RHEUMATE- l-5-methyltetrahydrofolate, methylcobalamin, curcuminoid turmerone complex capsule
Rheumate by
Drug Labeling and Warnings
Rheumate by is a Other medication manufactured, distributed, or labeled by Primus Pharmaceuticals, Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
-
SPL UNCLASSIFIED SECTION
Dispensed by prescription.
L-5-methyltetrahydrofolate 1 mg
Methylcobalamin (vitamin B-12) 1 mg
Curcuminoid turmerone complex 500 mg
1mg L-5-methyltetrahydrofolate equals 1.7mg dietary folate equivalent.
RHEUMATE is a specially formulated prescription medical food product for the clinical dietary management of metabolic effects of methotrexate therapy administered to patients with chronic inflammatory diseases. RHEUMATE must be administered under physician supervision.
-
DESCRIPTION
RHEUMATE consists of a proprietary mixture of L-5-methyltetrahydrofolate((6S)-5-methyltetrahydrofolic acid, methylcobalamin (vitamin B-12) and curcuminoid turmerone complex (high purity curcuminoids comprised of curcumin, desmethoxycurcumin, bis-desmethoxycurcumin and essential oils derived from turmeric rhizome).Reduced folate is required for one-carbon metabolic cycles. L-5-methyltetrahydrofolate addresses the methotrexate-induced distinctive nutritional requirement for folate due to competitive inhibition of reduced folate production, which may be more pronounced in people with a genetic polymorphism in the folate metabolic pathway. Methylcobalamin provides adequate vitamin B-12 stores to avoid masking borderline deficiency of B-12 with exogenous folate use. Curcumin helps to address the increased nutritional requirement for antioxidants associated with methotrexate induced oxidative stress.
L-5-methyltetrahydrofolate
L-5-methyltetrahydrofolate ((6S)-5-methyltetrahydrofolic acid) is chemically described as N-[4-[[[(6S)-2-amino-1,4,5,6,7,8-hexa-5-methyl-4-oxo-6-pteridinyl]methyl]amino]benzoyl]-L-glutamic acid with a molecular weight of 817.8. The empirical formula for L-5-methyltetrahydrofolic acid is C20H23N7O6(C6H14NO5)2. The structural formula is:
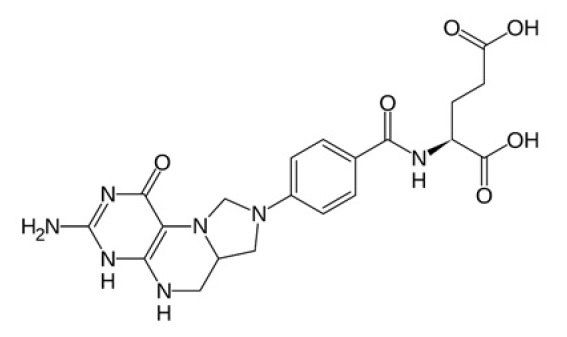
Methylcobalamin
Methylcobalamin is chemically described as carbanide; cobalt(2+);[(2R,3S,4R,5S)-5-(5,6-dimethylbenzimidazol-1-yl)-4-hydroxy-2-(hydroxymethyl)oxolan-3-yl][(2R)-1-[3-[(1R,2R,3R,4Z,7S,9Z,12S,13S,14Z,17S,18S,19R)-2,13,18-tris(2-amino-2-oxoethyl)-7,12,17-tris(3-amino-3-oxopropyl)-3,5,8,8,13,15,18,19-octamethyl-2,7,12,17-tetrahydro- 1H-corrin-21-id-3-yl]propanoylamino]propan-2-yl] phosphate with a molecular weight of 1344.4. The empirical formula for methylcobalamin is C63H91CoN13O14P. The structural formula is:
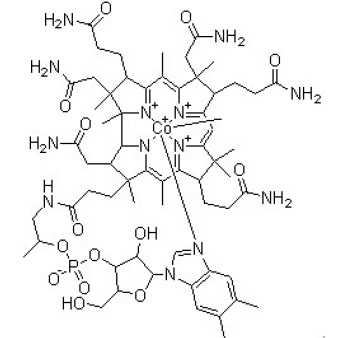
Curcuminoid turmerone complex
Curcuminoid turmerone complex is a composition of three curcuminoids and an essential oil, all sourced from turmeric.
Curcumin is chemically described as (1E,6E)-1,7-bis(4-hydroxy-3-methoxyphenyl)hepta-1,6-diene-3,5-dione with a molecular weight of 368.4. The empirical formula for curcumin is C21H20O6. The structural formula is:
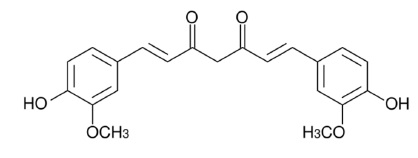
Desmethoxycurcumin is chemically described as (1E,4Z,6E)-5-hydroxy-7-(4-hydroxy-3-methoxyphenyl)-1-(4-hydroxyphenyl)hepta-1,4,6-trien-3-one with a molecular weight of 338.4. The empirical formula for desmethoxycurcumin is C20H18O5. The structural formula is:
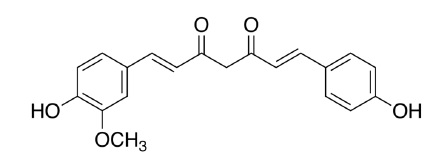
Bis-desmethoxycurcumin is chemically described as (1E,6E)-1,7-bis(4- hydroxyphenyl)hepta-1,6-diene-3,5-dione with a molecular weight of 308.3. The empirical formula for Bis-desmethoxycurcumin is C19H16O4. The structural formula is:
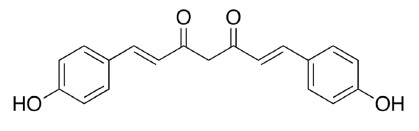
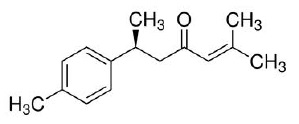
ar-turmerone, an essential oil of turmeric, is chemically described as (6S)-2-methyl-6-(4-methylphenyl)hept-2-en-4-one with a molecular weight of 216.3. The empirical formula for ar-turmerone is C15H20O. The structural formula is:
Additional Contents
RHEUMATE also contains the following: microcrystalline cellulose, bovine gelatin, magnesium stearate, titanium dioxide, silicone dioxide, FD&C yellow 5, and FD&C green 3.
Capsule contains bovine gelatin. RHEUMATE does not contain fructose, glucose, sucrose, lactose, gluten, maltodextrin, tree nuts, peanuts, soy, or flavors or product of seafood origin.
Medical Foods
The U.S. Congress defined “medical food” in the Orphan Drug Act and Amendments of 1988 as “a food which is formulated to be consumed or administered enterally under the supervision of a physician, and which is intended for the specific dietary management of a disease or condition for which distinctive nutritional requirements, based on recognized scientific principles, are established by medical evaluation.” RHEUMATE has been developed, manufactured and labeled in accordance with the statutory definition of a medical food. RHEUMATE is to be used only under a physician’s supervision.
Safety of Rheumate Ingredients
The ingredients in RHEUMATE meet the U.S. Food and Drug Administration (FDA) safety standards for food products. Ingredients may be used in food if they are Generally Recognized as Safe (GRAS), approved food additives, or approved color additives for the intended use. A use of a food substance may be GRAS either through scientific procedures or, for a substance used in food before 1958, through experience based on common use in food. General recognition of safety through scientific procedures ordinarily is based on published studies (which may be corroborated by
unpublished studies and other data and information). General recognition of safety through experience based on common use in foods requires a substantial history of consumption for food use by a significant number of consumers before 1958. All ingredients in RHEUMATE are either GRAS, approved food additives, or approved color additives.METHOTREXATE-INDUCED DISTINCT NUTRITIONAL REQUIREMENTS
Methotrexate is the most widely used disease modifying drug in the treatment of inflammatory diseases such as rheumatoid arthritis, psoriatic arthritis or psoriasis. However, as many as 30% of patients must discontinue or reduce dosage of methotrexate because of adverse effects of methotrexate due to the failure to meet the increased need for folate. Methotrexate associated adverse events leading to the increased need for folate involve interference with the 1-carbon methylation cycle and other enzymatic processes for which reduced folate is a cofactor. Competitive inhibition of dihydrofolate reductase (DHFR) by methotrexate results in depletion of endogenous reduced folate stores due to an increased need for folate. Methylcobalamin provides adequate vitamin B-12 stores to avoid masking borderline deficiency of B-12 with exogenous folate supplementation.
Methotrexate adverse events are thought to be due in large part to a disruption of cellular redox homeostasis (severe oxidative stress) with depletion of natural antioxidant mechanisms (SOD, GTH, catalase and other peroxidases) and marked rise in oxidative products such as thiobarbituric acid reactive substances (TBARS) and malondialdehyde. This creates increased nutritional requirement for exogenous antioxidants. This leads to a distinct nutritional requirement for compounds such as dietary polyphenols, e.g., curcuminoids, which are powerful anti-oxidants and induce augmentation of endogenous antioxidant mechanisms, thus reducing oxidative burden.
-
CLINICAL PHARMACOLOGY
L-5-methyltetrahydrofolate
L-5-methyltetrahydrofolate is the bioactive form of folate. It is well absorbed and able to participate without further metabolism in all biochemical processes for which folate is a cofactor.
Cobalamin
Cobalamin (vitamin B-12) is well absorbed under normal conditions. However, many older people as well as people with atrophic gastritis and gastric hypochlorhydria (reduced stomach acid production) may not absorb cobalamin properly. This defect can be overcome with ingestion of additional dietary B-12 except in people with autoimmune atrophic gastritis who must be supplemented with parenteral B-12.
Curcuminoids
Because curcuminoids are known to be poorly absorbed, various methods have been devised to increase absorption of the molecules. The proprietary mixture of purified curcuminoids and essential turmeric oil used in product RHEUMATE increases the absorption and serum retention time of the curcuminoids molecules approximately 7- fold (Antony 2006). None of the ingredients in RHEUMATE are known to affect bioavailability by interacting with each other.
Mechanism of Action
L-5-methyltetrahydrofolate
L-5-methyltetrahydrofolate addresses the distinctive nutritional requirement for folate of patients taking methotrexate for treatment of inflammatory diseases including, but not limited to, rheumatoid arthritis, psoriatic arthritis or psoriasis. Folate is an essential cofactor in the NADP/NADH dependent 1-carbon transformation system involving methionine, homocysteine and serine and side pathways of their metabolism. These reactions are important in the metabolism and biosynthesis of amino acids, nucleic acids, membrane lipids, cholesterol and neurotransmitters. A significant proportion of the general population, however, has one or another polymorphisms in the methyltetrahydrofolate reductase (MTHFR) gene which catalyzes the rate limiting final conversion of 5,10-MTHF into the active 5-MTHF and, therefore, require 5-MTHF rather than folic acid. At least 90 polymorphisms have been found in the MTHFR gene locus. The most common and important of these, found in up to 20% of people in the U.S. and up to 50% of Europeans, is the C677T polymorphism which in its various forms greatly reduces enzyme activity or renders the enzyme inactive. L-5-methyltetrahydrofolate is the bioactive form of folate. Substitution of folic acid with L-5-methyltetrahydrofolate salts that do not require 10-demethylation therefore may obviate the effect of these polymorphisms (Figure 1).

Methylcobalamin
Methylcobalamin is an essential cofactor in many folate dependent metabolic pathways and minor deficiencies in cobalamin may be obscured in the presence of supraphysiological folate levels. While this may suppress such clinical signs as megaloblastic anemia, it does not prevent neurological damage associated with B12 deficiency. Cobalamin supplementation provides sufficient vitamin B-12 to assure that L-5-methyltetrahydrofolate administration does not mask cobalamin deficiency.
Curcuminoids
Antioxidant EffectsMethotrexate is a drug that results in an increased nutritional requirement for liver antioxidant capacity because it increases levels of oxidative species and depletes levels of endogenous antioxidants. A significant adverse event associated with methotrexate use is liver injury. Drug induced liver injury (DILI) and its most serious outcome, acute fulminating hepatic failure, involve, among other things, increased production of reactive oxygen species (ROS) and nitric oxide and severe depletion of endogenous antioxidants such as superoxide dismutase (SOD), peroxidases and glutathione (GSH). These oxidative mechanisms cause activation of NFkB and increased expression of TNFα and other downstream inflammatory molecules which enhance the inflammatory response and leads to activation of cellular apoptotic and fibrotic mechanisms. Curcumin is an excellent antioxidant that scavenges a variety of reactive oxygen species (ROS) and inhibits lipid peroxidation about 10-fold more than vitamin E. Curcumin reduces formation of ROS and nitric oxide, helps restore endogenous antioxidant levels toward normal, reduces markers of lipid peroxidation, and inhibits the activation of NFkB without affecting the levels of constitutively expressed NFkB.
Other Clinical Effects
Curcumin also possesses significant anti-inflammatory effects related to inhibition of multiple aspects of arachidonic acid metabolism including suppression of prostaglandin (PG) and leukotriene (LT) synthesis by cyclooxygenase (COX) and lipoxygenase (LOX) by blocking respective enzyme induction. Curcumin down-regulates COX-2 and 5-LOX, IL-1β and TNFα by suppression of NFκB transcription and pre-transcriptional translocation to the nucleus. Curcumin also modulates phospholipase A2 and blocks up-regulation of MMP-4 and COX-2 induced by TNF and IL-1. Leukotrienes are potent molecules that attract and activate leukocytes and promote their adhesion to vessel walls.
Animal Studies
Numerous animal studies have shown the ability of curcumin to protect against or reverse liver damage induced by hepatic toxins such as carbon tetrachloride, sodium arsenite and other experimental hepatotoxins. Similarly, curcumin significantly reduced the severity of hepatic damage induced by methotrexate, acetaminophen, and multidrug tuberculosis regimens (INH, rifampin, pyrazinamide, ethambutol). In these experiments, methotrexate induced fatty degeneration and necrosis of liver hepatocytes and sinusoidal cells, reduction of levels of the endogenous antioxidants catalase, SOD and GSH and elevation of malondialdehyde, a marker of lipid peroxidation. Administration of curcumin (100mg/kg I.P.) ameliorated all the biochemical abnormalities and histological changes by preserving adequate oxidative status in hepatic tissue.
-
TOXICITY
L-5-methyltetrahydrofolate
No toxicity issues have been reported from folate supplementation. However, care must be taken to assure adequate cobalamin repletion to avoid masking cobalamin deficiency by correcting megaloblastic hematologic abnormalities with folate while allowing progression of neurologic disease due to cobalamin deficiency.
Curcuminoids
No significant curcumin toxicity has been reported. Acute doses as high as 12 gm and chronic doses of 10gm/d have been found to be safe. Dozens of human clinical trials have uniformly demonstrated the lack of toxicity from curcumin. Minor gastrointestinal complaints have occasionally been noted in these trials. A few in vitro studies suggest that curcumin may interfere with platelet aggregation.
-
SPECIAL POPULATIONS
Pregnancy and Nursing Women
The use of RHEUMATE during pregnancy and lactation is up to the individual physician.
There is limited published data on curcumin toxicity in animal models. These data generally do not report any significant adverse events although one rodent study noted slight reduction in neonatal weight. Curcumin toxicity during pregnancy has not been formally studied in humans.
- INDICATIONS AND USAGE
-
PRECAUTIONS AND CONTRAINDICATIONS
Hypersensitivity
RHEUMATE is contraindicated in patients with known hypersensitivity to any of the ingredients in the product. See “ingredients” for a full list of ingredients.
Warnings
Folate may interfere with absorption of tetracycline. Patients receiving both RHEUMATE and tetracycline should take these medications at least one hour apart. Sulfasalazine, phenytoin, statins and some birth control medications may lower levels of serum folate and impair utilization of folate.
-
HOW SUPPLIED
RHEUMATE is supplied in two-part green and yellow capsules with a smooth surface imprinted X502 on one side of the yellow body of the capsule and 52012 on the other side of the yellow body of the capsule supplied as:
Product # Size 68040-750-43 Bottles of 30 capsules 68040-750-08 Carton of 10 capsules as a sample (Not For Resale) Store at room temperature, 59-86°F (15-30°C) [see USP Controlled Room Temperature]. Protect from light and moisture. RHEUMATE is supplied to pharmacies in a recyclable plastic bottle with a child-resistant cap. Dispense in a light-resistant container as defined in the USP/NF with a child-resistant closure.
Dispensed by prescription.
Distributed by:
Primus Pharmaceuticals, Inc.
Scottsdale, AZ 85253(480) 483-1410
www.primusrx.com
www.rheumaterx.com
Patent Nos. 7,736,679; 7,879,373; 7,883,728; 8,153,172; 8,197,869; 8,329,233; 8,623,431; 8,859,020; 8,895.087; 8,993,013
© 2020 Primus Pharmaceuticals, Inc. All rights reserved. 18003-1020 - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
RHEUMATE
l-5-methyltetrahydrofolate, methylcobalamin, curcuminoid turmerone complex capsuleProduct Information Product Type MEDICAL FOOD Item Code (Source) NHRIC:68040-750 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength METHYLCOBALAMIN (UNII: BR1SN1JS2W) (METHYLCOBALAMIN - UNII:BR1SN1JS2W) METHYLCOBALAMIN 1 mg LEVOMEFOLIC ACID (UNII: 8S95DH25XC) (LEVOMEFOLIC ACID - UNII:8S95DH25XC) LEVOMEFOLIC ACID 1 mg CURCUMINOIDS (UNII: R7T2XCO5ZU) (CURCUMINOIDS - UNII:R7T2XCO5ZU) CURCUMINOIDS 500 mg Inactive Ingredients Ingredient Name Strength MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) GELATIN TYPE B BOVINE (150 BLOOM) (UNII: F5AJW0ONK4) MAGNESIUM STEARATE (UNII: 70097M6I30) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) FD&C GREEN NO. 3 (UNII: 3P3ONR6O1S) Product Characteristics Color green, yellow Score no score Shape capsule Size 25mm Flavor Imprint Code X502;52012 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:68040-750-43 30 in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date medical food 03/05/2014 Labeler - Primus Pharmaceuticals, Inc. (130834745)
Trademark Results [Rheumate]
Mark Image Registration | Serial | Company Trademark Application Date |
|---|---|
 RHEUMATE 86098562 4795883 Live/Registered |
Primus Pharmaceuticals, Inc. 2013-10-22 |
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.
