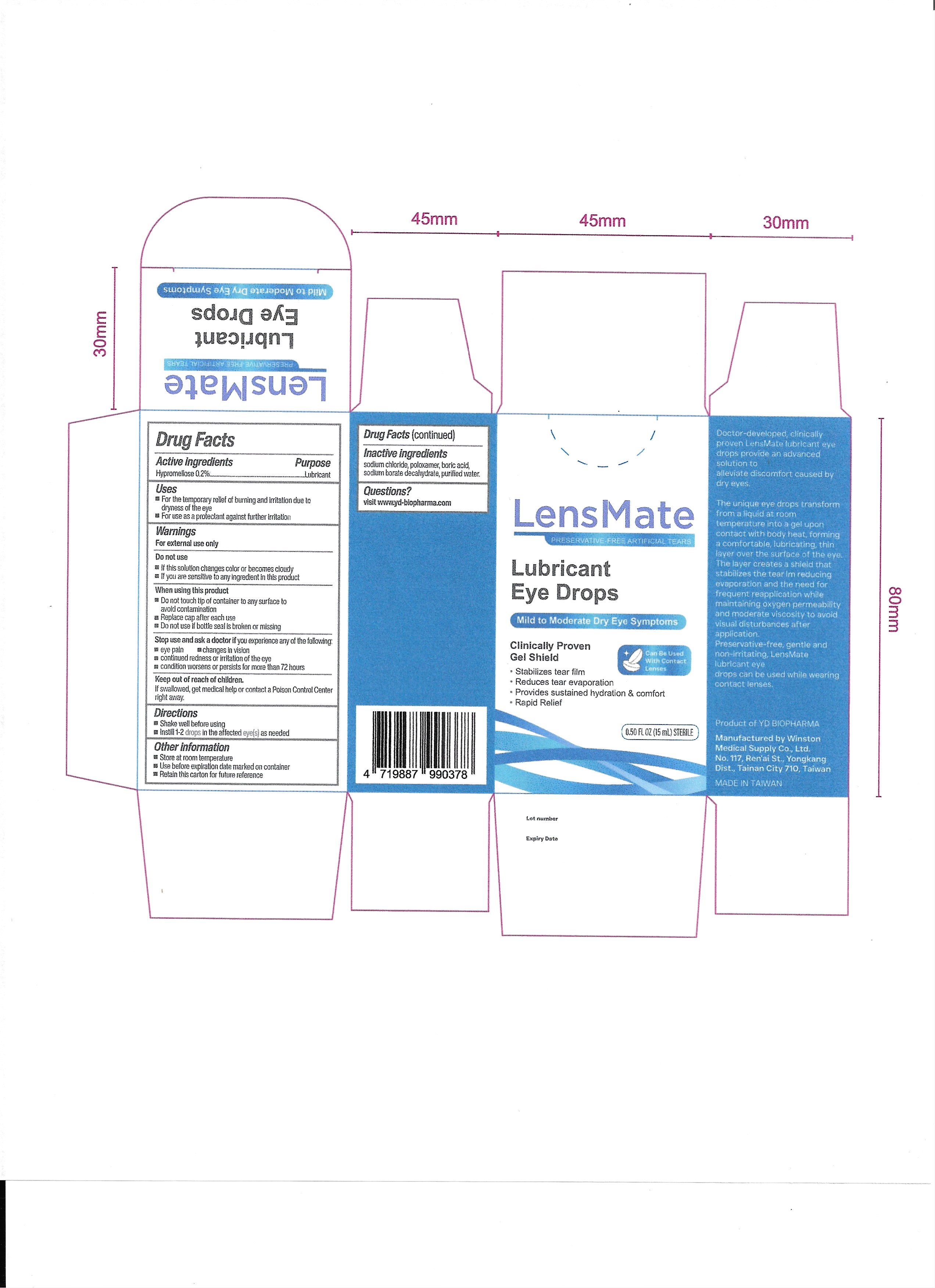
LensMate Preservative-Free Artificial Tears by YD BIO USA, INC. / WINSTON MEDICAL SUPPLY CO., LTD. 001-01
LensMate Preservative-Free Artificial Tears by
Drug Labeling and Warnings
LensMate Preservative-Free Artificial Tears by is a Otc medication manufactured, distributed, or labeled by YD BIO USA, INC., WINSTON MEDICAL SUPPLY CO., LTD.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
LENSMATE PRESERVATIVE-FREE ARTIFICIAL TEARS- lubricant eye drops liquid
YD BIO USA, INC.
----------
001-01
Stop use and ask a doctor if you experience any of the following:
Eye pain
Changes in vision
Continued redness or irritation of the eye
Condition worsens or persists for more than 72 hours
If this solution changes color or becomes cloudy
If you are sensitive to any ingredient in this product
Stop use and ask a doctor if you experience any of the following:
eye pain
Changes in vision
Continued redness or irritation of the eye
Condition worsens or persists for more than 72 hours
Do not touch tip of container to any surface to avoid contamination
Replace cap after each use
Do not use if bottle seal is broken or missing
Store at room temperature
Use before expiration date marked on container
Retain this carton for future reference
| LENSMATE PRESERVATIVE-FREE ARTIFICIAL TEARS
lubricant eye drops liquid |
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
|
||||||||||||||||||||
| Labeler - YD BIO USA, INC. (118330659) |
| Registrant - YD BIO USA, INC. (118330659) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| WINSTON MEDICAL SUPPLY CO., LTD. | 656080751 | manufacture(87106-001) | |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.