CETAPHIL REDNESS RELIEVING DAILY FACIAL MOISTURIZER WITH SUNSCREEN SPF 40- titanium dioxide, zinc oxide cream
Cetaphil Redness Relieving Daily Facial Moisturizer with sunscreen SPF 40 by
Drug Labeling and Warnings
Cetaphil Redness Relieving Daily Facial Moisturizer with sunscreen SPF 40 by is a Otc medication manufactured, distributed, or labeled by Galderma Laboratories, L.P., Knowlton Development Corporation Inc.. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
- Active Ingredients
- Purpose
- Use(s)
- Warnings
- Keep out of reach of children.
-
Directions
Apply liberally 15 minutes before sun exposure.
Use a water resistant sunscreen if swimming or sweating
Reapply at least every 2 hours.
Children under 6 months: ask a doctor
SUN PROTECTION MEASURES: Spending time in the sun increases your risk of cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
Limit time in the sun, especially 10am – 2 pm.
Wear long-sleeved shirts, pants, hats, and sun glasses. - Other Information
-
Inactive Ingredients
ALLANTOIN, BENZYL ALCOHOL, C12-15 ALKYL BENZOATE, CAFFEINE, CAPRYLYL METHICONE, CETYL DIGLYCERYL TRIS(TRIMETHYLSILOXY) SILYLETHYL DIMETHICONE, CHROMIUM OXIDE GREENS, DICAPRYLYL CARBONATE, DIMETHICONE, DIPOTASSIUM GLYCYRRHIZATE, ETHYLHEXYLGLYCERIN, GLYCERIN, HYDROGENATED LECITHIN, IRON OXIDES, ISOHEXADECANE, MAGNESIUM SULFAGE, MEHYLPROPANEDIOL, SILICA, TOCOPHEROL, TRIETHOXYCAPRYLYLSILANE, TRISILOXANE, WATER
- Questions?
-
PRINCIPAL DISPLAY PANEL - 1.7 FL OZ (50 mL) carton
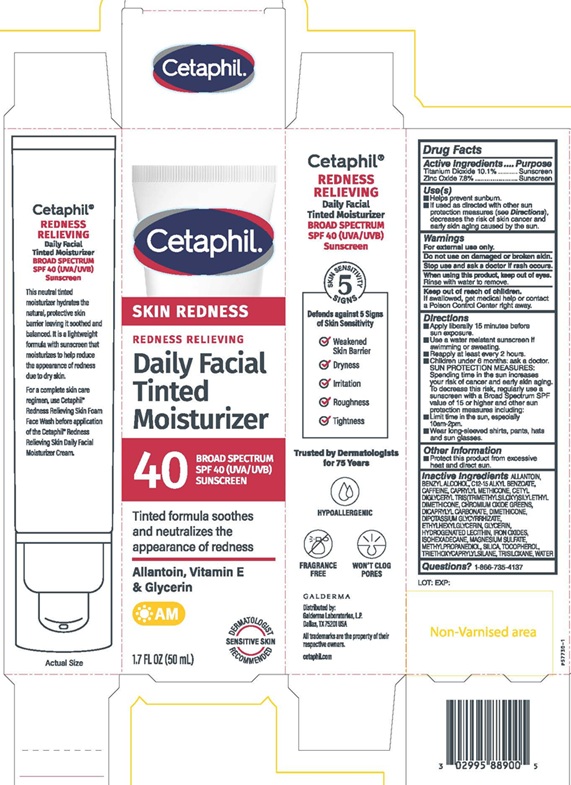
Cetaphil®
SKIN REDNESS
REDNESS RELIEVING
Daily Facial
Tinted
Moisturizer
40 BROAD SPECTRUM
SPF 40 (UVA/UVB)
SUNSCREEN
Tinted formula soothes
and neutralizes the
appearance of redness
Allantoin, Vitamin E
& Glycerin
AM
DERMATOLOGIST
SENSITIVE SKIN
RECOMMENDED
1.7 FL OZ (50 mL)Distributed by:
Galderma Laboratories, L.P.
Dallas, TX 75201 USA
All trademarks are the property of their
respective owners.
cetaphil.com
P57730-1
-
INGREDIENTS AND APPEARANCE
CETAPHIL REDNESS RELIEVING DAILY FACIAL MOISTURIZER WITH SUNSCREEN SPF 40
titanium dioxide, zinc oxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC: 0299-4138 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 101 mg in 1 mL ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC OXIDE - UNII:SOI2LOH54Z) ZINC OXIDE 78 mg in 1 mL Inactive Ingredients Ingredient Name Strength ALLANTOIN (UNII: 344S277G0Z) BENZYL ALCOHOL (UNII: LKG8494WBH) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) CAFFEINE (UNII: 3G6A5W338E) CAPRYLYL TRISILOXANE (UNII: Q95M2P1KJL) CHROMIC OXIDE (UNII: X5Z09SU859) DICAPRYLYL CARBONATE (UNII: 609A3V1SUA) DIMETHICONE (UNII: 92RU3N3Y1O) GLYCYRRHIZINATE DIPOTASSIUM (UNII: CA2Y0FE3FX) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) GLYCERIN (UNII: PDC6A3C0OX) HYDROGENATED SOYBEAN LECITHIN (UNII: H1109Z9J4N) FERRIC OXIDE RED (UNII: 1K09F3G675) ISOHEXADECANE (UNII: 918X1OUF1E) Magnesium Sulfate, Unspecified Form (UNII: DE08037SAB) METHYLPROPANEDIOL (UNII: N8F53B3R4R) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) TOCOPHEROL (UNII: R0ZB2556P8) TRIETHOXYCAPRYLYLSILANE (UNII: LDC331P08E) TRISILOXANE (UNII: 9G1ZW13R0G) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC: 0299-4138-00 1 in 1 CARTON 07/20/2023 1 50 mL in 1 TUBE; Type 0: Not a Combination Product 2 NDC: 0299-4138-05 10 mL in 1 TUBE; Type 0: Not a Combination Product 07/20/2023 11/05/2025 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 07/20/2023 Labeler - Galderma Laboratories, L.P. (047350186) Establishment Name Address ID/FEI Business Operations Port Jervis Laboratories, Inc 001535103 manufacture(0299-4138)
© 2026 FDA.report
This site is not affiliated with or endorsed by the FDA.