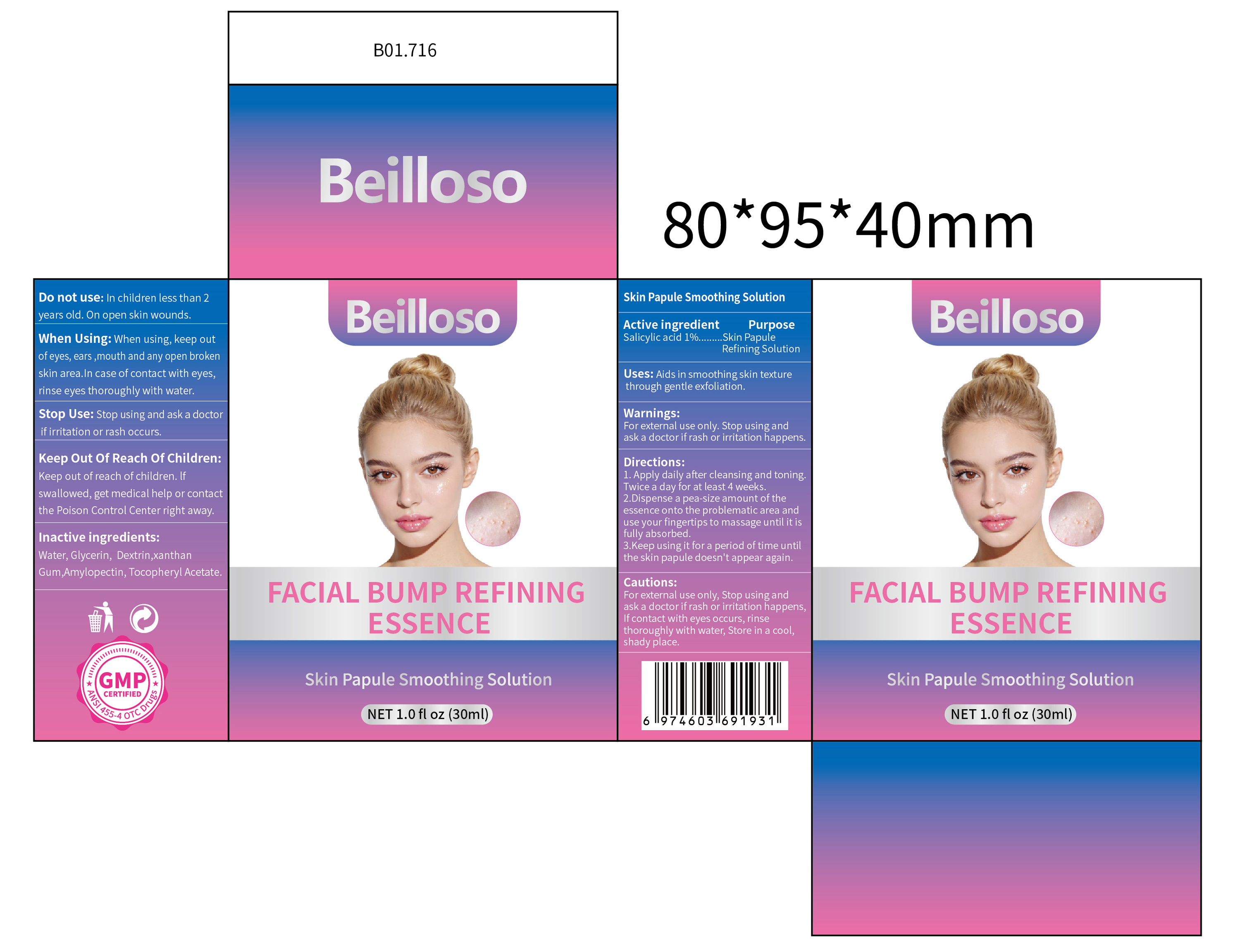
Beilloso FACIAL BUMP REFINING ESSENCE by Jiangxi Hemei Pharmaceutical Co., Ltd 84010-234complete
Beilloso FACIAL BUMP REFINING ESSENCE by
Drug Labeling and Warnings
Beilloso FACIAL BUMP REFINING ESSENCE by is a Otc medication manufactured, distributed, or labeled by Jiangxi Hemei Pharmaceutical Co., Ltd. Drug facts, warnings, and ingredients follow.
Drug Details [pdf]
BEILLOSO FACIAL BUMP REFINING ESSENCE- salicylic acid 1% facial bump refining essence liquid
Jiangxi Hemei Pharmaceutical Co., Ltd
----------
84010-234complete
When Using
When using, keep out of eyes, ears ,mouth and any open broken skin area.In case of contact with eyes, rinse eyes thoroughly with water.
Keep Oot Of Reach Of Children
Keep out of reach of children. lf swallowed, get medical help or contact the Poison Control Center right away.
Directions
1. Apply daily after cleansing and toning.Twice a day for at least 4 weeks. 2.Dispense a pea-size amount of the essence onto the problematic area and use fingertips to massage until it is fully absorbed. 3,Keep using it for a period of time untiL the skin papule doesn't appear again.
| BEILLOSO FACIAL BUMP REFINING ESSENCE
salicylic acid 1% facial bump refining essence liquid |
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
|
||||||||||||||||
| Labeler - Jiangxi Hemei Pharmaceutical Co., Ltd (724892056) |
| Establishment | |||
| Name | Address | ID/FEI | Business Operations |
|---|---|---|---|
| Jiangxi Hemei Pharmaceutical Co., Ltd | 724892056 | manufacture(84010-234) | |