SODIUM CHLORIDE solution/ drops
Sodium Chloride by
Drug Labeling and Warnings
Sodium Chloride by is a Otc medication manufactured, distributed, or labeled by CVS Pharmacy, Akorn, Inc., Akorn, Inc, Akorn, Inc. . Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Use
-
Warnings
Do not use
- this product except under the advice and supervision of a doctor
- if imprinted seal on bottle is missing or broken
- if solution changes color or becomes cloudy
When using this product
- temporary burning and irritation upon being instilled into eye may occur
- to avoid contamination, do not touch tip of container to any surface
- replace cap after each use
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
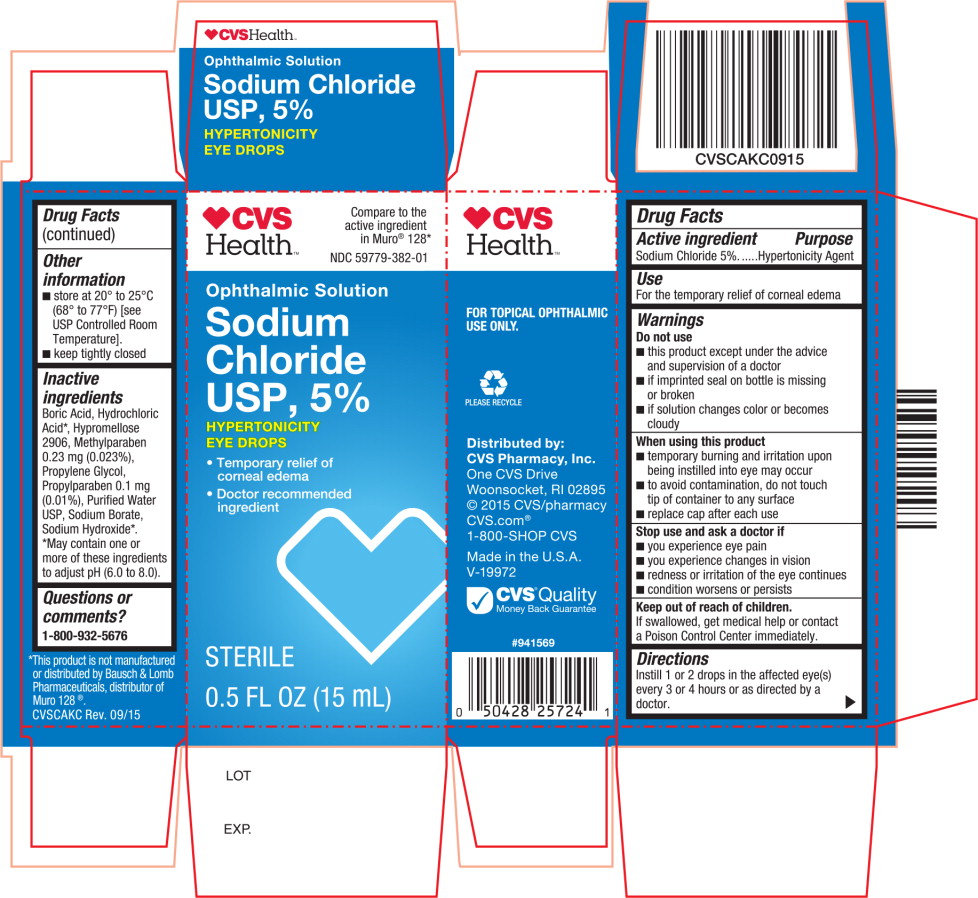
PRINCIPAL DISPLAY PANEL
Principal Display Panel Text for Container Label:
CVS Health™ Logo
NDC: 59779-382-01
Ophthalmic Solution
Sodium
Chloride
USP, 5%
HYPERTONICITY
EYE DROPS
STERILE
0.5 FL OZ (15 mL) -
PRINCIPAL DISPLAY PANEL
Principal Display Panel Text for Carton Label:
CVS Health™ Logo Compare to the
active ingredient
in Muro® 128*
NDC: 59779-382-01
Ophthalmic Solution
Sodium
Chloride
USP, 5%
HYPERTONICITY
EYE DROPS
- Temporary relief of
corneal edema - Doctor recommended
ingredient
STERILE
0.5 FL OZ (15 mL)
- Temporary relief of
-
INGREDIENTS AND APPEARANCE
SODIUM CHLORIDE
sodium chloride solution/ dropsProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 59779-382 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Sodium Chloride (UNII: 451W47IQ8X) (Sodium Cation - UNII:LYR4M0NH37) Sodium Chloride 50 mg in 1 mL Inactive Ingredients Ingredient Name Strength Boric Acid (UNII: R57ZHV85D4) Hypromellose 2906 (4 MPA.S) (UNII: 6531BH494O) Propylene Glycol (UNII: 6DC9Q167V3) Sodium Borate (UNII: 91MBZ8H3QO) Sodium Hydroxide (UNII: 55X04QC32I) Hydrochloric Acid (UNII: QTT17582CB) Water (UNII: 059QF0KO0R) Methylparaben (UNII: A2I8C7HI9T) Propylparaben (UNII: Z8IX2SC1OH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 59779-382-01 1 in 1 CARTON 1 15 mL in 1 BOTTLE, DROPPER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part349 06/14/2013 Labeler - CVS Pharmacy (062312574) Registrant - Akorn, Inc. (062649876) Establishment Name Address ID/FEI Business Operations Akorn, Inc 063434679 PACK(59779-382) , LABEL(59779-382) Establishment Name Address ID/FEI Business Operations Akorn, Inc. 155135783 MANUFACTURE(59779-382) , ANALYSIS(59779-382) , STERILIZE(59779-382)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.