Acetaminophen Tablets extra strength
Acetaminophen by
Drug Labeling and Warnings
Acetaminophen by is a Otc medication manufactured, distributed, or labeled by Elysium Pharmaceuticals Ltd., Elysium Pharmaceuticals Limited. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
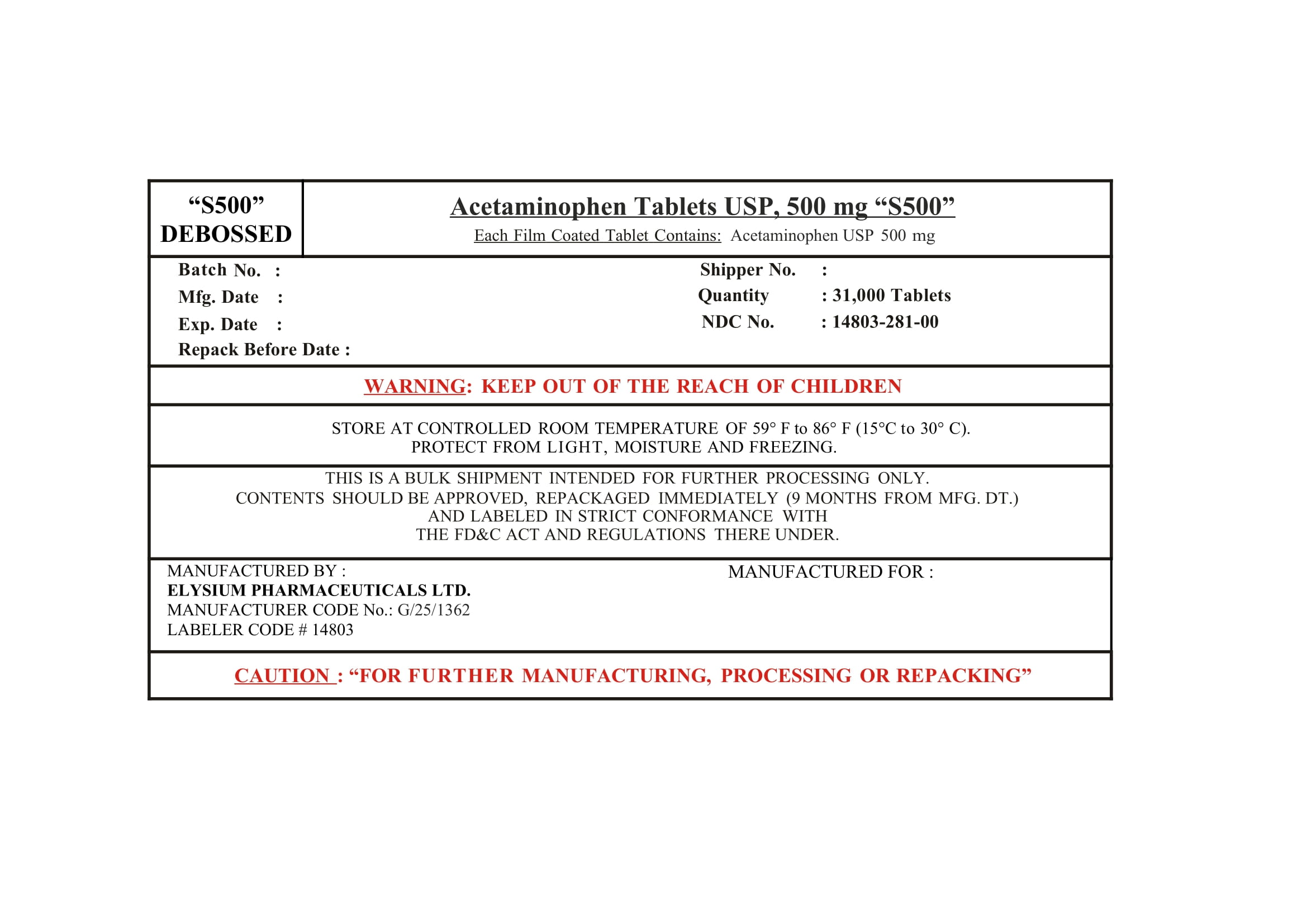
ACETAMINOPHEN- acetaminophen tablet
Elysium Pharmaceuticals Ltd.
----------
Acetaminophen Tablets extra strength
| ACETAMINOPHEN
acetaminophen tablet |
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
|
||||||||||||||||||||||
| Labeler - Elysium Pharmaceuticals Ltd. (915664486) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Elysium Pharmaceuticals Ltd. | 915664486 | manufacture(14803-281) , analysis(14803-281) | |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Elysium Pharmaceuticals Limited | 863182240 | manufacture(14803-281) , pack(14803-281) | |
Revised: 11/2025
Document Id: 44a372b0-69e7-21b5-e063-6294a90ab111
Set id: 44a36fdb-23bc-2b67-e063-6294a90aa77b
Version: 2
Effective Time: 20251127
Trademark Results [Acetaminophen]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 ACETAMINOPHEN 85615223 not registered Dead/Abandoned |
General Merchandise importers and Expoters 2012-05-03 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.