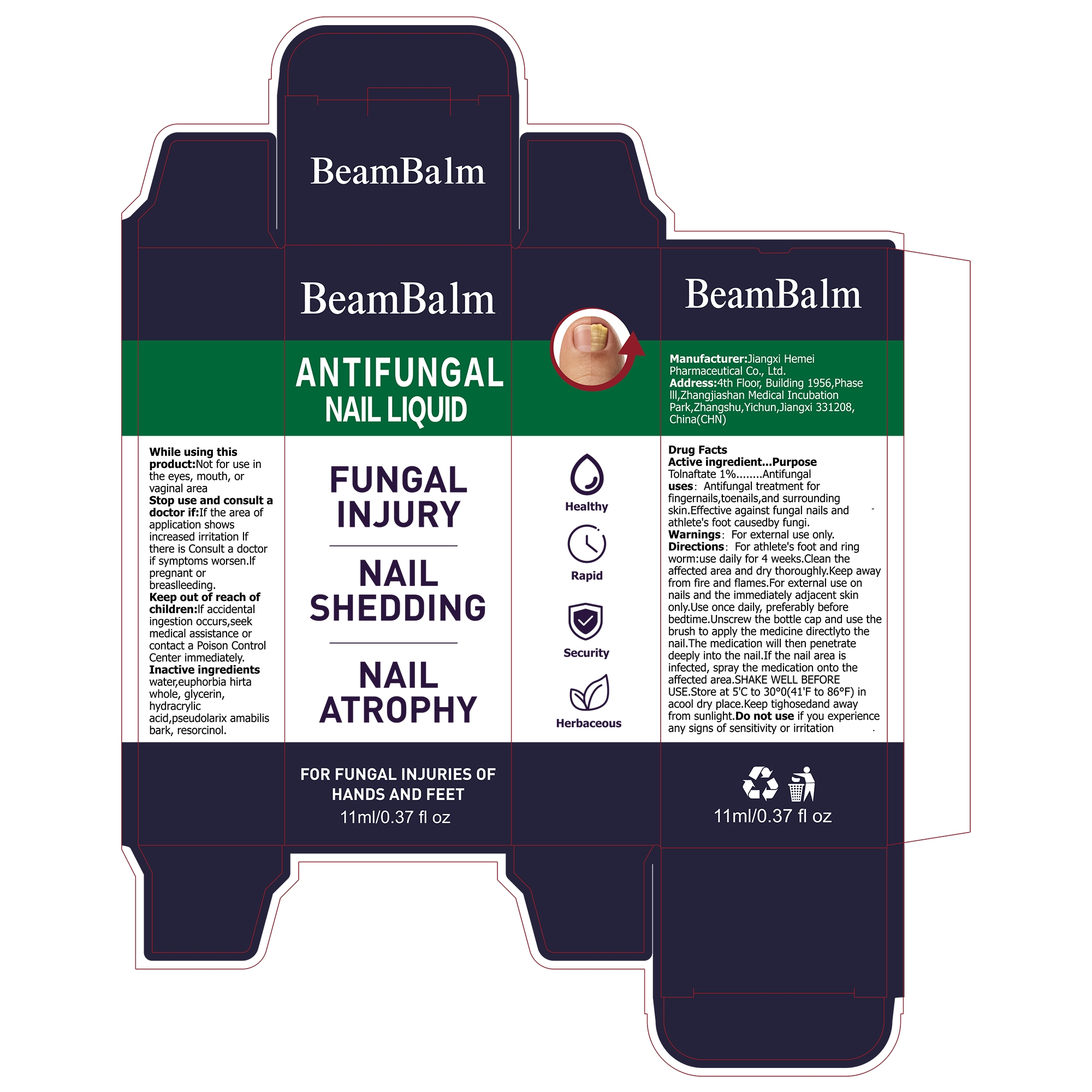
BeamBalm ANTIFUNGAL NAIL by Jiangxi Hemei Pharmaceutical Co., Ltd 84010-205 complete
BeamBalm ANTIFUNGAL NAIL by
Drug Labeling and Warnings
BeamBalm ANTIFUNGAL NAIL by is a Otc medication manufactured, distributed, or labeled by Jiangxi Hemei Pharmaceutical Co., Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
BEAMBALM ANTIFUNGAL NAIL- tolnaftate 1% antifungal nail liquid
Jiangxi Hemei Pharmaceutical Co., Ltd
----------
84010-205 complete
Use
Antifungal treatment for fingernails,toenails, and surrounding skin.Effective against fungal nails and athlete's foot caused by fungi.
Stop Use
lf the area of application shows increased irritation lf there is Consult a doctor if symptoms worsen.If pregnant or breaslleeding
Ask Doctor
lf the area of application shows increased irritation lf there is Consult a doctor if symptoms worsen.If pregnant or breaslleeding
Keep Oot Of Reach Of Children
lf accidental ingestion occurs,seek medical assistance or contact a Poison Control Centerimmediately.
Directions
For athlete's foot and ringworm:use daily for 4 weeks.Clean the affected area and dry thoroughly.Keep away from fire and flames.For external use on nails and the immediately adjacent skin only.Use once daily, preferably before bedtime.
Unscrew the bottle cap and use the brush to apply the medicine directlyto the nail.The medication will then penetrate deeply into the nail.If the nail area is infected, spray the medication onto the affected area.SHAKE WELL BEFORE USE.Store at 5'c to 30°0(41'F to 86°F) in a cool dry place. Keep tighosedand away from sunlight.
| BEAMBALM ANTIFUNGAL NAIL
tolnaftate 1% antifungal nail liquid |
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
|
|||||||||||||||
| Labeler - Jiangxi Hemei Pharmaceutical Co., Ltd (724892056) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Jiangxi Hemei Pharmaceutical Co., Ltd | 724892056 | manufacture(84010-205) | |