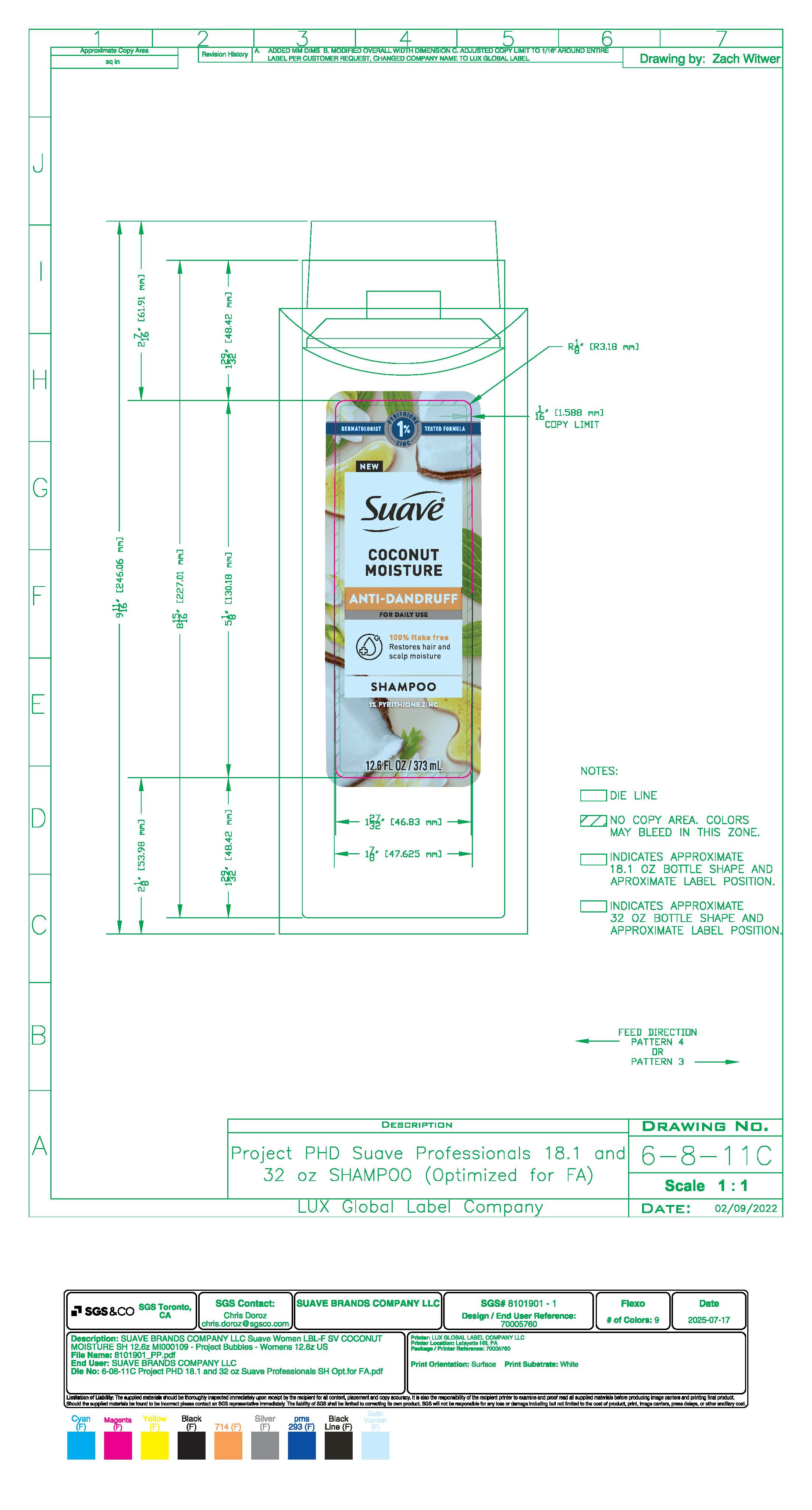
SUAVE ANTI DANDRUFF COCONUT MOISTURE- anti dandruff shampoo shampoo
Suave Anti dandruff Coconut Moisture by
Drug Labeling and Warnings
Suave Anti dandruff Coconut Moisture by is a Otc medication manufactured, distributed, or labeled by Suave Brands Co., LLC, Consumer Product Partners (CPP) (MO). Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- DOSAGE & ADMINISTRATION
-
WARNINGS
Warnings
For external use only
When using this product avoid contact with eyes. If contact occurs, rinse eyes thoroughly with water.
Stop and ask a doctor if condition worsens or does not improve after regular use of this product as desired.
Keep this and all drugs out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away -
KEEP OUT OF REACH OF CHILDREN
Warnings
For external use only
When using this product avoid contact with eyes. If contact occurs, rinse eyes thoroughly with water.
Stop and ask a doctor if condition worsens or does not improve after regular use of this product as desired.
Keep this and all drugs out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away -
INACTIVE INGREDIENT
WATER (AQUA), SODIUM LAURETH SULFATE, COCAMIDOPROPYL BETAINE, SODIUM CHLORIDE, PHENOXYETHANOL, CITRIC ACID, CARBOMER, DIMETHICONOL, CAPRYLYL GLYCOL, FRAGRANCE
(PARFUM), SODIUM BENZOATE, PPG-9, SODIUM HYDROXIDE, ZINC SULFATE, GUAR HYDROXYPROPYLTRIMONIUM CHLORIDE, TEA-DODECYLBENZENESULFONATE, COCOS NUCIFERA (COCONUT) FRUIT EXTRACT, HONEY EXTRACT, LIMONENE, CITRUS LIMON (LEMON) PEEL OIL, CINNAMYL ALCOHOL - ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SUAVE ANTI DANDRUFF COCONUT MOISTURE
anti dandruff shampoo shampooProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 83711-308 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PYRITHIONE ZINC (UNII: R953O2RHZ5) (PYRITHIONE ZINC - UNII:R953O2RHZ5) PYRITHIONE ZINC 1 g in 100 mL Inactive Ingredients Ingredient Name Strength CITRIC ACID (UNII: 2968PHW8QP) CARBOMER (UNII: 0A5MM307FC) ZINC SULFATE (UNII: 89DS0H96TB) TEA-DODECYLBENZENESULFONATE (UNII: 8HM7ZD48HN) SODIUM BENZOATE (UNII: OJ245FE5EU) GUAR HYDROXYPROPYLTRIMONIUM CHLORIDE (UNII: B16G315W7A) SODIUM CHLORIDE (UNII: 451W47IQ8X) DIMETHICONOL (100000 CST) (UNII: OSA9UP217S) PPG-9 (UNII: I29VQH0G0B) SODIUM HYDROXIDE (UNII: 55X04QC32I) LIMONENE, (+/-)- (UNII: 9MC3I34447) PHENOXYETHANOL (UNII: HIE492ZZ3T) HONEY (UNII: Y9H1V576FH) COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) CAPRYLYL GLYCOL (UNII: 00YIU5438U) SODIUM LAURETH SULFATE (UNII: BPV390UAP0) COCONUT (UNII: 3RT3536DHY) CINNAMYL ALCOHOL (UNII: SS8YOP444F) CITRUS LIMON (LEMON) PEEL OIL (UNII: I9GRO824LL) WATER (UNII: 059QF0KO0R) FRAGRANCE CLEAN ORC0600327 (UNII: 329LCV5BTF) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 83711-308-75 373 mL in 1 BOTTLE; Type 0: Not a Combination Product 10/23/2025 2 NDC: 83711-308-28 828 mL in 1 BOTTLE; Type 0: Not a Combination Product 10/23/2025 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M032 10/23/2025 Labeler - Suave Brands Co., LLC (118965468) Establishment Name Address ID/FEI Business Operations Consumer Product Partners (CPP) (MO) 119091520 manufacture(83711-308)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.