NASALASALT- sodium chloride spray
NasalAsalt by
Drug Labeling and Warnings
NasalAsalt by is a Otc medication manufactured, distributed, or labeled by Nasal and Sinus Health, Inc., Gerolymatos International. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- USES
-
DIRECTIONS
Tilt head forward, spray 2-3 times in each nostril directing spray upward and outward toward ear and away from center of nose. Blow nose. For best results, see package insert for the Vanderpool Technique™ for a more thorough nasal cleansing procedure or go to the NasalAsalt® web site for a video demonstration.
- WARNINGS
- PRECAUTIONS
- ACTIVE INGREDIENT
- SPL UNCLASSIFIED SECTION
-
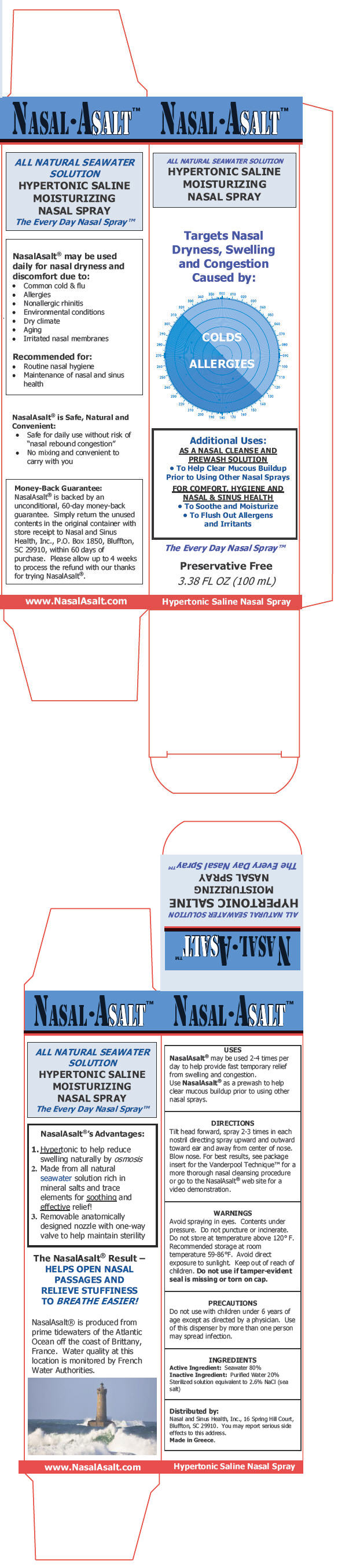
PRINCIPAL DISPLAY PANEL - 100 mL Canister Carton
NASALASALT™
ALL NATURAL SEAWATER SOLUTION
HYPERTONIC SALINE
MOISTURIZING
NASAL SPRAYTargets Nasal
Dryness, Swelling
and Congestion
Caused by:COLDS
ALLERGIESAdditional Uses:
AS A NASAL CLEANSE AND
PREWASH SOLUTION-
To Help Clear Mucous Buildup
Prior to Using Other Nasal Sprays
FOR COMFORT, HYGIENE AND
NASAL & SINUS HEALTH- To Soothe and Moisturize
-
To Flush Out Allergens
and Irritants
The Every Day Nasal Spray™
Preservative Free
3.38 FL OZ (100 mL)
Hypertonic Saline Nasal Spray
-
To Help Clear Mucous Buildup
-
INGREDIENTS AND APPEARANCE
NASALASALT ALL-NATURAL SEAWATER
sodium chloride sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 52429-123 Route of Administration NASAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Sodium Chloride (UNII: 451W47IQ8X) (Sodium Cation - UNII:LYR4M0NH37) Sodium Chloride 2600 mg in 100 mL Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 52429-123-01 1 in 1 BOX 1 100 mL in 1 CANISTER Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part349 11/01/2011 Labeler - Nasal and Sinus Health, Inc. (968415740) Establishment Name Address ID/FEI Business Operations Nasal and Sinus Health, Inc. 968415740 RELABEL Establishment Name Address ID/FEI Business Operations Gerolymatos International 423075886 MANUFACTURE
Trademark Results [NasalAsalt]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 NASALASALT 77739957 3783601 Dead/Cancelled |
Innovative Consulting, Inc. 2009-05-19 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.