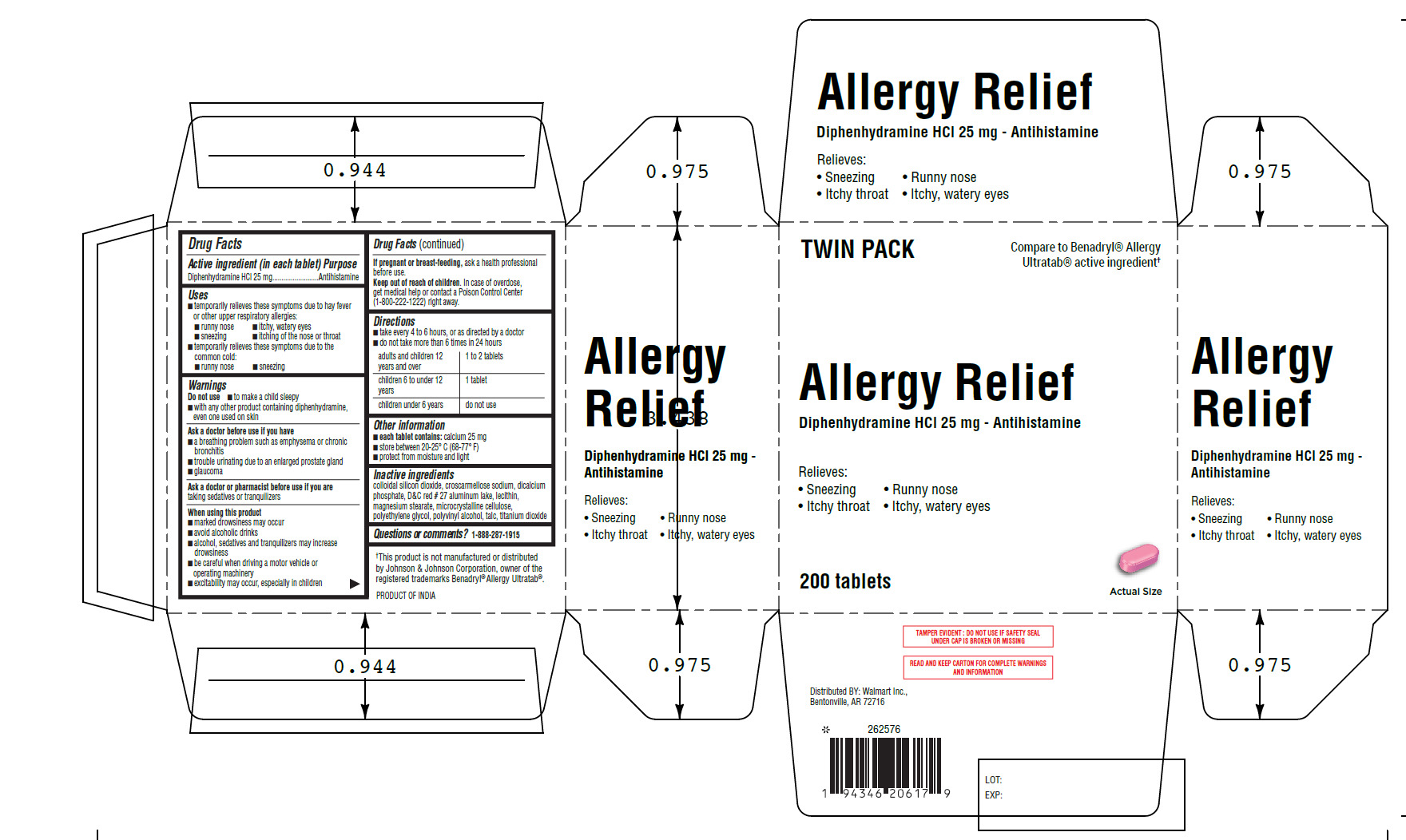
Equate Allergy Relief Tablets 25mg
ALLERGY RELIEF by
Drug Labeling and Warnings
ALLERGY RELIEF by is a Otc medication manufactured, distributed, or labeled by Walmart Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
ALLERGY RELIEF- diphenhydramine hydrochloride tablet, film coated
Walmart Inc.
----------
Equate Allergy Relief Tablets 25mg
Uses ■ temporarily relieves these symptoms due to hay fever
or other upper respiratory allergies:
■ runny nose ■ itchy, watery eyes
■ sneezing ■ itching of the nose or throat
■ temporarily relieves these symptoms due to the
common cold:
■ runny nose ■ sneezing
Do not use■ to make a child sleepy
■ with any other product containing diphenhydramine,
even one used on skin
Ask a doctor before use if you have
■ a breathing problem such as emphysema or chronic
bronchitis
■ trouble urinating due to an enlarged prostate gland
■ glaucoma
When using this product
■ marked drowsiness may occur
■ avoid alcoholic drinks
■ alcohol, sedatives and tranquilizers may increase
drowsiness
■ be careful when driving a motor vehicle or
operating machinery
■ excitability may occur, especially in children
Keep out of reach of children.In case of overdose,
get medical help or contact a Poison Control Center
(1-800-222-1222) right away.
Directions
■ take every 4 to 6 hours, or as directed by a doctor
■ do not take more than 6 times in 24 hours
adults and children 12 years and over 1 to 2 tablets
children 6 to under 12 years 1 tablet
children under 6 years do not use
Other information
■
each tablet contains:calcium 25 mg
■ store between 20-25° C (68-77° F)
■ protect from moisture and light
| ALLERGY RELIEF
diphenhydramine hydrochloride tablet, film coated |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
| Labeler - Walmart Inc. (051957769) |
Revised: 1/2026
<
Document Id: 497415bd-68d9-2c4d-e063-6394a90a650f
Set id: 3d4902aa-be01-4b0b-af13-27c9424fa917
Version: 3
Effective Time: 20260128
Trademark Results [ALLERGY RELIEF]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 ALLERGY RELIEF 98236984 not registered Live/Pending |
Dmytro Kononenko 2023-10-24 |
 ALLERGY RELIEF 90457167 not registered Live/Pending |
American Textile Company, Inc. 2021-01-10 |
 ALLERGY RELIEF 78838437 3358249 Live/Registered |
Meshbesher Health Corporation 2006-03-16 |
 ALLERGY RELIEF 76619855 3066888 Live/Registered |
AMERICAN TEXTILE COMPANY 2004-11-09 |
 ALLERGY RELIEF 74668018 not registered Dead/Abandoned |
NaturaLife Corporation 1995-05-01 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.