat or flame.
Do not use
- in children less than 2 months of age
- on open skin wounds
When using this product keep out of eyes, ears and mouth. In case of contact with eyes, rinse eyes thoroughly with water.
Stop use and ask a doctor if irritation or ash occurs. These may be signs of a serious condition.
Directions :
- Place enough product on hands to cover all surfaces. Rub hands together until dry
- Supervise children under 6 years of age when using this product to avoid swallowing
Other information :
- Store between 15-30°C (59-86°F)
- Avoid freezing and excessive heat above 40°C (104°F)
- May discolour fabrics
Inactive Ingredients : Aloe Barbadensis Leaf Juice, Carbomer, Citric Acid, Denatonium Benzoate, Glycerin, Triethanolamine, Water (Aqua)
| ALCLENE HAND SANITIZER
alcohol gel |
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||
| Labeler - Global Cosmetics (China) Company Limited (527852739) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Global Cosmetics (China) Company Limited | 527852739 | manufacture(71650-010) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.