LIPID SHIELD SPF 30- zinc oxide cream
Lipid Shield SPF 30 by
Drug Labeling and Warnings
Lipid Shield SPF 30 by is a Otc medication manufactured, distributed, or labeled by Allure Labs. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
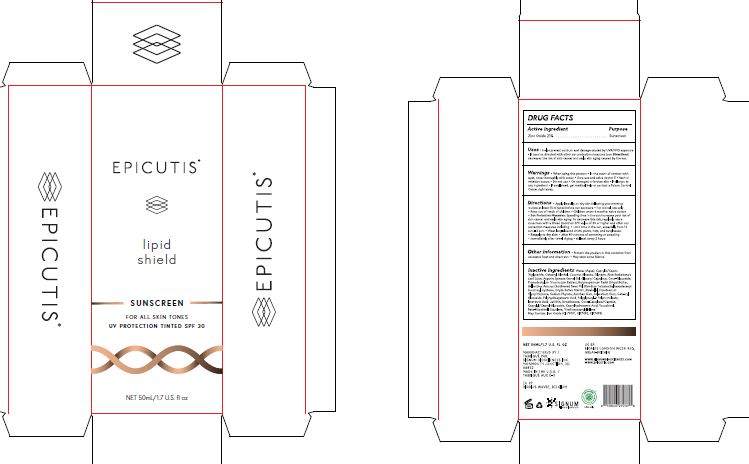
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- DO NOT USE
- WHEN USING
- STOP USE
- KEEP OUT OF REACH OF CHILDREN
-
DOSAGE & ADMINISTRATION
Directions Apply liberally on dry skin following your morning routine at least 15 minutes before sun exposure For topical use only Keep out of reach of children Children under 6 months: ask a doctor Sun Protection Measures: Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 30 or higher and other sun protection measures including: Limit time in the sun, especially from 10 a.m. - 2 p.m. Wear long-sleeved shirts, pants, hats, and sunglasses. Reapply to dry skin: after 80 minutes of swimming or sweating immediately after towel drying at least every 2 hours
- OTHER SAFETY INFORMATION
-
INACTIVE INGREDIENT
Inactive Ingredients: Water (Aqua), Caprylic/Capric Triglyceride, Cetearyl Alcohol, Coconut Alkanes, Glycerin, Aloe Barbadensis Leaf Juice, Argania Spinosa Kernel Oil, Glyceryl Caprylate, Coco-Glucoside, Phaeodactylum Tricornutum Extract, Butyrospermum Parkii (Shea) Butter, Helianthus Annuus (Sunflower) Seed Oil, Disodium Tetramethylhexadecenyl Succinoyl Cysteine, Oryza Sativa Starch, Bisabolol, Dipotassium Glycyrrhizinate, Sodium Phytate, Xanthan Gum, Sclerotium Gum, Cetearyl Glucoside, Polyhydroxystearic Acid, Polyglyceryl-3 Polyricinoleate, Isostearic Acid, Lecithin, Dimethicone, Coco-Caprylate/Caprate, Caprylyl/Capryl Glucoside, Caprylhydroxamic Acid, Tocopherol, Beta-Sitosterol, Squalene, Triethoxycaprylylsilane
May Contain: Iron Oxide (CI 77491, CI77492, CI77499) - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
LIPID SHIELD SPF 30
zinc oxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 62742-4273 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 21 g in 100 g Inactive Ingredients Ingredient Name Strength GLYCERYL CAPRYLATE (UNII: TM2TZD4G4A) TOCOPHEROL (UNII: R0ZB2556P8) PHAEODACTYLUM TRICORNUTUM (UNII: Y5W63E7HLV) SHEA BUTTER (UNII: K49155WL9Y) DISODIUM TETRAMETHYLHEXADECENYL SUCCINOYL CYSTEINE (UNII: CZA8L664YS) COCONUT ALKANES (UNII: 1E5KJY107T) DIPOTASSIUM GLYCYRRHIZATE (UNII: CA2Y0FE3FX) CI 77499 (UNII: XM0M87F357) ALOE BARBADENSIS LEAF JUICE (UNII: RUE8E6T4NB) COCO-GLUCOSIDE (UNII: ICS790225B) HELIANTHUS ANNUUS (SUNFLOWER) SEED OIL (UNII: 3W1JG795YI) BISABOLOL (UNII: 24WE03BX2T) XANTHAN GUM (UNII: TTV12P4NEE) SCLEROTIUM GUM (UNII: 2X51AD1X3T) ISOSTEARIC ACID (UNII: X33R8U0062) LECITHIN, SOYBEAN (UNII: 1DI56QDM62) CAPRYLYL/CAPRYL GLUCOSIDE (UNII: E00JL9G9K0) CAPRYLIC/CAPRIC TRIGLYCERIDE (UNII: C9H2L21V7U) GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) CETEARYL ALCOHOL (UNII: 2DMT128M1S) CAPRYLHYDROXAMIC ACID (UNII: UPY805K99W) .BETA.-SITOSTEROL (UNII: S347WMO6M4) CI 77492 (UNII: EX438O2MRT) POLYGLYCERYL-3 PENTARICINOLEATE (UNII: 7Q0OK5DOT4) ORYZA SATIVA (RICE) STARCH (UNII: 4DGK8B7I3S) POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) CETEARYL GLUCOSIDE (UNII: 09FUA47KNA) ARGANIA SPINOSA KERNEL OIL (UNII: 4V59G5UW9X) SODIUM PHYTATE (UNII: 88496G1ERL) DIMETHICONE (UNII: 92RU3N3Y1O) COCO-CAPRYLATE/CAPRATE (UNII: 8D9H4QU99H) TRIETHOXYCAPRYLYLSILANE (UNII: LDC331P08E) CI 77491 (UNII: 1K09F3G675) SQUALENE (UNII: 7QWM220FJH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 62742-4273-2 1 in 1 CARTON 08/07/2025 1 NDC: 62742-4273-1 50 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 08/07/2025 Labeler - Allure Labs (926831603) Registrant - Allure Labs (926831603) Establishment Name Address ID/FEI Business Operations Allure Labs 926831603 manufacture(62742-4273)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.