CEFPODOXIME PROXETIL tablet
Cefpodoxime Proxetil by
Drug Labeling and Warnings
Cefpodoxime Proxetil by is a Animal medication manufactured, distributed, or labeled by BUTLER ANIMAL HEALTH SUPPLY, LLC DBA COVETRUS NORTH AMERICA, FELIX PHARMACEUTICALS PRIVATE LIMITED. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- CAUTION
-
DESCRIPTION
Cefpodoxime proxetil is an orally administered, extended spectrum, semi-synthetic cephalosporin antibiotic. The chemical name is: (+/-)-1-Hydroxyethyl(+)-(6R,7R)-7-[2-(2-amino-4-thiazolyl) glyoxylamido]-3-methoxymethyl)-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate, 72-(Z)- (O-methyloxime), isopropyl carbonate (ester) [87239-81-4].
Cefpodoxime proxetil Chemical Structure:
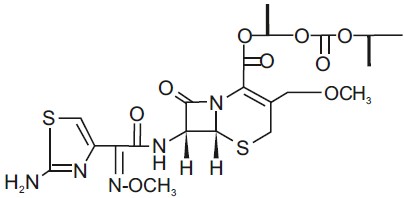
Cefpodoxime proxetil is a prodrug; its active metabolite is cefpodoxime. All doses of Cefpodoxime Proxetil Tablets are expressed in terms of the active cefpodoxime moiety. Cefpodoxime Proxetil Tablets are available as:
100 mg Tablet, each red-colored, oval-shaped, film-coated, scored tablet contains cefpodoxime proxetil equivalent to 100 mg of cefpodoxime.
200 mg Tablet, each orange-colored, oblong-shaped, film-coated, scored tablet contains cefpodoxime proxetil equivalent to 200 mg of cefpodoxime.
-
INDICATION
Cefpodoxime Proxetil Tablets are indicated for the treatment of skin infections (wounds and abscesses) in dogs caused by susceptible strains of Staphylococcus pseudintermedius, Staphylococcus aureus, Streptococcus canis (group G, ß hemolytic), Escherichia coli, Pasteurella multocida, and Proteus mirabilis.
-
DOSAGE AND ADMINISTRATION
Dose range
The dose range of Cefpodoxime Proxetil Tablets is 5-10 mg/kg (2.3-4.5 mg/lb) body weight, administered orally, once a day. The dose may be given with or without food. The determination of dosage for any particular patient must take into consideration such factors as the severity and nature of the infection, the susceptibility of the causative organisms, and the integrity of the patient's host-defense mechanisms. Obtain a sample of the pathogenic organism for culture and sensitivity testing prior to beginning antimicrobial therapy. Once results become available, continue with appropriate therapy.
Duration
Cefpodoxime Proxetil Tablets should be administered once daily for 5-7 days or for 2-3 days beyond the cessation of clinical signs, up to a maximum of 28 days. Treatment of acute infections should not be continued for more than 3-4 days if no response to therapy is seen.
Dosing Charts
For daily oral administration of Cefpodoxime Proxetil Tablets at 5 mg/kg (Table 1) and 10 mg/kg (Table 2).
Table 1. Dose Table for Cefpodoxime Proxetil Tablets at 5 mg/kg Total Daily Dosage
Weight of Dog (lbs)
Daily Dose
22
44
66
88
132
No. of 100 mg tablets
0.5
1
1.5
1
No. of 200 mg tablets
1
1
Weight of Dog (kgs)
Daily Dose
10
20
30
40
60
No. of 100 mg tablets
0.5
1
1.5
1
No. of 200 mg tablets
1
1
Table 2. Dose Table for Cefpodoxime Proxetil Tablets at 10 mg/kg Total Daily Dosage
Weight of Dog (lbs)
Daily Dose
11
22
44
66
88
132
No. of 100 mg tablets
0.5
1
1
No. of 200 mg tablets
1
1
2
3
Weight of Dog (kgs)
Daily Dose
5
10
20
30
40
60
No. of 100 mg tablets
0.5
1
1
No. of 200 mg tablets
1
1
2
3 - CONTRAINDICATIONS
-
WARNINGS
Not for human use. Keep this and all drugs out of reach of children. Antimicrobial drugs, including penicillins and cephalosporins, can cause allergic reactions in sensitized individuals. To minimize the possibility of allergic reactions, those handling such antimicrobials, including cefpodoxime, are advised to avoid direct contact of the product with the skin and mucous membranes.
- PRECAUTIONS
-
ADVERSE REACTIONS
A total of 216 dogs of various breeds and ages ranging from 2 months to 15 years were included in the field study safety analysis. The following table shows the number of dogs displaying each clinical observation.
Table 3. Abnormal Health Findings in the U.S. Field Study*
Clinical Observation
cefpodoxime proxetil tablets (n=118)
Active Control (n=98)
Vomiting
2
4
Diarrhea
1
1
Increased water drinking
0
2
Decreased appetite
1
1*Dogs may have experienced more than one of the observations during the study.
-
CONTACT INFORMATION
To report suspected adverse drug events, for technical assistance or to obtain a copy of the Safety Data Sheet, contact Covetrus® North America at (855) 724-3461 or covetrus.com.
For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888- FDA-VETS or http://www.fda.gov/reportanimalae
-
CLINICAL PHARMACOLOGY
Pharmacokinetics/Pharmacodynamics
Cefpodoxime proxetil is a prodrug that is absorbed from and de-esterified in the gastrointestinal tract to its active metabolite, cefpodoxime. Following oral administration to fasting Beagles, oral bioavailability was 63.1 ± 5.3%.
Figure 1. Canine Plasma Concentration of Cefpodoxime After a Single Oral Dose of 10 mg/kg cefpodoxime proxetil tablets
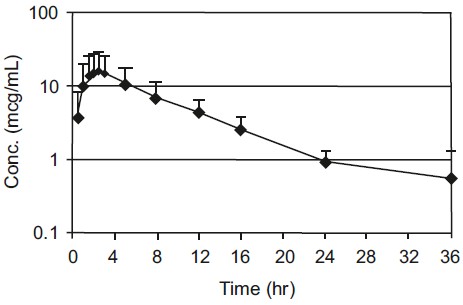
Cefpodoxime is distributed in the body with an apparent volume of distribution of 151 ± 27 mL/kg. Like other β-lactam antibiotics, cefpodoxime is eliminated from the body primarily in the urine, with an apparent elimination half-life of approximately 5-6 hours after oral administration. This is similar to the 4.7 hour apparent elimination half-life observed after intravenous dosing. Following intravenous administration of 10 mg/kg, the average total body clearance (CIB) was 22.7 ± 4.19 mL/hr/kg.
Table 4. Summary of Pharmacokinetic Parameters Obtained after a Single Oral Dose of 10 mg Cefpodoxime/kg BW, Administered as a Tablet
PK Parameter
Unit
Tablet (SD)
AUC0-∞
mcghr/mL
145 (77.6)
AUC0-LOQ
mcghr/mL
142 (77.5)
Maximum concentration (Cmax)
mcg/mL
16.4 (11.8)
Terminal plasma elimination half-life (t1/2,Z)
hr
5.61 (1.15)
Time of maximum concentration (tmax)
hr
2.21 (0.542)
Mean residence time (MRT0-∞)
hr
9.21 (1.97)
Microbiology
Like other β-lactam antibiotics, cefpodoxime exerts its inhibitory effect by interfering with bacterial cell wall synthesis. This interference is primarily due to its covalently binding to the penicillin-binding proteins (PBPs) (i.e. transpeptidase and/or carboxypeptidase), which are essential for synthesis of the bacterial cell wall. Therefore, cefpodoxime is bactericidal. Cefpodoxime is stable in the presence of many common β-lactamase enzymes. As a result, many organisms resistant to other β-lactam antibiotics (penicillins and some cephalosporins) due to the production of β-lactamases may be susceptible to cefpodoxime.
Cefpodoxime has a broad spectrum of clinically useful antibacterial activity that includes staphylococci, streptococci, and Gram-negative species (including Pasteurella, Escherichia, and Proteus). The compound is not active against most obligate anaerobes, Pseudomonas spp., or enterococci. The minimum inhibitory concentrations (MICs) for cefpodoxime against Gram-positive and Gram-negative pathogens isolated from canine skin infections (wounds and abscesses) in a 2002 U.S. field study are presented in Table 5. All MICs were determined in accordance with the National Committee for Clinical Laboratory Standards (NCCLS). Appropriate quality control (QC) ranges for in vitro susceptibility testing are presented in Table 6.
Table 5. Cefpodoxime Minimum Inhibitory Concentration Values (mcg/mL) from a 2002 Field Study Evaluating Skin Infections (wounds and abscesses) of Canines in the United States.
Organism*
# of Isolates
MIC50
MIC90
Range
Staphylococcus pseudintermedius
118
0.12
0.50
0.12->32.0
Streptococcus canis (group G, β hemolytic)
33
≤0.03
≤0.03
≤0.03†
Escherichia coli
41
0.25
0.50
0.12->32.0
Pasteurella multocida
32
≤0.03
≤0.03
≤0.03-0.12
Proteus mirabilis
14
≤0.03
0.06
≤0.03-0.06
Staphylococcus aureus
19
2.0
2.0
0.12-2.0* Veterinary specific interpretive criteria have not been established for the above listed canine pathogens by the NCCLS at this time.
† No Range, all isolates yielded the same value.
Table 6. Acceptable Quality Control Ranges for Cefpodoxime
QC ATCC strain
KB Disk Diffusion Method
Broth Micro-dilution Method
Drug concentration
Zone diameter
MIC
Escherichia coli 25922
10 mcg
23-28 mm*
0.25-1 mcg/mL*
Staphylococcus aureus 25923
10 mcg
19-25 mm*
Staphylococcus aureus 29213
1-8 mcg/mL*
Streptococcus pneumoniae 49619
10 mcg
28-34 mm†
0.03-0.12 mcg/mL†*These ranges are for quality control strains used to monitor accuracy of minimum inhibitory concentrations (MICs) of non-fastidious organisms using cation-adjusted Mueller-Hinton agar or broth medium. The dilution range should encompass the QC ranges of these strains in the broth micro-dilution method.
†These ranges are for quality control strains used to monitor accuracy of minimum inhibitory concentrations (MICs) of fastidious organisms. When susceptibility testing is performed for Streptococcus canis (group G, β hemolytic), Streptococcus pneumoniae ATCC 49619 should be included as a QC strain in the presence of 5% lysed sheep blood (KB disk diffusion method) or 2.5% lysed horse blood (broth micro-dilution method).
-
EFFECTIVENESS
The clinical effectiveness of cefpodoxime proxetil tablets was established in a multi-location (23 site) field study. In this study, 216 dogs with infected wounds or abscesses were treated with either cefpodoxime proxetil tablets (n=118) once daily at 5 mg/kg (2.3 mg/lb) body weight or with an active control antibiotic (n=98) administered twice daily for 5-7 days. In this study, cefpodoxime proxetil tablets were considered noninferior to the active control (88.7% versus 88.4% respectively) in the treatment of canine skin infections (wounds and abscesses) caused by susceptible strains of Staphylococcus pseudintermedius, Staphylococcus aureus, Streptococcus canis (group G, β hemolytic), Escherichia coli, Pasteurella multocida, and Proteus mirabilis.
-
ANIMAL SAFETY
In target animal safety studies, cefpodoxime was well tolerated at exaggerated daily oral doses of 100 mg/kg/day (10 times the maximum label dose) for 13 weeks in adult dogs and for 28 days in puppies (18-23 days of age). Therefore, once daily administration of cefpodoxime oral tablets at the maximum labeled dose of 10 mg/kg for up to 28 days was shown to be safe in adult dogs and puppies.
Blood dyscrasia including neutropenias, may be seen following high doses of cephalosporins. Cephalosporin administration should be discontinued in such cases.
- STORAGE INFORMATION
-
HOW SUPPLIED
Cefpodoxime Proxetil Tablets are available in the following strengths (cefpodoxime equivalent), colors, and sizes:
100 mg (Red-colored, oval-shaped, film-coated tablet, debossed with "F" & "3" on either side of score line on one side of tablets and "1" & "00" on either side of score line on other side of the tablet.)
Bottles of 100
NDC: 11695-7040-2
Bottles of 250
NDC: 11695-7040-1
200 mg (Orange-colored, oblong-shaped, film-coated tablet, debossed with "F" & "3" on either side of score line on one side of tablets and "2" & "00" on either side of score line on other side of the tablet.)
Bottles of 100
NDC: 11695-7041-2
Bottles of 250
NDC: 11695-7041-1 - SPL UNCLASSIFIED SECTION
-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
NDC: 11695-7040-2
Cefpodoxime Proxetil Tablets
100 mg
Antimicrobial For Oral Use in Dogs Only
Caution: Federal law restricts this drug to use by or on the order of a licensed veterinarian.
Approved by FDA under ANADA # 200-815
100 Tablets

-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
NDC: 11695-7040-1
Cefpodoxime Proxetil Tablets
100 mg
Antimicrobial For Oral Use in Dogs Only
Caution: Federal law restricts this drug to use by or on the order of a licensed veterinarian.
Approved by FDA under ANADA # 200-815
250 Tablets

-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
NDC: 11695-7041-2
Cefpodoxime Proxetil Tablets
200 mg
Antimicrobial For Oral Use in Dogs Only
Caution: Federal law restricts this drug to use by or on the order of a licensed veterinarian.
Approved by FDA under ANADA # 200-815
100 Tablets

-
PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
NDC: 11695-7041-1
Cefpodoxime Proxetil Tablets
200 mg
Antimicrobial For Oral Use in Dogs Only
Caution: Federal law restricts this drug to use by or on the order of a licensed veterinarian.
Approved by FDA under ANADA # 200-815
250 Tablets

-
INGREDIENTS AND APPEARANCE
CEFPODOXIME PROXETIL
cefpodoxime proxetil tabletProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC: 11695-7040 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CEFPODOXIME PROXETIL (UNII: 2TB00A1Z7N) (CEFPODOXIME - UNII:7R4F94TVGY) CEFPODOXIME 100 mg Product Characteristics Color RED Score 2 pieces Shape OVAL Size 11mm Flavor Imprint Code F;3;1;00 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 11695-7040-2 100 in 1 BOTTLE 2 NDC: 11695-7040-1 250 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANADA ANADA200815 07/22/2025 CEFPODOXIME PROXETIL
cefpodoxime proxetil tabletProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC: 11695-7041 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CEFPODOXIME PROXETIL (UNII: 2TB00A1Z7N) (CEFPODOXIME - UNII:7R4F94TVGY) CEFPODOXIME 200 mg Product Characteristics Color ORANGE Score 2 pieces Shape OVAL (Oblong) Size 16mm Flavor Imprint Code F;3;2;00 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 11695-7041-2 100 in 1 BOTTLE 2 NDC: 11695-7041-1 250 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANADA ANADA200815 07/22/2025 Labeler - BUTLER ANIMAL HEALTH SUPPLY, LLC DBA COVETRUS NORTH AMERICA (603750329) Registrant - FELIX PHARMACEUTICALS PRIVATE LIMITED (985612369)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.