CHLORDIAZEPOXIDE HYDROCHLORIDE AND CLIDINIUM BROMIDE capsule
Chlordiazepoxide Hydrochloride and Clidinium Bromide by
Drug Labeling and Warnings
Chlordiazepoxide Hydrochloride and Clidinium Bromide by is a Prescription medication manufactured, distributed, or labeled by Xiromed, LLC, Misemer Pharmaceuticals, Inc., Belcher Pharmaceuticals, LLC. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
BOXED WARNING
(What is this?)
WARNING: RISKS FROM CONCOMITANT USE WITH OPIOIDS; ABUSE, MISUSE, AND ADDICTION; and DEPENDENCE AND WITHDRAWAL REACTIONS
- Concomitant use of benzodiazepines and opioids may result in profound sedation, respiratory depression, coma, and death. Reserve concomitant prescribing of these drugs in patients for whom alternative treatment options are inadequate. Limit dosages and durations to the minimum required. Follow patients for signs and symptoms of respiratory depression and sedation (see WARNINGS and PRECAUTIONS and PRECAUTIONS, Drug Interactions).
- The use of benzodiazepines, including chlordiazepoxide hydrochloride, a component of chlordiazepoxide hydrochloride and clidinium bromide capsules, exposes users to risks of abuse, misuse, and addiction, which can lead to overdose or death. Abuse and misuse of benzodiazepines commonly involve concomitant use of other medications, alcohol, and/ or illicit substances, which is associated with an increased frequency of serious adverse outcomes. Before prescribing chlordiazepoxide hydrochloride and clidinium bromide capsules and throughout treatment, assess each patient’s risk for abuse, misuse, and addiction (see WARNINGS).
- The continued use of benzodiazepines, including chlordiazepoxide hydrochloride and clidinium bromide dependence. The risks of dependence and withdrawal increase with longer treatment duration and higher daily dose. Abrupt discontinuation or rapid dosage reduction of chlordiazepoxide hydrochloride and clidinium bromide capsules after continued use may precipitate acute withdrawal reactions, which can be life-threatening. To reduce the risk of withdrawal reactions, use a gradual taper to discontinue chlordiazepoxide hydrochloride and clidinium bromide capsules or reduce the dosage (see WARNINGS and DOSAGE AND ADMINISTRATION).
-
DESCRIPTION
Chlordiazepoxide hydrochloride and clidinium bromide capsules are a fixed-combination of chlordiazepoxide hydrochloride, a benzodiazepine, and clidinium bromide, an anticholinergic.
Each capsule contains the active ingredients 5 mg chlordiazepoxide hydrochloride and 2.5 mg clidinium bromide. Each capsule also contains the inactive ingredients lactose monohydrate, pregelatinized starch, talc. Gelatin capsule shells may contain titanium dioxide with the following dye systems: D&C Yellow #10, FD&C Green #3 and gelatin. Imprinting ink is composed of shellac, propylene glycol, black iron oxide, strong ammonia solution and potassium hydroxide.
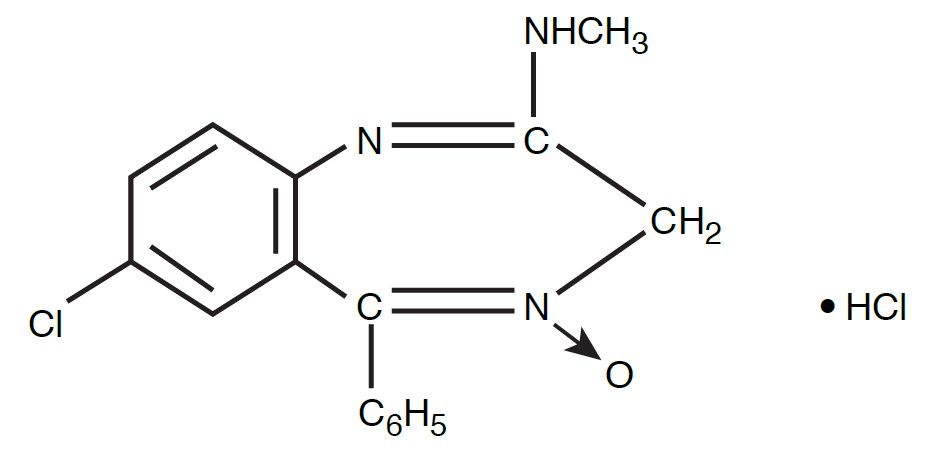
Chlordiazepoxide hydrochloride is 7-chloro-2-methylamino- 5-phenyl-3H-1,4-benzodiazepine 4-oxide hydrochloride. A colorless, crystalline substance, it is soluble in water. It is unstable in solution and the powder must be protected from light. The molecular weight is 336.22. The structural formula of chlordiazepoxide hydrochloride is as follows:
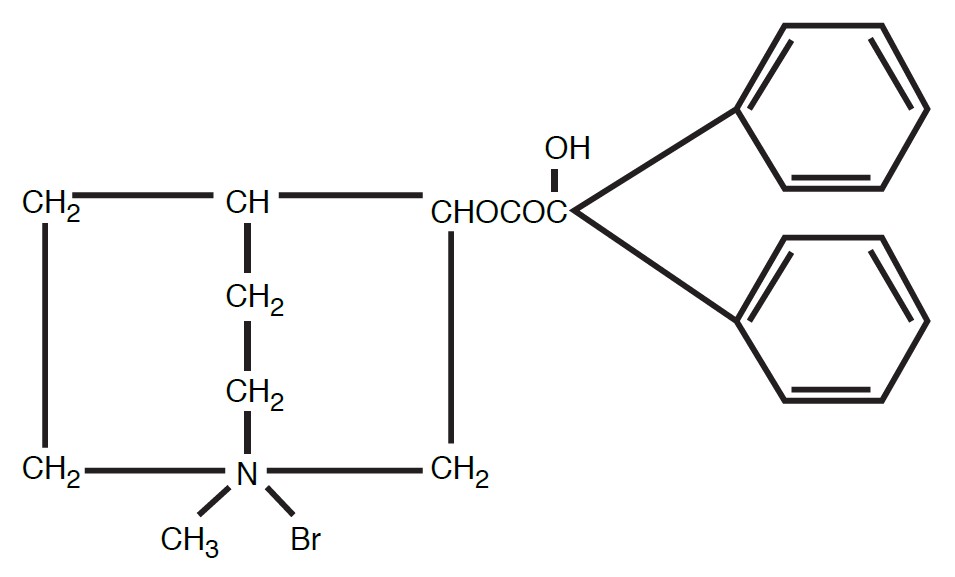
Clidinium bromide is a synthetic anticholinergic agent which has been shown in experimental and clinical studies to have a antispasmodic and antisecretory effects on the gastrointestinal tract.
Structurally clidinium bromide is:
-
ANIMAL PHARMACOLOGY
Chlordiazepoxide hydrochloride has been studied extensively in many species of animals and these studies are suggestive of action on the limbic system of the brain, which recent evidence indicates is involved in emotional responses. Hostile monkeys were made tame by oral drug doses, which did not cause sedation. Chlordiazepoxide hydrochloride revealed a “taming-action with the elimination of fear and aggression”. The taming effect of chlordiazepoxide hydrochloride was further demonstrated in rats made vicious by lesions in the septal area of the brain. The drug dosage which effectively blocked the vicious reaction was well below the dose which caused sedation in these animals.
The oral LD50 of single doses of chlordiazepoxide hydrochloride, calculated according to the method of Miller and Tainter, is 720 ± 51 mg/kg as determined in mice observed over a period of 5 days following dosage.
Clidinium bromide is an effective anticholinergic agent with activity approximating that of atropine sulfate against acetylcholineinduced spasms in isolated intestinal strips. On oral administration in mice, it proved an effective antisialagogue in preventing pilocarpine- induced salivation. Spontaneous intestinal motility in both rats and dogs is reduced following oral dosing with 0.1 to 0.25 mg/kg. Potent cholinergic ganglionic blocking effects (vagal) were produced with intravenous usage in anesthetized dogs.
Oral doses of 2.5 mg/kg to dogs produced signs of nasal dryness and slight pupillary dilation. In two other species, monkeys and rabbits, doses of 5 mg/kg, po, given three times daily for 5 days did not produce apparent secretory or visual changes.
The oral LD50 of single doses of clidinium bromide is 860 ± 57 mg/kg as determined in mice observed over a period of 5 days following dosage; the calculations were made according to the method of Miller and Tainter.
Effects on Reproduction
Effects on Reproduction:
Reproduction studies in rats fed chlordiazepoxide hydrochloride, 10, 20 and 80 mg/kg daily, and bred through one or two matings showed no congenital anomalies, nor were there adverse effects on lactation of the dams or growth of the newborn. However, in another study at 100 mg/kg daily there was noted a significant decrease in the fertilization rate and a marked decrease in the viability and body weight of offspring which may be attributable to sedative activity, thus resulting in lack of interest in mating and lessened maternal nursing and care of the young. One neonate in each of the first and second matings in the rat reproduction study at the 100 mg/kg dose exhibited major skeletal defects. Further studies are in progress to determine the significance of these findings.
Two series of reproduction experiments with clidinium bromide were carried out in rats, employing dosages of 2.5 and 10 mg/ kg daily in each experiment. In the first experiment, clidinium bromide was administered for a 9-week interval prior to mating; no untoward effect on fertilization or gestation was noted. The offspring were taken by caesarean section and did not show a significant incidence of congenital anomalies when compared to control animals. In the second experiment, adult animals were given clidinium bromide for 10 days prior to and through two mating cycles. No significant effects were observed on fertility, gestation, viability of offspring or lactation, as compared to control animals, nor was there a significant incidence of congenital anomalies in the offspring derived from these experiments.
A reproduction study of Chlordiazepoxide hydrochloride and clidinium bromide capsules was carried out in rats through two successive matings. Oral daily doses were administered in two concentrations: 2.5 mg/kg chlordiazepoxide hydrochloride with 1.25 mg/kg clidinium bromide or 25 mg/kg chlordiazepoxide hydrochloride with 12.5 mg/kg clidinium bromide. In the first mating, no significant differences were noted between the control or the treated groups, with the exception of a slight decrease in the number of animals surviving during lactation among those receiving the highest dosage. As with all anticholinergic drugs, an inhibiting effect on lactation may occur. In the second mating, similar results were obtained except for a slight decrease in the number of pregnant females and in the percentage of offspring surviving until weaning. No congenital anomalies were observed in both matings in either the control or treated groups. Additional animal reproduction studies are in progress.
-
INDICATIONS AND USAGE
Chlordiazepoxide hydrochloride and clidinium bromide capsules is indicated to control emotional and somatic factors in gastrointestinal disorders. Chlordiazepoxide hydrochloride and clidinium bromide capsules may also be used as adjunctive therapy in the treatment of peptic ulcer and in the treatment of the irritable bowel syndrome (irritable colon, spastic colon, mucous colitis) and acute enterocolitis.
-
CONTRAINDICATIONS
Chlordiazepoxide hydrochloride and clidinium bromide capsules are contraindicated in the presence of glaucoma (since the anticholinergic component may produce some degree of mydriasis) and in patients with prostatic hypertrophy and benign bladder neck obstruction. It is contraindicated in patients with known hypersensitivity to chlordiazepoxide hydrochloride and/or clidinium bromide.
-
WARNINGS
Risks From Concomitant Use with Opioids
Concomitant use of benzodiazepines, including chlordiazepoxide hydrochloride and clidinium bromide capsules, and opioids may result in profound sedation, respiratory depression, coma, and death. Because of these risks, reserve concomitant prescribing of these drugs—in patients for whom alternative treatment options are inadequate.
Observational studies have demonstrated that concomitant use of opioid analgesics and benzodiazepines increases the risk of drugrelated mortality compared to use of opioids alone. If a decision is made to prescribe chlordiazepoxide hydrochloride and clidinium bromide capsules concomitantly with opioids, prescribe the lowest effective dosages and minimum durations of concomitant use, and follow patients closely for signs and symptoms of respiratory depression and sedation. Advise both patients and caregivers about the risks of respiratory depression and sedation when chlordiazepoxide hydrochloride and clidinium bromide capsules is used with opioids (see PRECAUTIONS).
Abuse, Misuse, and Addiction
The use of benzodiazepines, including chlordiazepoxide hydrochloride, a component of chlordiazepoxide hydrochloride and clidinium bromide capsules, exposes users to the risks of abuse, misuse, and addiction, which can lead to overdose or death. Abuse and misuse of benzodiazepines often (but not always) involve the use of doses greater than the maximum recommended dosage and commonly involve concomitant use of other medications, alcohol, and/or illicit substances, which is associated with an increased frequency of serious adverse outcomes, including respiratory depression, overdose, or death (see DRUG ABUSE AND DEPENDENCE).
Before prescribing chlordiazepoxide hydrochloride and clidinium bromide capsules and throughout treatment, assess each patient’s risk for abuse, misuse, and addiction (e.g., using a standardized screening tool). Use of chlordiazepoxide hydrochloride and clidinium bromide capsules, particularly in patients at elevated risk, necessitates counseling about the risks and proper use of chlordiazepoxide hydrochloride and clidinium bromide capsules along with monitoring for signs and symptoms of abuse, misuse, and addiction. Prescribe the lowest effective dosage; avoid or minimize concomitant use of CNS depressants and other substances associated with abuse, misuse, and addiction (e.g., opioid analgesics, stimulants); and advise patients on the proper disposal of unused drug. If a substance use disorder is suspected, evaluate the patient and institute (or refer them for) early treatment, as appropriate.
Dependence and Withdrawal Reactions
To reduce the risk of withdrawal reactions, use a gradual taper to discontinue chlordiazepoxide hydrochloride and clidinium bromide capsules or reduce the dosage (a patient-specific plan should be used to taper the dosage) (see DOSAGE AND ADMINISTRATION).
Patients at an increased risk of withdrawal adverse reactions after benzodiazepine discontinuation or rapid dosage reduction include those who take higher dosages, and those who have had longer durations of use.
Acute Withdrawal Reactions
The continued use of benzodiazepines, including chlordiazepoxide hydrochloride and clidinium bromide capsules, may lead to clinically significant physical dependence. Abrupt discontinuation or rapid dosage reduction of chlordiazepoxide hydrochloride and clidinium bromide capsules after continued use, or administration of flumazenil (a benzodiazepine antagonist) may precipitate acute withdrawal reactions, which can be life-threatening (e.g., seizures) (see DRUG ABUSE AND DEPENDENCE).
Protracted Withdrawal Syndrome
In some cases, benzodiazepine users have developed a protracted withdrawal syndrome with withdrawal symptoms lasting weeks to more than 12 months (see DRUG ABUSE AND DEPENDENCE).
Effects on the Ability to Drive or Operate Machinery
As in the case of other preparations containing CNS-acting drugs, patients receiving chlordiazepoxide hydrochloride and clidinium bromide capsules should be cautioned about possible combined effects with opioids, alcohol and other CNS depressants. For the same reason, they should be cautioned against hazardous occupations requiring complete mental alertness, such as operating machinery or driving a motor vehicle.
Usage in Pregnancy
An increased risk of congenital malformations associated with the use of minor tranquilizers (chlordiazepoxide, diazepam and meprobamate) during the first trimester of pregnancy has been suggested in several studies. Because use of these drugs is rarely a matter of urgency, their use during this period should almost always be avoided. The possibility that a woman of childbearing potential may be pregnant at the time of institution of therapy should be considered. Patients should be advised that if they become pregnant during therapy or intend to become pregnant they should communicate with their physicians about the desirability of discontinuing the drug.
As with all anticholinergic drugs, an inhibiting effect on lactation may occur (see ANIMAL PHARMACOLOGY).
-
OVERDOSAGE
Manifestations of chlordiazepoxide hydrochloride overdosage include somnolence, confusion, coma and diminished reflexes. Respiration, pulse and blood pressure should be monitored, as in all cases of drug overdosage, although, in general, these effects have been minimal following chlordiazepoxide hydrochloride overdosage.
While the signs and symptoms of chlordiazepoxide hydrochloride and clidinium bromide capsules overdosage may be produced by either of its components, usually such symptoms will be overshadowed by the anticholinergic actions of clidinium bromide. The symptoms of overdosage of clidinium bromide are excessive dryness of mouth, blurring of vision, urinary hesitancy and constipation.
General supportive measures should be employed, along with immediate gastric lavage. Administer physostigmine 0.5 to 2 mg at a rate of no more than 1 mg per minute. This may be repeated in 1 to 4 mg doses if arrhythmias, convulsions or deep coma recur.
Intravenous fluids should be administered and an adequate airway maintained. Hypotension may be combated by the use of levarterenol or metaraminol.
Methylphenidate or caffeine and sodium benzoate may be given to combat CNS-depressive effects.
Dialysis is of limited value. Should excitation occur, barbiturates should not be used. As with the management of intentional overdosage with any drug, it should be borne in mind that multiple agents may have been ingested.
Withdrawal symptoms of the barbiturate type have occurred after the discontinuation of benzodiazepines (see DRUG ABUSE AND DEPENDENCE).
-
PRECAUTIONS
In debilitated patients, it is recommended that the dosage be limited to the smallest effective amount to preclude the development of ataxia, oversedation or confusion (not more than 2 Chlordiazepoxide Hydrochloride and Clidinium Bromide capsules per day initially, to be increased gradually as needed and tolerated). In general, the concomitant administration of Chlordiazepoxide Hydrochloride and Clidinium Bromide and other psychotropic agents is not recommended. If such combination therapy seems indicated, careful consideration should be given to the pharmacology of the agents to be employed - particularly when the known potentiating compounds such as the MAO inhibitors and phenothiazines are to be used. The usual precautions in treating patients with impaired renal or hepatic function should be observed.
Paradoxical reactions to chlordiazepoxide hydrochloride, e.g., excitement, stimulation and acute rage, have been reported in psychiatric patients and should be watched for during Chlordiazepoxide Hydrochloride and Clidinium Bromide therapy. The usual precautions are indicated when chlordiazepoxide hydrochloride is used in the treatment of anxiety states where there is any evidence of impending depression; it should be borne in mind that suicidal tendencies may be present and protective measures may be necessary. Although clinical studies have not established a cause and effect relationship, physicians should be aware that variable effects on blood coagulation have been reported very rarely in patients receiving oral anticoagulants and chlordiazepoxide hydrochloride.
CNS Adverse Reactions
In geriatric or debilitated patients, it is recommended that the dosage be limited to the smallest effective amount to preclude the development of ataxia, oversedation or confusion (not more than 2 chlordiazepoxide hydrochloride and clidinium bromide capsules per day initially, to be increased gradually as needed and tolerated). In general, the concomitant administration of chlordiazepoxide hydrochloride and clidinium bromide capsules and other psychotropic agents is not recommended. If such combination therapy seems indicated, careful consideration should be given to the pharmacology of the agents to be employed— particularly when the known potentiating compounds such as the MAO inhibitors and phenothiazines are to be used. The usual precautions in treating patients with impaired renal or hepatic function should be observed.
Paradoxical reactions to chlordiazepoxide hydrochloride, e.g., excitement, stimulation and acute rage, have been reported in psychiatric patients and should be watched for during chlordiazepoxide hydrochloride and clidinium bromide capsules therapy. The usual precautions are indicated when chlordiazepoxide hydrochloride is used in the treatment of anxiety states where there is any evidence of impending depression; it should be borne in mind that suicidal tendencies may be present and protective measures may be necessary.
Information for Patients
Abuse, Misuse, and Addiction
Inform patients that the use of chlordiazepoxide hydrochloride and clidinium bromide capsules, even at recommended dosages, exposes users to risks of abuse, misuse, and addiction, which can lead to overdose and death, especially when used in combination with other medications (e.g., opioid analgesics), alcohol, and/or illicit substances. Inform patients about the signs and symptoms of benzodiazepine abuse, misuse, and addiction; to seek medical help if they develop these signs and/or symptoms; and on the proper disposal of unused drug (see WARNINGS).
Withdrawal Reactions
Inform patients that the continued use of chlordiazepoxide hydrochloride and clidinium bromide capsules may lead to clinically significant physical dependence and that abrupt discontinuation or rapid dosage reduction of chlordiazepoxide hydrochloride and clidinium bromide capsules may precipitate acute withdrawal reactions, which can be life-threatening. Inform patients that in some cases, patients taking benzodiazepines have developed a protracted withdrawal syndrome with withdrawal symptoms lasting weeks to more than 12 months. Instruct patients that discontinuation or dosage reduction of chlordiazepoxide hydrochloride and clidinium bromide capsules may require a slow taper (see WARNINGS and DRUG ABUSE AND DEPENDENCE).
Concomitant Use With Opioids and Other CNS Depressants
Inform patients and caregivers that potentially fatal additive effects may occur if chlordiazepoxide hydrochloride and clidinium bromide capsules are used with opioids or other CNS depressants, including alcohol, and not to use these concomitantly unless supervised by a health care provider (see1 and PRECAUTIONS, Drug Interactions).
- 1 WARNINGS
Drug Interactions
Opioids
The concomitant use of benzodiazepines, including chlordiazepoxide hydrochloride, a component of chlordiazepoxide hydrochloride and clidinium bromide capsules, and opioids increases the risk of respiratory depression because of actions at different receptor sites in the CNS that control respiration.
Benzodiazepines interact at GABAA sites and opioids interact primarily at mu receptors. When benzodiazepines and opioids are combined, the potential for benzodiazepines to significantly worsen opioid-related respiratory depression exists. Limit dosage and duration of concomitant use of chlordiazepoxide hydrochloride and clidinium bromide capsules and opioids, and follow patients closely for respiratory depression and sedation.
Oral Anticoagulants
Although clinical studies have not established a cause and effect relationship, physicians should be aware that variable effects on blood coagulation have been reported very rarely in patients receiving oral anticoagulants and chlordiazepoxide hydrochloride, a component of chlordiazepoxide hydrochloride and clidinium bromide capsules.
Geriatric Use
Geriatric subjects may be particularly prone to experiencing drowsiness, ataxia and confusion while receiving chlordiazepoxide hydrochloride and clidinium bromide capsules. These effects can usually be avoided with proper dosage adjustment, although they have occasionally been observed even at the lower dosage ranges. Dosing in geriatric subjects should be initiated cautiously (no more than 2 capsules per day) and increased gradually if needed and tolerated (see DOSAGE AND ADMINISTRATION). Chlordiazepoxide hydrochloride and clidinium bromide capsules are contraindicated in the presence of glaucoma, prostatic hypertrophy and benign bladder neck obstruction (see CONTRAINDICATIONS).
-
ADVERSE REACTIONS
No side effects or manifestations not seen with either compound alone have been reported with the administration of chlordiazepoxide hydrochloride and clidinium bromide capsules. However, since chlordiazepoxide hydrochloride and clidinium bromide capsules contain chlordiazepoxide hydrochloride and clidinium bromide, the possibility of untoward effects which may be seen with either of these two compounds cannot be excluded.
When chlordiazepoxide hydrochloride has been used alone the necessity of discontinuing therapy because of undesirable effects has been rare. Drowsiness, ataxia and confusion have been reported in some patients—particularly the elderly and debilitated. While these effects can be avoided in almost all instances by proper dosage adjustment, they have occasionally been observed at the lower dosage ranges. In a few instances syncope has been reported.
Other adverse reactions reported during therapy with chlordiazepoxide hydrochloride include isolated instances of skin eruptions, edema, minor menstrual irregularities, nausea and constipation, extrapyramidal symptoms, as well as increased and decreased libido. Such side effects have been infrequent and are generally controlled with reduction of dosage. Changes in EEG patterns (low-voltage fast activity) have been observed in patients during and after chlordiazepoxide hydrochloride treatment.
Blood dyscrasias, including agranulocytosis, jaundice and hepatic dysfunction have occasionally been reported during therapy with chlordiazepoxide hydrochloride. When chlordiazepoxide hydrochloride treatment is protracted, periodic blood counts and liver function tests are advisable.
Adverse effects reported with use of Chlordiazepoxide hydrochloride and clidinium bromide capsules are those typical of anticholinergic agents, i.e., dryness of the mouth, blurring of vision, urinary hesitancy and constipation.
Constipation has occurred most often when Chlordiazepoxide hydrochloride and clidinium bromide capsules therapy has been combined with other spasmolytic agents and/or a low residue diet.
To report SUSPECTED ADVERSE REACTIONS, contact Xiromed, LLC at 1-844-XIROMED (1-844-947-6633) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
-
DRUG ABUSE AND DEPENDENCE
Withdrawal symptoms, similar in character to those noted with barbiturates and alcohol (convulsions, tremor, abdominal and muscle cramps, vomiting and sweating), have occurred following abrupt discontinuance of chlordiazepoxide. The more severe withdrawal symptoms have usually been limited to those patients who had received excessive doses over an extended period of time. General milder withdrawal symptoms (e.g., dysphoria and insomnia) have been reported following abrupt discontinuance of benzodiazepines taken continuously at therapeutic levels for several months. Consequently, after extended therapy, abrupt discontinuation should generally be avoided and a gradual dosage tapering schedule followed. Addiction-prone individuals (such as drug addicts or alcoholics) should be under careful surveillance when receiving chlordiazepoxide or other psychotropic agents because of the predisposition of such patients to habituation and dependence.
Controlled Substance
Chlordiazepoxide hydrochloride and clidinium bromide capsules contain chlordiazepoxide hydrochloride, a Schedule IV controlled substance and clidinium bromide, which is not a controlled substance. Chlordiazepoxide hydrochloride and clidinium bromide capsules are exempted from Schedule IV and are not controlled under the Controlled Substances Act.
Abuse
Chlordiazepoxide hydrochloride, a component of chlordiazepoxide hydrochloride and clidinium bromide capsules, is a CNS depressant with a potential for abuse and addiction. Abuse is the intentional, non-therapeutic use of a drug, even once, for its desirable psychological or physiological effects. Misuse is the intentional use, for therapeutic purposes, of a drug by an individual in a way other than prescribed by a health care provider or for whom it was not prescribed. Drug addiction is a cluster of behavioral, cognitive, and physiological phenomena that may include a strong desire to take the drug, difficulties in controlling drug use (e.g., continuing drug use despite harmful consequences, giving a higher priority to drug use than other activities and obligations), and possible tolerance or physical dependence. Even taking benzodiazepines as prescribed may put patients at risk for abuse and misuse of their medication. Abuse and misuse of benzodiazepines may lead to addiction.
Abuse and misuse of benzodiazepines often (but not always) involve the use of doses greater than the maximum recommended dosage and commonly involve concomitant use of other medications, alcohol, and/or illicit substances, which is associated with an increased frequency of serious adverse outcomes, including respiratory depression, overdose, or death. Benzodiazepines are often sought by individuals who abuse drugs and other substances, and by individuals with addictive disorders (see WARNINGS).
The following adverse reactions have occurred with benzodiazepine abuse and/or misuse: abdominal pain, amnesia, anorexia, anxiety, aggression, ataxia, blurred vision, confusion, depression, disinhibition, disorientation, dizziness, euphoria, impaired concentration and memory, indigestion, irritability, muscle pain, slurred speech, tremors, and vertigo.
The following severe adverse reactions have occurred with benzodiazepine abuse and/or misuse: delirium, paranoia, suicidal ideation and behavior, seizures, coma, breathing difficulty, and death. Death is more often associated with polysubstance use (especially benzodiazepines with other CNS depressants such as opioids and alcohol).
Dependence
Physical Dependence
Chlordiazepoxide hydrochloride and clidinium bromide capsules may produce physical dependence from continued therapy. Physical dependence is a state that develops as a result of physiological adaptation in response to repeated drug use, manifested by withdrawal signs and symptoms after abrupt discontinuation or a significant dose reduction of a drug. Abrupt discontinuation or rapid dosage reduction of benzodiazepines or administration of flumazenil, a benzodiazepine antagonist, may precipitate acute withdrawal reactions, including seizures, which can be life-threatening. Patients at an increased risk of withdrawal adverse reactions after benzodiazepine discontinuation or rapid dosage reduction include those who take higher dosages (i.e., higher and/or more frequent doses) and those who have had longer durations of use (see WARNINGS).
To reduce the risk of withdrawal reactions, use a gradual taper to discontinue chlordiazepoxide hydrochloride and clidinium bromide capsules or reduce the dosage (see WARNINGS and DOSAGE AND ADMINISTRATION).
Acute Withdrawal Signs and Symptoms
Acute withdrawal signs and symptoms associated with benzodiazepines have included abnormal involuntary movements, anxiety, blurred vision, depersonalization, depression, derealization, dizziness, fatigue, gastrointestinal adverse reactions (e.g., nausea, vomiting, diarrhea, weight loss, decreased appetite), headache, hyperacusis, hypertension, irritability, insomnia, memory impairment, muscle pain and stiffness, panic attacks, photophobia, restlessness, tachycardia, and tremor. More severe acute withdrawal signs and symptoms, including life-threatening reactions, have included catatonia, convulsions, delirium tremens, depression, hallucinations, mania, psychosis, seizures and suicidality.
Protracted Withdrawal Syndrome
Protracted withdrawal syndrome associated with benzodiazepines is characterized by anxiety, cognitive impairment, depression, insomnia, formication, motor symptoms (e.g., weakness, tremor, muscle twitches), paresthesia, and tinnitus that persists beyond 4 to 6 weeks after initial benzodiazepine withdrawal. Protracted withdrawal symptoms may last weeks to more than 12 months. As a result, there may be difficulty in differentiating withdrawal symptoms from potential re-emergence or continuation of symptoms for which the benzodiazepine was being used.
Tolerance
Tolerance to chlordiazepoxide hydrochloride and clidinium bromide capsules may develop from continued therapy. Tolerance is a physiological state characterized by a reduced response to a drug after repeated administration (i.e., a higher dose of a drug is required to produce the same effect that was once obtained at a lower dose). Tolerance to the therapeutic effects of chlordiazepoxide hydrochloride and clidinium bromide capsules may develop; however, little tolerance develops to the amnestic reactions and other cognitive impairments caused by benzodiazepines.
-
DOSAGE AND ADMINISTRATION
Recommended Dosage
Because of the varied individual responses to tranquilizers and anticholinergics, the optimum dosage of chlordiazepoxide hydrochloride and clidinium bromide capsules varies with the diagnosis and response of the individual patient. The dosage, therefore, should be individualized for maximum beneficial effects. The usual maintenance dose is 1 or 2 capsules, 3 or 4 times a day administered before meals and at bedtime.
Recommended Geriatric Dosage
Dosage should be limited to the smallest effective amount to preclude the development of ataxia, oversedation or confusion. The initial dose should not exceed 2 chlordiazepoxide hydrochloride and clidinium bromide capsules per day, to be increased gradually as needed and tolerated. Elderly patients have an increased risk of dose-related adverse reactions (see PRECAUTIONS).
Discontinuation or Dosage Reduction of Chlordiazepoxide Hydrochloride and Clidinium Bromide Capsules
To reduce the risk of withdrawal reactions, use a gradual taper to discontinue chlordiazepoxide hydrochloride and clidinium bromide capsules or reduce the dosage. If a patient develops withdrawal reactions, consider pausing the taper or increasing the dosage to the previous tapered dosage level. Subsequently decrease the dosage more slowly (see WARNINGS and DRUGABUSE AND DEPENDENCE).
-
HOW SUPPLIED
Chlordiazepoxide hydrochloride and clidinium bromide capsules are available in light green opaque capsules, each containing 5 mg chlordiazepoxide hydrochloride and 2.5 mg clidinium bromide in bottles of 100 capsules (NDC: 70700-185-01), with “VP302” imprinted on the cap of the capsule.
Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F).
Keep out of reach of children.
Dispense in tight, light-resistant container as defined in USP/NF.Manufactured for:
Xiromed, LLC
Florham Park, NJ 07932Made in the USA
PI-185-00 Rev. 03/2021
L41I-XIR, R-2103 -
MEDICATION GUIDE
This Medication Guide has been approved by the U.S. Food and Drug Administration.
PIL-185-00 Rev. 03/2021
L41I-XIR, R-2103MEDICATION GUIDE
Chlordiazepoxide Hydrochloride and Clidinium Bromide Capsules
(klor´´ dye az´´ e pox´ ide hye´´ droe klor´ ide and kli din´ ee um broe´ mide)
for oral useWhat is the most important information I should know about chlordiazepoxide hydrochloride and clidinium bromide capsules?
-
Chlordiazepoxide hydrochloride and clidinium bromide capsules contain a benzodiazepine medicine. Taking chlordiazepoxide hydrochloride and clidinium bromide capsules with opioid medicines, alcohol, or other central nervous system (CNS) depressants (including street drugs) can cause severe drowsiness, breathing problems (respiratory depression), coma, and death. Get emergency help right away if any of the following happens:
- shallow or slowed breathing
- breathing stops (which may lead to the heart stopping)
- excessive sleepiness (sedation)
Do not drive or operate heavy machinery until you know how taking chlordiazepoxide hydrochloride and clidinium bromide capsules with opioids affects you.
-
Risk of abuse, misuse, and addiction. There is a risk of abuse, misuse, and addiction with benzodiazepines, including chlordiazepoxide hydrochloride and clidinium bromide capsules, which can lead to overdose or death.
- Serious side effects including coma and death have happened in people who have abused or misused benzodiazepines, including chlordiazepoxide hydrochloride and clidinium bromide capsules. These serious side effects may also include delirium, paranoia, suicidal thoughts or actions, seizures, and difficulty breathing. Call your healthcare provider or go to the nearest hospital emergency room right away if you get any of these serious side effects.
- You can develop an addiction even if you take chlordiazepoxide hydrochloride and clidinium bromide capsules as prescribed by your healthcare provider.
- Take chlordiazepoxide hydrochloride and clidinium bromide capsules exactly as your healthcare provider prescribed.
- Do not share your chlordiazepoxide hydrochloride and clidinium bromide capsules with other people.
- Keep chlordiazepoxide hydrochloride and clidinium bromide capsules in a safe place and away from children.
-
Physical dependence and withdrawal reactions. Chlordiazepoxide hydrochloride and clidinium bromide capsules can cause physical dependence and withdrawal reactions.
- Do not suddenly stop using chlordiazepoxide hydrochloride and clidinium bromide capsules. Stopping chlordiazepoxide hydrochloride and clidinium bromide capsules suddenly can cause serious and life-threatening side effects, including unusual movements, responses, or expressions, seizures, sudden and severe mental or nervous system changes, depression, seeing or hearing things that others do not see or hear, an extreme increase in activity or talking, losing touch with reality, and suicidal thoughts or actions. Call your healthcare provider or go to the nearest hospital emergency room right away if you get any of these symptoms.
- Some people who suddenly stop benzodiazepines have symptoms that can last for several weeks to more than 12 months, including anxiety, trouble remembering, learning, or concentrating, depression, problems sleeping, feeling like insects are crawling under your skin, weakness, shaking, muscle twitches, burning or prickling feeling in your hands, arms, legs or feet, and ringing in your ears.
- Physical dependence is not the same as drug addiction. Your healthcare provider can tell you more about the differences between physical dependence and drug addiction.
- Do not take more chlordiazepoxide hydrochloride and clidinium bromide capsules than prescribed or take chlordiazepoxide hydrochloride and clidinium bromide capsules for longer than prescribed.
What are chlordiazepoxide hydrochloride and clidinium bromide capsules?
- Chlordiazepoxide hydrochloride and clidinium bromide capsules are a prescription medicine that is used with other therapies for the treatment of:
- stomach (peptic) ulcers
- irritable bowel syndrome (IBS)
- inflammation of the colon called acute enterocolitis
- Chlordiazepoxide hydrochloride and clidinium bromide capsules contain the medicines chlordiazepoxide HCl and clidinium bromide.
- Chlordiazepoxide hydrochloride and clidinium bromide capsules contain chlordiazepoxide hydrochloride that can be abused or lead to dependence. Keep chlordiazepoxide hydrochloride and clidinium bromide capsules in a safe place to prevent misuse and abuse. Selling or giving away chlordiazepoxide hydrochloride and clidinium bromide capsules may harm others. Tell your healthcare provider if you have abused or been dependent on alcohol, prescription medicines or street drugs.
- It is not known if chlordiazepoxide hydrochloride and clidinium bromide capsules is safe and effective in children.
Do not take chlordiazepoxide hydrochloride and clidinium bromide capsules if you:
- have glaucoma
- have an enlarged prostate
- have a blockage of your bladder that causes problems with urination
- are allergic to chlordiazepoxide hydrochloride or clidinium bromide
Before you take chlordiazepoxide hydrochloride and clidinium bromide capsules, tell your healthcare provider about all of your medical conditions, including if you:
- have eye problems
- have problems urinating or emptying your bladder
- have coordination problems
- have kidney or liver problems
- have a history of depression, mental illness, or suicidal thoughts
- have a history of drug or alcohol abuse or addiction
- have bleeding problems
- are pregnant or plan to become pregnant. Chlordiazepoxide hydrochloride and clidinium bromide capsules may harm your unborn baby. Avoid taking chlordiazepoxide hydrochloride and clidinium bromide capsules during the first trimester of pregnancy. Tell your healthcare provider right away if you become pregnant during treatment with chlordiazepoxide hydrochloride and clidinium bromide capsules.
- are breastfeeding or plan to breastfeed. Chlordiazepoxide hydrochloride and clidinium bromide capsules may pass through your breast milk and may harm your baby. Talk to your healthcare provider about the best way to feed your baby if you take Chlordiazepoxide hydrochloride and clidinium bromide capsules. Chlordiazepoxide hydrochloride and clidinium bromide capsules may decrease the amount of breast milk your body makes.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. Taking Chlordiazepoxide hydrochloride and clidinium bromide capsules with certain other medicines can cause side effects or affect how well chlordiazepoxide hydrochloride and clidinium bromide capsules or the other medicines work. Do not start or stop other medicines without talking to your healthcare provider.
Especially tell your healthcare provider if you:
- take a monoamine oxidase inhibitor (MAOI) medicine or an anti-psychotic medicine called phenothiazine.
How should I take chlordiazepoxide hydrochloride and clidinium bromide capsules?
- Take chlordiazepoxide hydrochloride and clidinium bromide capsules exactly as your healthcare provider tells you to take it.
- Your healthcare provider may change your dose of chlordiazepoxide hydrochloride and clidinium bromide capsules if needed. Do not change your dose of chlordiazepoxide hydrochloride and clidinium bromide capsules or suddenly stop taking chlordiazepoxide hydrochloride and clidinium bromide capsules without talking with your healthcare provider.
- If you take too much chlordiazepoxide hydrochloride and clidinium bromide capsules, call your healthcare provider or go to the nearest hospital emergency room right away.
What are the possible side effects of chlordiazepoxide hydrochloride and clidinium bromide capsules?
Chlordiazepoxide hydrochloride and clidinium bromide capsules may cause serious side effects, including: See “What is the most important information I should know about Chlordiazepoxide hydrochloride and clidinium bromide capsules?”
-
Chlordiazepoxide hydrochloride and clidinium bromide capsules can make you sleepy or dizzy and can slow your thinking and motor skills.
- Do not drive, operate heavy machinery, or do other dangerous activities until you know how chlordiazepoxide hydrochloride and clidinium bromide capsules affect you.
- Do not drink alcohol or take other drugs that may make you sleepy or dizzy while taking chlordiazepoxide hydrochloride and clidinium bromide capsules without first talking to your healthcare provider. When taken with alcohol or drugs that cause sleepiness or dizziness, chlordiazepoxide hydrochloride and clidinium bromide capsules may make your sleepiness or dizziness much worse.
The most common side effects of Chlordiazepoxide Hydrochloride/Clidinium Bromide include:
- dry mouth
- blurred vision
- nausea
- constipation
- skin problems
- swelling
- irregular menstrual (periods) cycles
- increase and decreased desire for sex (libido)
- problems starting to urinate
- drowsiness, coordination problems, and confusion may happen, especially in people who are elderly or weak
These are not all the possible side effects of chlordiazepoxide hydrochloride and clidinium bromide capsules.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store chlordiazepoxide hydrochloride and clidinium bromide capsules?
- Store chlordiazepoxide hydrochloride and clidinium bromide capsules at room temperature 77°F (25°C).
- Keep chlordiazepoxide hydrochloride and clidinium bromide capsules and all medicines out of the reach of children.
General information about the safe and effective use of chlordiazepoxide hydrochloride and clidinium bromide capsules.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not take chlordiazepoxide hydrochloride and clidinium bromide capsules for a condition for which it was not prescribed. Do not give chlordiazepoxide hydrochloride and clidinium bromide capsules to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about chlordiazepoxide hydrochloride and clidinium bromide capsules that is written for health professionals.
What are the ingredients in chlordiazepoxide hydrochloride and clidinium bromide capsules?
Active ingredients: chlordiazepoxide hydrochloride and clidinium bromide
Inactive ingredients: lactose monohydrate, pregelatinized starch, talc. Gelatin capsule shells may contain titanium dioxide with the following dye systems: D&C Yellow #10, FD&C Green #3 and gelatin.
Manufactured for:
Xiromed, LLC
Florham Park, NJ 07932Made in the USA
-
Chlordiazepoxide hydrochloride and clidinium bromide capsules contain a benzodiazepine medicine. Taking chlordiazepoxide hydrochloride and clidinium bromide capsules with opioid medicines, alcohol, or other central nervous system (CNS) depressants (including street drugs) can cause severe drowsiness, breathing problems (respiratory depression), coma, and death. Get emergency help right away if any of the following happens:
- Package Label
-
INGREDIENTS AND APPEARANCE
CHLORDIAZEPOXIDE HYDROCHLORIDE AND CLIDINIUM BROMIDE
chlordiazepoxide hydrochloride and clidinium bromide capsuleProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC: 70700-185 Route of Administration ORAL DEA Schedule CIV Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHLORDIAZEPOXIDE (UNII: 6RZ6XEZ3CR) (CHLORDIAZEPOXIDE - UNII:6RZ6XEZ3CR) CHLORDIAZEPOXIDE 5 mg CLIDINIUM BROMIDE (UNII: 91ZQW5JF1Z) (CLIDINIUM - UNII:BO76JF850N) CLIDINIUM BROMIDE 2.5 Inactive Ingredients Ingredient Name Strength STARCH, PREGELATINIZED CORN (UNII: O8232NY3SJ) TALC (UNII: 7SEV7J4R1U) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) GELATIN (UNII: 2G86QN327L) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C GREEN NO. 3 (UNII: 3P3ONR6O1S) Product Characteristics Color green (light green opaque capsules) Score 4 pieces Shape CAPSULE Size 14mm Flavor Imprint Code VP302 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 70700-185-01 100 in 1 BOTTLE; Type 0: Not a Combination Product 04/05/2021 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA210579 04/05/2021 Labeler - Xiromed, LLC (080228637) Registrant - Misemer Pharmaceuticals, Inc. (784121365) Establishment Name Address ID/FEI Business Operations Belcher Pharmaceuticals, LLC 965082543 manufacture(70700-185) , analysis(70700-185) , pack(70700-185) , label(70700-185)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
